Abstract
Background and aims
Because the outcome of glioblastoma multiforme (GBM) remains dismal, there is an urgent need for a better molecular characterization of this malignancy. The aim of this prospective study was to investigate the prognostic impact of the expression of c-mesenchymal-epithelial transition (c-Met) a receptor tyrosine kinase implicated in expression growth, survival, motility/migration, and invasion in GMB patients managed according to the established diagnostic and therapeutic protocols.
Methods
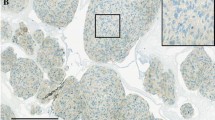
Between May 2003 and March 2011, a total of 69 patients (33 males and 36 females; mean age: 52.2 ± 12.9 years, age range: 23–81 years) referred to our Department for the surgical removal of GBM were evaluated immunohistochemically for c-Met expression. Progression-free survival (PFS) and overall survival (OS) served as the main outcome measures.
Results
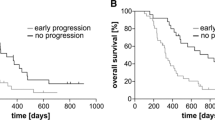
Compared with c-Met− subjects (n = 38), c-Met+ subjects (n = 31) had both a significantly lower OS (15.3 ± 2.3 vs. 22.6 ± 2.5 months, respectively, p < 0.01) and PFS (12.3 ± 2.1 vs. 19.1 ± 2.6 months, respectively, p < 0.05). After allowance for potential confounders, multivariate Cox regression analysis identified c-Met+ as an independent predictor of both OS (hazard ratio = 1.7; 95 % confidence interval = 1.2–1.9, p < 0.01) and PFS (hazard ratio = 1.6; 95 % confidence interval = 1.1–2.3, p < 0.05).
Conclusions
Our findings suggest that c-Met immunohistochemical expression is an independent predictor of outcomes in patients with GBM treated by standard of care.


Similar content being viewed by others
References
Rekers NH, Sminia P, Peters GJ. Towards tailored therapy of glioblastoma multiforme. J Chemother. 2011;23:187–99.
Gilbert CA, Daou MC, Moser RP, Ross AH. Gamma-secretase inhibitors enhance temozolomide treatment of human gliomas by inhibiting neurosphere repopulation and xenograft recurrence. Cancer Res. 2010;70:6870–9.
Tran B, Rosenthal MA. Survival comparison between glioblastoma multiforme and other incurable cancers. J Clin Neurosci. 2010;17:417–21.
Zhang X, Zhang W, Cao WD, Cheng G, Zhang YQ. Glioblastoma multiforme: molecular characterization and current treatment strategy (review). Exp Ther Med. 2012;3:9–14.
Kanu OO, Mehta A, Di C, Lin N, Bortoff K, Bigner DD, et al. Glioblastoma multiforme: a review of therapeutic targets. Exp Opin Ther Targ. 2009;13:701–18.
Masui K, Cloughesy TF, Mischel PS. Molecular pathology in adult high-grade gliomas from molecular diagnostics to target therapies. Neuropathol Appl Neurobiol. 2012;38:271–91.
Jung KH, Park BH, Hong SS. Progress in cancer therapy targeting c-Met signaling pathway. Arch Pharm Res. 2012;35:595–604.
Gherardi E, Birchmeier W, Birchmeier C, Vande Woude G. Targeting MET in cancer: rationale and progress. Nat Rev Cancer. 2012;12:89–103.
Cañadas I, Rojo F, Arumí-Uría M, Rovira A, Albanell J, Arriola E. C-MET as a new therapeutic target for the development of novel anticancer drugs. Clin Transl Oncol. 2010;12:253–60.
Yap TA, Sandhu SK, Alam SM, de Bono JS. HGF/c-MET targeted therapeutics: novel strategies for cancer medicine. Curr Drug Targ. 2011;12:2045–58.
Shiota G, Kawasaki H, Nakamura T. Coexpression of hepatocyte growth factor and its receptor (c-Met oncogene) in HGL4 glioblastoma cells. Oncology. 1996;53:511–6.
Uchinokura S, Miyata S, Fukushima T, Itoh H, Nakano S, Wakisaka S, et al. Role of hepatocyte growth factor activator (HGF activator) in invasive growth of human glioblastoma cells in vivo. Int J Cancer. 2006;118:583–92.
Liu W, Fu Y, Xu S, Ding F, Zhao G, Zhang K, et al. c-Met expression is associated with time to recurrence in patients with glioblastoma multiforme. J Clin Neurosci. 2011;18:119–21.
Xie Q, Bradley R, Kang L, Koeman J, Ascierto ML, Worschech A, et al. Hepatocyte growth factor (HGF) autocrine activation predicts sensitivity to MET inhibition in glioblastoma. Proc Natl Acad Sci USA. 2012;109:570–5.
Welsh JW, Mahadevan D, Ellsworth R, Cooke L, Bearss D, Stea B. The c-Met receptor tyrosine kinase inhibitor MP470 radiosensitizes glioblastoma cells. Radiat Oncol. 2009;4:69.
Guessous F, Zhang Y, diPierro C, Marcinkiewicz L, Sarkaria J, Schiff D, et al. An orally bioavailable c-Met kinase inhibitor potently inhibits brain tumor malignancy and growth. Anticancer Agents Med Chem. 2010;10:28–35.
Buchanan SG, Hendle J, Lee PS, Smith CR, Bounaud PY, Jessen KA, et al. SGX523 is an exquisitely selective, ATP-competitive inhibitor of the MET receptor tyrosine kinase with antitumor activity in vivo. Mol Cancer Ther. 2009;8:3181–90.
Terzolo M, Boccuzzi A, Bovio S, Cappia S, De Giuli P, Alì A, et al. Immunohistochemical assessment of Ki-67 in the differential diagnosis of adrenocortical tumors. Urology. 2001;57:176–82.
Hou XZ, Liu W, Fan HT, Liu B, Pang B, Xin T, et al. Expression of hepatocyte growth factor and its receptor c-Met in human pituitary adenomas. Neurol Oncol. 2010;12:799–803.
Minoretti P, Falcone C, Calcagnino M, Emanuele E, Buzzi MP, Coen E, et al. Prognostic significance of plasma osteopontin levels in patients with chronic stable angina. Eur Heart J. 2006;27:802–7.
Huang TJ, Wang JY, Lin SR, Lian ST, Hsieh JS. Overexpression of the c-met protooncogene in human gastric carcinoma—correlation to clinical features. Acta Oncol. 2001;40:638–43.
Kong DS, Song SY, Kim DH, Joo KM, Yoo JS, Koh JS, et al. Prognostic significance of c-Met expression in glioblastomas. Cancer. 2009;115:140–8.
Christensen JG, Burrows J, Salgia R. c-Met as a target for human cancer and characterization of inhibitors for therapeutic intervention. Cancer Lett. 2005;225:1–26.
Liu X, Wang Q, Yang G, Marando C, Koblish HK, Hall LM, et al. A novel kinase inhibitor, INCB28060, blocks c-MET-dependent signaling, neoplastic activities, and cross-talk with EGFR and HER-3. Clin Cancer Res. 2011;17:7127–38.
Barker FG 2nd, Chang SM, Larson DA, Sneed PK, Wara WM, Wilson CB, et al. Age and radiation response in glioblastoma multiforme. Neurosurgery. 2001;49:1288–97.
Pierga JY, Hoang-Xuan K, Feuvret L, Simon JM, Cornu P, Baillet F, et al. Treatment of malignant gliomas in the elderly. J Neurooncol. 1999;43:187–93.
Whittle IR, Basu N, Grant R, Walker M, Gregor A. Management of patients aged >60 years with malignant glioma: good clinical status and radiotherapy determine outcome. Br J Neurosurg. 2002;16:343–7.
Lamborn KR, Chang SM, Prados MD. Prognostic factors for survival of patients with glioblastoma: recursive partitioning analysis. Neurol Oncol. 2004;6:227–35.
Stewart LA. Chemotherapy in adult high-grade glioma: a systematic review and meta-analysis of individual patient data from 12 randomised trials. Lancet. 2002;359:1011–8.
Conflict of interest
The authors declare that they have no conflict of interest relating to the publication of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Olmez, O.F., Cubukcu, E., Evrensel, T. et al. The immunohistochemical expression of c-Met is an independent predictor of survival in patients with glioblastoma multiforme. Clin Transl Oncol 16, 173–177 (2014). https://doi.org/10.1007/s12094-013-1059-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-013-1059-4