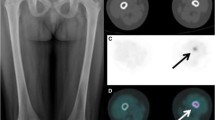
Abstract
Objective
To investigate the value of 18F-FDG PET/CT in diagnosis and disease evaluation of Langerhans cell histiocytosis (LCH).
Methods
A retrospective analysis of 31 patients with LCH confirmed by histopathology was performed. A systematic analysis of the PET/CT imaging manifestations of LCH was performed, recording patients who were treated and receiving PET/CT for efficacy evaluation. In addition, clinical and laboratory data of LCH patients were collected, and the correlation between these data and PET/CT metabolic parameters was initially investigated.
Results
Of the 31 patients, thirty had at least 1 PET/CT positive lesions (96.7%), and one had only skin damage without abnormalities on PET/CT. Of 31 patients, fifteen (48.4%) had single system (SS) disease (9 cases with a single site and 6 cases with multiple sites) and 16 (51.6%) had multisystem (MS) disease (6 low risk and 10 high risk cases). The incidence of LCH lesions in the bone, lymphatic system, pituitary gland, liver, soft tissue, thyroid gland, thymus, and lungs was 20 cases (64.5%), 12 cases (38.7%), 3 cases (9.7%), 2 cases (6.5%), 2 cases (6.5%), 1 case (3.2%), 1 case (3.2%), and 8 cases (25.8%), respectively. A total of 21 PET/CT follow-up scanning were performed in 13 patients receiving chemotherapy, with 13 (61.9%) partial metabolic remission (PMR), 6 (28.6%) progressive metabolic disease (PMD), and 2 (9.5%) stable metabolic disease (SMD), according to PET Response Evaluation Criteria in Solid Tumors (PRECIST) 1.0. Erythrocyte sedimentation rate (ESR), C-reactive protein (CRP) and Lactic Dehydrogenase (LDH) were positively correlated with TTLG (total TLG) (R2 = 0.3256, 0.2409, 0.4205, P < 0.05). The Re-examine SUVmax is positively correlated with re-examine LDH (R2 = 0.7285, P < 0.05).
Conclusions
18F-FDG PET/CT is an effective way to diagnose and evaluate LCH. PET metabolic parameters were associated with laboratory inflammatory markers, suggesting that 18F-FDG PET/CT may be helpful in evaluating disease activity of LCH.



Similar content being viewed by others
Data availability
All relevant data are within the paper.
References
Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379(9):856–68.
Binkovitz LA, Olshefski RS, Adler BH. Coincidence FDG-PET in the evaluation of Langerhans’ cell histiocytosis: preliminary findings. Pediatr Radiol. 2003;33(9):598–602.
Kaste SC, Rodriguez-Galindo C, McCarville ME, Shulkin BL. PET-CT in pediatric Langerhans cell histiocytosis. Pediatr Radiol. 2007;37(7):615–22.
Phillips M, Allen C, Gerson P, McClain K. Comparison of FDG-PET scans to conventional radiography and bone scans in management of Langerhans cell histiocytosis. Pediatr Blood Cancer. 2009;52(1):97–101.
Lee HJ, Ahn BC, Lee SW, Lee J. The usefulness of F-18 fluorodeoxyglucose positron emission tomography/computed tomography in patients with Langerhans cell histiocytosis. Ann Nucl Med. 2012;26(9):730–7.
Zhou W, Wu H, Han Y, Wang S, Dong Y, Wang Q. Preliminary study on the evaluation of Langerhans cell histiocytosis using F-18-fluoro-deoxy-glucose PET/CT. Chin Med J (Engl). 2014;127(13):2458–62.
Garcia JR, Riera E, Bassa P, Mourelo S, Soler M. (18)F-FDG PET/CT in follow-up evaluation in pediatric patients with Langerhans histiocytosis. Rev Esp Med Nucl Imagen Mol. 2017;36(5):325–8.
Zhao Y, Zhou Y, Tian R, Su M. Solitary bone Langerhans cell histiocytosis demonstrated on multimodality imaging in an adult. Clin Nucl Med. 2020;45(1):78–80.
Xu T, Cen B, Li H, Wang Y, Chen Y. 99m Tc-MDP bone scan findings of recurrent Langerhans cell histiocytosis in an adult involving femur after joint replacement. Clin Nucl Med. 2023;48(9):804–5.
Rodriguez-Galindo C, Allen CE. Langerhans cell histiocytosis. Blood. 2020;135(16):1319–31.
Pinker K, Riedl C, Weber WA. Evaluating tumor response with FDG PET: updates on PERCIST, comparison with EORTC criteria and clues to future developments. Eur J Nucl Med Mol Imaging. 2017;44(Suppl 1):55–66.
Berres ML, Lim KP, Peters T, Price J, Takizawa H, Salmon H, et al. BRAF-V600E expression in precursor versus differentiated dendritic cells defines clinically distinct LCH risk groups. J Exp Med. 2014;211(4):669–83.
Laman JD, Leenen PJ, Annels NE, Hogendoorn PC, Egeler RM. Langerhans-cell histiocytosis “insight into DC biology.” Trends Immunol. 2003;24(4):190–6.
Senechal B, Elain G, Jeziorski E, Grondin V, Patey-Mariaud de Serre N, Jaubert F, et al. Expansion of regulatory T cells in patients with Langerhans cell histiocytosis. PLoS Med. 2007;4(8):e253.
Allen CE, Li L, Peters TL, Leung HC, Yu A, Man TK, et al. Cell-specific gene expression in Langerhans cell histiocytosis lesions reveals a distinct profile compared with epidermal Langerhans cells. J Immunol. 2010;184(8):4557–67.
Wang J, Song T, Wang J, Ma L, Jiang Y, Kong D, et al. Aggressive Langerhans cell histiocytosis transformation of T cell acute lymphoblastic leukemia detected on (18)F-FDG PET/CT. Eur J Nucl Med Mol Imaging. 2021;48(2):642–3.
Luo ZH, Lu PX, Qi WL, Liao FX, Jin AF, Zen QY. Role of (18)F-FDG PET/CT in the diagnosis and management of patients with Langerhans cell histiocytosis. Quant Imaging Med Surg. 2022;12(6):3351–63.
Albano D, Bosio G, Giubbini R, Bertagna F. Role of (18)F-FDG PET/CT in patients affected by Langerhans cell histiocytosis. Jpn J Radiol. 2017;35(10):574–83.
Kobayashi M, Ando S, Kawamata T, Makiyama J, Yokoyama K, Imai Y, et al. Clinical features and outcomes of adult Langerhans cell histiocytosis: a single-center experience. Int J Hematol. 2020;112(2):185–92.
Agarwal KK, Seth R, Behra A, Jana M, Kumar R. 18F-Fluorodeoxyglucose PET/CT in Langerhans cell histiocytosis: spectrum of manifestations. Jpn J Radiol. 2016;34(4):267–76.
Ohshima K, Fujiya K, Nagashima T, Ohnami S, Hatakeyama K, Urakami K, et al. Driver gene alterations and activated signaling pathways toward malignant progression of gastrointestinal stromal tumors. Cancer Sci. 2019;110(12):3821–33.
Gatenby RA, Gillies RJ. Why do cancers have high aerobic glycolysis? Nat Rev Cancer. 2004;4(11):891–9.
McCleland ML, Adler AS, Deming L, Cosino E, Lee L, Blackwood EM, et al. Lactate dehydrogenase B is required for the growth of KRAS-dependent lung adenocarcinomas. Clin Cancer Res. 2013;19(4):773–84.
de la Cruz-Lopez KG, Castro-Munoz LJ, Reyes-Hernandez DO, Garcia-Carranca A, Manzo-Merino J. Lactate in the regulation of tumor microenvironment and therapeutic approaches. Front Oncol. 2019;9:1143.
Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest. 2003;111(12):1805–12.
Zer A, Domachevsky L, Rapson Y, Nidam M, Flex D, Allen AM, et al. The Role of 18F-FDG PET/CT on staging and prognosis in patients with small cell lung cancer. Eur Radiol. 2016;26(9):3155–61.
Kwon HR, Cho J, Park S, Lee SH, Ahn MJ, Choi JY, et al. Metabolic parameters on baseline (18)F-FDG PET/CT are potential predictive biomarkers for immunotherapy in patients with head and neck squamous cell carcinoma. Front Med (Lausanne). 2022;9: 896494.
Lee JW, Kang CM, Choi HJ, Lee WJ, Song SY, Lee JH, et al. Prognostic value of metabolic tumor volume and total lesion glycolysis on preoperative (1)(8)F-FDG PET/CT in patients with pancreatic cancer. J Nucl Med. 2014;55(6):898–904.
Burger IA, Casanova R, Steiger S, Husmann L, Stolzmann P, Huellner MW, et al. 18F-FDG PET/CT of non-small cell lung carcinoma under neoadjuvant chemotherapy: background-based adaptive-volume metrics outperform TLG and MTV in predicting histopathologic response. J Nucl Med. 2016;57(6):849–54.
Funding
The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal
Not applicable.
Ethical approval
Not applicable.
Consent for participating and for publication
Patients signed an informed consent to participate and to use their anonymous data for analysis and publication of results.
Consent informed
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
An, R., Ma, X. & Wang, Y. The value of 18F-FDG PET/CT in Langerhans cell histiocytosis. Ann Nucl Med 38, 238–245 (2024). https://doi.org/10.1007/s12149-023-01892-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-023-01892-3