Abstract
Pulmonary mucormycosis (PM) is a rare disease. Literature about the surgical management of PM in India is sparse. Recently, there has been a surge in the number of cases of mucormycosis in association with the coronavirus disease 2019 (COVID-19) pandemic, igniting the interest in PM. Hence, we endeavoured to analyse our surgical experience in treating PM prior to the pandemic, in a tertiary care centre, to provide insight into this disease. Data of 10 adult patients with isolated PM, who underwent surgery between the years 2009 and 2020, and maintained in our departmental database, were retrieved and analysed.
Similar content being viewed by others
Introduction
Mucormycosis is a rare but life-threatening opportunistic mycosis, which usually affects immunocompromised individuals. It is caused by broad aseptate fungi belonging to the subphylum Mucoromycotina, order Mucorales [1]. Recently, this pathogen has come under intense focus, due to its association with the coronavirus disease 2019 (COVID-19) pandemic [2]. The lung is the third most frequently involved site after rhino-orbito-cerebral and cutaneous involvements [3]. Pulmonary mucormycosis (PM) is known for its aggressive clinical course with a mortality rate approaching 55% with medical management alone [4]. A combination of prompt diagnosis, appropriate medical therapy and early aggressive surgical intervention could reduce the mortality to 25% [3, 4]. The surgeries described for PM include lobectomy, wedge resection and pneumonectomy [4]. Only a few such surgically managed cases have been reported from India prior to the pandemic [5,6,7,8,9]. Now since this rare entity has ignited the interest of the world, we found a need to review our past surgical experience with PM in our tertiary care centre.
Description of cases
The protocols followed for pre-operative workup, antifungal therapy and surgical management, which were common to all cases, are presented below.
Pre-operative workup
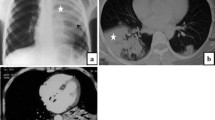
Patients with a clinical, radiological, microbiological or pathological suspicion of localised PM were advised surgery, if they were fit for surgery. Baseline blood tests were done to identify comorbidities. The chest x-ray and contrast-enhanced computed tomogram (CECT) helped to diagnose and characterise the lesion, and to ascertain the condition of the rest of the lung, which helped in deciding the possibility and the extent of the surgery. On CECT, PM appears as an intra-cavitatory mass with the reverse halo sign, also known as the atoll sign (Fig. 1a), which is defined as central ground glass opacity, surrounded by denser crescent-shaped sliver of pulmonary consolidation forming more than three-fourths of a circle or a complete ring of at least 2 mm thickness, and at times, with an air-fluid level (Fig. 1b). Other pathologies like an aspergilloma or a cavitating neoplasm can mimic it radiologically (Fig. 1c, d). A microbiological study of the sputum was done to identify any superadded bacterial infection and to rule out active tuberculosis. All patients underwent pulmonary function test (PFT) to help decide the feasibility of the planned resection. Bronchoscopy was done to study the bronchial anatomy, intra-bronchial spread and any associated incidental endobronchial pathology and to rule out active tuberculosis (TB). Broncho-alveolar lavage (BAL) cultures and trans-bronchial lung biopsy (TBLB) were done in all patients. The pre-operative management included nutritional buildup, cessation of smoking, control of diabetes mellitus, blood transfusions if the haemoglobin level was less than 9 gm%, treatment of any other comorbidities if any, and intensive physiotherapy. Any steroid or immunosuppressant medication was tapered down to the lowest dosage possible.
a CT of a patient with PM showing atoll sign (arrow). b Chest radiograph of the same patient showing right lower lobe lesion with air-fluid level (arrow). c Fungal ball with air crescent sign (arrow) mimicking an aspergilloma. d Cavitatory lesion (arrow) mimicking a malignancy. CT, computed tomography; PM, pulmonary mucormycosis
Antifungal protocol
Intravenous amphotericin B was the commonly used antifungal agent. It was started pre-operatively, if mucormycosis was diagnosed in BAL or TBLB. During amphotericin B therapy, the patients were kept sufficiently hydrated and the renal functions were monitored twice a week. After the surgery, amphotericin B was continued for 2 more weeks, concurrently with 6 weeks of oral posaconazole. Further continuation of antifungals was decided by assessing clinical improvement, C-reactive protein levels and radiological findings.
Criteria for surgical management
Only patients with PM localised to a single lobe were advised surgery. The surgery was expedited without any delay, if the PM was localised to a single lobe on diagnosis. Patients with extensive multi-lobar or bilateral disease were started on amphotericin B and were reassessed with weekly computed tomography (CT). If and when the disease got localised to a resectable level, surgery was undertaken. If the disease did not get localised, even after a cumulative dose of 4 g of amphotericin, they were discharged on posaconazole and were advised to follow up every month to assess the disease with a CT. The institutional protocol for surgical procedure and post-operative management of lobectomy has been reported in our previous publication [10]. Patients were asked to follow up at 6 months and then at yearly intervals.
Patient details
There were a total of 10 adult patients who underwent lung resection for isolated PM. The average age was 44 years (range—20 to 56). The male to female ratio was 2.3:1. The right lower lobe (RLL) was most commonly involved in 4 (40%) patients, followed by the right upper lobe in 2 (20%), left upper lobe in 2 (20%) and left lower and middle lobes in 1 (10%) patient each. Most of the patients, 6 (60%), had presented with intermittent haemoptysis. Other commonly reported symptoms were cough in 2 (20%) patients and fever in 2 (20%) patients. The average duration of symptoms was 21 months (range—1 to 120). Anaemia was the most common comorbidity seen in 6 (60%) patients. The other common comorbidities were diabetes mellitus in 5 (50%) patients, hypoproteinemia in 3 (30%) and chronic kidney disease (CKD) in 3 (30%) patients. Two patients had previous history of tuberculosis. Three (30%) patients were on steroids. Out of the three, one was a renal allograft recipient, one had CKD due to IgA nephropathy and one had pyoderma gangrenosum. The average duration of steroid intake was 3.3 months (range—2 to 6 months). The renal allograft recipient had received his transplant 1 month earlier and was also on immunosuppressive therapy with tacrolimus for 1 month. One patient had history of prior resection of a contra-lateral lobe for mucormycosis in a different centre. The mean haemoglobin level of the patients was 10.5 ± 2.2 g/dl. The average serum creatinine level was 1.23 ± 0.7 mg/dl. The mean serum albumin was 3.5 ± 0.6 g/dl. The average forced expiratory volume in the first second (FEV1) was 2.29 l. Bronchoscopy revealed normal bronchial anatomy in all and an occasional minimal traction-induced distortion. BAL cultures grew Rhizopus spp. (mucormycosis) in 4 (40%) and Klebsiella pneumonia in 2 (20%) patients. Other organisms encountered were Haemophilus influenza, Acinetobactor baumani and Morganella morgagni in one patient each. TBLB histopathology revealed mucormycosis in 4 (40%) patients. Three out of these 4 patients had grown fungus in their BAL cultures also. Overall, of the 5 (50%) patients diagnosed pre-operatively, 3 had evidence of mucor, both in BAL and in TBLB, and the other 2 had evidence either in BAL or in TBLB. Lobectomy by a thoracotomy was done in all the patients. The average operative time was 162 ± 45 min. The mean intra-operative blood loss was 324 ± 88 ml. Post-operative tissue cultures grew Rhizopus spp. in 5 (50%) patients (Fig. 2a, b). In addition, one patient grew Mycobacterium fortitium and another grew Klebsiella pneumonia. Histopathology confirmed the diagnosis in all the patients. Tissue or angio invasion was seen in all of the patients (Fig. 2c, d). There were 3 (30%) post-operative complications and a single (10%) in-hospital mortality. The complications were prolonged air leak in 2 (20%) and empyema in 1 (10%) patient. The mean post-operative hospital stay was 9.8 days (range—5 to 30). The mean follow-up in our series was 2.5 years (range 1–8 years). There was one late death. Two patients on follow-up developed disseminated mucormycosis after about 1 year.
a Direct microscopy from tissue with calcoflour white fluorescence stain showing broad aseptate hyphae (40 ×). b Lactophenol cotton blue preparation showing Rhizopus sporangiophore (black arrow), rhizoids (red star), sporangium and spores (red arrows) (40 ×). c Photomicrograph displaying broad aseptate fungal hyphae (star) in the wall of a necrotic vessel, periodic acid-Schiff (PAS) (40 ×). d Photomicrograph displaying broad aseptate fungal hyphae (star) in the wall of a necrotic vessel, Gomori methenamine silver (GMS) (40 ×)
Discussion
Our series of patients is one of the largest single-centre surgical experiences from India. Epidemiological studies from India are largely single-centre experiences, and hence, the real incidence of the disease is not clearly known. The largest single-centre experience was from Mayo Clinic, which reported 35 cases over a period of 15 years, and included patients managed medically and surgically [11]. The characteristics of patients who required surgical management in India are largely unknown. Our study could provide an insight into this rare disease which has resurfaced in large numbers now. The average age of the patient (44 years) and the sex ratio (2.3:1) were comparable to other studies [3, 4, 12]. The most commonly involved lobe was the right lower lobe, in contrast to other diseases by fungi like aspergillus species, which primarily involve the upper lobes [10]. This was because aspergillus infections or infestations often developed in pre-existent tubercular cavities, which are more common in the upper lobes [13]. The conidia (mitospores) of Mucorales probably preferentially settled down in the right lower lobe upon inhalation, since its bronchus is in line with the trachea and has more of zone 3 ventilation, and due to the effect of gravity [14, 15]. In healthy individuals, these spores are eliminated by phagocytes in the lungs and the respiratory tract [16, 17]. Hyperglycaemia and acidosis are known to impair chemotaxis and the killing activity of phagocytic cells by impairing its oxidative and non-oxidative mechanisms of phagocytosis [16, 17]. Corticosteroids impair migration, ingestion, and phagolysosome fusion in human macrophages. This explains the high incidence of mucormycosis in diabetics and in patients on steroids. However, the exact molecular mechanism that causes attenuation of phagocytic function in patients with hyperglycaemia, ketoacidosis or steroids is not well understood. In our study, 5 (50%) patients had diabetes mellitus and 3 (30%) patients were on steroids. Anaemia was common in our cohort, which was not observed in other studies. This may be because of the long-standing haemoptysis, the higher incidence of CKD (30%) or the chronic disease itself. Three patients had neutropenia (< 1500 cells/mm3), including one renal allograft recipient on immunosuppressive therapy. The reasons for neutropenia in the other two patients were not clear. Literature reports neutropenia as a risk factor for mucormycosis in patients on immunosuppressive agents or cancer chemotherapy [14]. Neutrophils also act in a way similar to that of macrophages to combat fungal invasion [18]. Classically, the duration of symptoms in mucormycosis was supposed to be very short (< 6 months) [14], but our patients were symptomatic for an average of 21 months (range—1 to 120). This was probably because of the association of PM with TB in our series in 2 (20%) patients, aspergillosis in 1 (10%) patient and both in another 2 (20%) of patients. Aspergilloma and TB have chronic courses and these patients might have developed superadded mucormycosis only recently. If patients with aspergilloma and TB were excluded, the average duration of symptoms in our cohort was only 5 months. Haematological malignancies and solid organ transplants, like kidney and lung transplants, are other common risk factors quoted in other studies [4, 11], but our study had only one patient who was a renal allograft recipient. BAL cultures and TBLB had low positivity (40% each) for fungus. This might be because of sampling error, due to delayed plating or fragmentation of the fungal elements during tissue processing for biopsy [19]. Rhizopus spp. were the most common fungus isolated both pre-operatively (40%) and post-operatively (50%), as in other studies [3]. Pre-operative initiation of antifungal agents healed areas of minor fungal involvement in other parts of the lungs, localising the disease to the worst affected lobe with refractory disease, thus enabling a safe and complete resection, as demonstrated in one patient (Fig. 3a, b). Two patients developed acute kidney injury due to the amphotericin B and were managed successfully with increased hydration and adjustment of the dose. Posaconazole, an orally broad-spectrum triazole, is the only drug of this class that appears to possess potent activity against Mucorales. However, long-term use of this drug could lead to gastro-intestinal disturbances and cause various drug interactions. One of our operated patients, who had previously undergone a lingulectomy in a different centre for mucormycosis, presented to us 12 months later, with right upper lobe mucormycosis. This patient had been treated with amphotericin B for 2 months and posaconazole for 6 months after the first surgery. This could suggest that even with appropriate antifungal therapy, mucormycosis is notorious for recurrence and dissemination. All our patients underwent a lobectomy to remove the entire diseased lobe. Since mucormycosis is invasive, it is paramount to remove the whole lobe to avoid leaving behind infected lung tissue and to prevent dissemination. Post-operative tissue biopsy was confirmative in all patients while the post-operative cultures were positive only in 50%. Out of the 5 (50%) patients who received pre-operative antifungal therapy, only one patient grew mucormycetes in the post-operative culture. The single mortality was due to refractory fungal and bacterial septicaemia. This patient had multiple comorbidities like anaemia, diabetes mellitus and pyoderma gangrenosum, for which she was on long-term steroid therapy. The post-surgical mortality of 10% and morbidity of 30% are higher than those for other fungal pulmonary involvements like aspergilloma, since aspergillus involvement is infrequently invasive [10]. It was observed that if a patient had chronic steroid intake or hypoproteinemia as a risk factor, it resulted definitely (100%) in an adverse surgical outcome (post-operative complication or in-hospital mortality) (Table 1). This suggests that steroids have to be tapered to the lowest acceptable dose, and stopped if possible, prior to surgery. This might not be feasible in allograft recipients. Chronic steroid intake and hypoproteinemia lead to poor bronchial stump, pulmonary and wound healings, resulting in complications like prolonged air leak, empyema and surgical site infections. Patients who had an acute infection (< 3 months) and lower lobar involvement had a higher (80%) chance of developing an adverse outcome after surgery (Table 1). Aggressive disease would have presented early and might have poorer outcomes as seen in patients with shorter duration of pre-operative symptoms. Lower lobes were commonly involved, and hence, the number of complications was also higher in that anatomical subset. Patients with diabetes mellitus or CKD had a 60–70% chance of an adverse outcome (Table 1). It is common knowledge that poor glycaemic control in the peri-operative period results in poor outcomes after any surgery [20]. There was an unrelated late mortality, 3 years after surgery, due to COVID-19. The major limitation of our study was the inadequate follow-up.
Conclusion
PM is rare and it is life-threatening, unless aggressive management is undertaken. A combination of medical and surgical management yields best results. Diabetes mellitus and chronic steroid intake are the common risk factors for acquiring PM. Optimisation of pre-operative risk factors like diabetes mellitus, steroid intake and hypoproteinemia would improve outcomes after surgery. Increasing incidence of mucormycosis due to the current pandemic makes this study important.
References
Ibrahim AS, Spellberg B, Walsh TJ, Kontoyiannis DP. Pathogenesis of mucormycosis. Clin Infect Dis. 2012;54:S16-22.
John TM, Jacob CN, Kontoyiannis DP. When uncontrolled diabetes mellitus and severe COVID-19 converge: the perfect storm for mucormycosis. J Fungi (Basel). 2021;7:298.
Manesh A, Rupali P, Sullivan MO, et al. Mucormycosis- a clinicoepidemiological review of cases over 10 years. Mycoses. 2019;62:391–8.
Lee FY, Mossad SB, Adal KA. Pulmonary mucormycosis: the last 30 years. Arch Intern Med. 1999;159:1301–9.
Agarwal R, Kumar V, Gupta D. Pulmonary mucormycosis: two of a kind. Eur J Intern Med. 2006;17:63–5.
Garg R, Marak RS, Verma SK, Singh J, Sanjay, Prasad R. Pulmonary mucormycosis mimicking as pulmonary tuberculosis: a case report. Lung India. 2008;25:129–31.
Mani RK, Mishra V, Sharma M, Kumar RA. Isolated pulmonary mucormycosis. BMJ Case Rep. 2017 ;2017:bcr2017219342.
Aggarwal D, Chander J, Janmeja AK, Katyal R. Pulmonary tuberculosis and mucormycosis co-infection in a diabetic patient. Lung India. 2015;32:53–5.
Panigrahi MK, Manju R, Kumar SV, Toi PC. Pulmonary mucormycosis presenting as nonresolving pneumonia in a patient with diabetes mellitus. Respir Care. 2014;59:e201–5.
Sameer M, David N, Rao VM, et al. Surgical management of pulmonary aspergilloma—12 years’ experience from a tertiary care centre in India. Indian J Thorac Cardiovasc Surg. 2021;37:402–10.
Lin E, Moua T, Limper AH. Pulmonary mucormycosis: clinical features and outcomes. Infection. 2017;45:443–8.
Priya P, Ganesan V, Rajendran T, Geni VG. Mucormycosis in a tertiary care center in South India: a 4-year experience. Indian J Crit Care Med. 2020;24:168–71.
Ong CWM, Elkington PT, Friedland JS. Tuberculosis, pulmonary cavitation, and matrix metalloproteinases. Am J Respir Crit Care Med. 2014;190:9–18.
Hamilos G, Samonis G, Kontoyiannis DP. Pulmonary mucormycosis. Semin Respir Crit Care Med. 2011;32:693–702.
Powers KA, Dhamoon AS. Physiology, pulmonary ventilation and perfusion. [Updated 2021 Mar 24]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK539907/
Spellberg B, Edwards J Jr, Ibrahim A. Novel perspectives on mucormycosis: pathophysiology, presentation, and management. Clin Microbiol Rev. 2005;18:556–69.
Kontoyiannis DP, Lewis RE. Invasive zygomycosis: update on pathogenesis, clinical manifestations, and management. Infect Dis Clin North Am. 2006;20:581–607.
Ghuman H, Voelz K. Innate and adaptive immunity to mucorales. J Fungi (Basel). 2017;3:48.
Reichenberger F, Habicht J, Matt P, et al. Diagnostic yield of bronchoscopy in histologically proven invasive pulmonary aspergillosis. Bone Marrow Transplant. 1999;24:1195–9.
Duggan EW, Carlson K, Umpierrez GE. Perioperative hyperglycemia management: an update. Anesthesiology. 2017;126:547–60.
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Santhosh Regini Benjamin, Deepak Narayanan and Sujith Thomas Chandy. The first draft of the manuscript was written by Santhosh Regini Benjamin and all authors commented on previous versions of the manuscript before the final one. Microbiological images and data were provided by Joy Sarojini Michael. Pathological images and data were provided by Thomas Alex Kodiatte. The final manuscript was written by Santhosh Regini Benjamin and Birla Roy Gnanamuthu. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The approval of the institutional review board has been obtained.
Informed consent
Written consent for studies and publication was obtained from the patients prior to the surgery.
Statement of human and animal rights
The study has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Benjamin, S.R., Narayanan, D., Chandy, S.T. et al. Pulmonary mucormycosis—a case series. Indian J Thorac Cardiovasc Surg 38, 177–182 (2022). https://doi.org/10.1007/s12055-021-01272-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12055-021-01272-4