Abstract
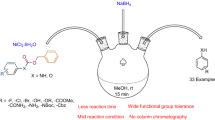
An efficient one-pot procedure for the direct reductive amination of aldehyde and ketones was achieved in the presence of sodium borohydride by using B(OSO3H)3/SiO2(SBSA) as the reusable solid catalyst in acetonitrile and solvent-free conditions. Both aromatic and aliphatic aldehyde reacted well to give the corresponding amines in excellent yields. All the products are known and well-characterized. The catalyst is recoverable and could be easily recycled by filtration and reused several times without any significant loss of its activity. SBSA acts as a dual Brønsted/Lewis acid that is an air-stable and cost-effective solid acid.

An efficient one-pot procedure for the direct reductive amination of aldehyde and ketones in the presence of sodium borohydride by using B(OSO3H)3/SiO2(SBSA) as the reusable solid catalyst in acetonitrile and under solvent-free conditions is described.



Similar content being viewed by others
References
Hutchins R O and Hutchins M K 1991 In Comprehensive Organic Synthesis B M Trost and I Fleming (Eds.) Vol. 8 (Oxford: Pergamon) p. 25
Nguyen Q P B and Kim T H 2013 Solvent- and catalyst-free direct reductive amination of aldehydes and ketones with Hantzsch ester: synthesis of secondary and tertiary amines Tetrahedron 69 4938
Sousa S C A and Fernandes A C 2010 Efficient and Highly Chemoselective Direct Reductive Amination of Aldehydes using the System Silane/Oxorhenium Complexes Adv. Synth. Catal. 352 2218
Apodaca R and Xiao W 2001 Direct Reductive Amination of Aldehydes and Ketones Using Phenylsilane: Catalysis by Dibutyltin Dichloride Org. Lett. 3 1745
Zhua X, Zhoua X and Zhanga W 2015 One-pot reductive amination of araldehydes by aniline using borohydride with CeCl3.7H2O as catalyst J. Chem. Res. 39 390
Lane C F 1975 Sodium Cyanoborohydride - A Highly Selective Reducing Agent for Organic Functional Groups Synthesis 135
Bomann M D, Guch I C and Dimare M 1995 A mild, pyridine-borane-based reductive amination protocol J. Org. Chem. 60 5995
Abdel-Magid A F, Carson K G, Harris B D, Maryanoff C A and Shah R D 1996 Reductive Amination of Aldehydes and Ketones with Sodium Triacetoxyborohydride. Studies on Direct and Indirect Reductive Amination Procedures J. Org. Chem. 61 3849
Matsumura T and Nakada M 2014 Direct reductive amination using triethylsilane and catalytic bismuth(III) chloride Tetrahedron Lett. 55 1829
Salmi C, Letourneux Y and Brunel J M 2006 Efficient Diastereoselective Titanium(IV) Reductive Amination of Ketones Lett. Org. Chem. 3 384
Pieri C and Brunel J M 2015 First Efficient Two-Step/One-Pot Zirconium (IV)isopropoxide–Mediated Reductive Amination of Carbonyl Compounds Lett. Drug Des. Discov. 12 448
Sydnes M O 2016 The Use of Microwave For One-pot Reductive Amination. Curr. Green Chem. 3 101
Alinezhad H and Tollabian Z 2010 One-Pot Reductive Amination of Carbonyl Compounds Using Sodium Borohydride-Cellulose Sulfuric Acid Bull. Korean Chem. Soc. 31 1927
Zhua X, Zhoua X and Zhanga W 2015 One-pot reductive amination of araldehydes by aniline using borohydride with CeCl3.7H2O as catalyst J. Chem. Res. 39 390
Ranu B C, Majee A and Sarkar A 1998 One-Pot Reductive Amination of Conjugated Aldehydes and Ketones with Silica Gel and Zinc Borohydride J. Org. Chem. 63 370
Yang K and Liu J -T 2015 One-pot asymmetric reductive amination of ketones induced by polyfluoroalkanesulfinamide J. Fluorine Chem. 173 18
Varma R S and Dahiya R 1998 Sodium borohydride on wet clay: Solvent-free reductive amination of carbonyl compounds using microwaves Tetrahedron 54 6293
Verardo G, Giumanin A G, Strazzolini P and Poiana M 1993 Reductive N-Monoalkylation of Primary Aromatic Amines Synthesis 121
Bhattacharyya S 1995 Reductive alkylation of dimethylamine using titanium(IV) isopropoxide and sodium borohydride: an efficient, safe, and convenient method for the synthesis of N,N-dimethylated tertiary amines J. Org. Chem. 60 4928
Saxena I, Borah R and Sarma J C 2000 Reductive amination of aromatic aldehydes and ketones with nickel boride J. Chem. Soc. Perkin Trans. 1 503
Cho B T and Kang S K 2005 Direct and indirect reductive amination of aldehydes and ketones with solid acid-activated sodium borohydride under solvent-free conditions Tetrahedron 61 5725
Alinezhad H, Tajbakhsh M and Hamidi N 2010 Reductive amination of aldehydes and ketones using sodium borohydride in the presence of silica chloride under solvent-free conditions Chin. Chem. Lett. 21 7
Alinezhad H, Tajbakhsh M and Salehian F 2005 Reductive Amination of Aldehydes and Ketones to Their Corresponding Amines with NaBH4 in Micellar Media Monatsh. Chem. 136 2029
Alinezhad H and Ardestani E 2007 Amination of Carbonyl Compounds with NaBH4-H3 PW 12 O 40 in THF and Under Solvent-Free Conditions Lett. Org. Chem. 4 473
Alinezhad H, Tajbakhsh M and Enayati Ahangar R 2008 Reductive Amination of Aldehydes and Ketones in a Heterogeneous System in THF and under Solvent-Free Conditions Using Sodium Borohydride-Silica Phosphoric Acid Monatsh. Chem. 13 21
Alinezhad H, Tajbakhsh M and Zare M 2009 Reductive Amination of Aldehydes and Ketones Under Heterogeneous and Solvent-Free Conditions Using Sodium-Borohydride and Silica-Gel–Supported Sulfuric Acid Syn. Commun. 39 2907
Alinezhad H, Tajbakhsh M and Mahdavi N 2010 One-Pot Reductive Amination of Carbonyl Compounds Using Sodium Borohydride–Amberlyst 15 Syn. Commun. 40 951
Setamdideh D and Sepehraddin F 2014 Convenient Reductive Amination of Aldehydes by NaBH4/Cation Exchange Resin J. Mex. Chem. Soc. 58 22
Nagarsenkar A, Prajapti S K and Babu B N 2015 An efficient catalytic reductive amination: A facile one-pot access to 1,2-dihydropyrrolo[3,4-b]indol-3(4 H)-ones by using B(C6 F 5)3/NaBH4 J. Chem. Sci. 127 711
Surya P R H and Senthilkumar S P 2004 Microwave-mediated reductive amination-cyclization of 4-aryl-4-oxobutanoates: Facile synthesis of 3-methylidene-5-phenyl-2,3-dihydropyrrolidones J. Chem. Sci. 116 163
Sharma R K, Dutta S and Sharma S 2016 Nickel(II) complex covalently anchored on core shell structured SiO2@Fe3 O 4 nanoparticles: A robust and magnetically retrievable catalyst for direct one-pot reductive amination of ketone New J. Chem. 40 2089
Javaherian M and Doraghi F 2015 Nano silica boron sulfuric acid as a dual Brønsted/Lewis acid and a heterogeneous catalyst in Baeyer-Villiger oxidation of ketones with hydrogen peroxide React. Kinet. Mech. Cat. 116 235
Khalafi-Nezhad A, Foroughi H, Doroodmand M and Panahi F 2011 Silica boron–sulfuric acid nanoparticles (SBSANs): preparation, characterization and their catalytic application in the Ritter reaction for the synthesis of amide derivatives J. Mater. Chem. 21 12842
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information (SI)
All additional information pertaining to characterization of the products and catalyst using NMR technique (Figures S3–S5) and IR spectra (Figures S1, S6–S23) are given in the supporting information. Supplementary Information is available at http://www.ias.ac.in/chemsci.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
HAMADI, H., JAVADI, S. One-pot Reductive Amination of Carbonyl Compounds with NaBH 4 -B(OSO 3 H) 3 /SiO 2 in Acetonitrile and in Solvent-free Condition. J Chem Sci 129, 75–80 (2017). https://doi.org/10.1007/s12039-016-1208-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-016-1208-8