Abstract
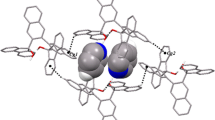
Tetrakis(2,6-dimethyl-4-acetoxyphenyl)pyrene H2 containing flexible acetate functionalities at the para positions of sterically-hindered and rigid aryl rings functions as an inclusion host system. Depending on the orientations of the acetate functionalities, a variety of conformers may indeed be expected. A limited number of the crystal structures of the inclusions compounds of H2 reveal that one indeed observes 2 different conformations for the host based on the orientations of the acetate functionalities. The inclusion compound of H2 with benzene guest molecules is particularly appealing in terms of how the latter are held in trough domains of the host by weak C-H…O and C-H…π hydrogen bonds. More experimentation and analyses of crystal structures of such systems is expected to lead to better insights toward realizing multicomponent molecular crystals in a rational manner.
Similar content being viewed by others
References
Atwood J L, Davies J E D and Macnicol D D 1984 In Inclusion compounds (London: Academic Press Inc)
Weber E 1987 and 1988 In Molecular inclusion and molecular recognition-Clathrates I and II topics in current chemistry (Berlin-Heidelberg: Springer-Verlag) vols 140 and 149
Desiraju G R 1989 Crystal engineering: The design of organic solids (Amsterdam, Netherlands: Elsevier)
Atwood J L, Davies J E D and Macnicol D D 1991 In Inclusion compounds (Oxford: Oxford University Press)
Atwood J L, Davies J E D, MacNicol D D, Vogtle F and Lehn J-M 1996 In Comprehensive supramolecular chemistry (Oxford: Pergamon)
Ramamurthy V 1991 Photochemistry in constrained and organized media (New York: VCH Publishers)
Desiraju G R 1997 Curr. Opin. Solid. State. Mater. Sci. 2 451
Toda F and Bishop R 2004 Separation and reactions in organic supramolecular chemistry (New York: Wiley)
Miyata M 2004 Inclusion reactions and polymerization: Encyclopedia of supramolecular chemistry (New York: Marcel Decker) p. 705
Sozzani P, Bracco S, Comotti A, Ferretti L and Simonutti R 2005 Angew. Chem., Int. Ed. 44 1816
Vishweshwar P, McMahon J A, Bis J A and Zaworotko M J 2006 J. Pharm. Sci. 95 499
Macnicol D D, Toda F and Bishop R 1996 Comprehensive supramolecular chemistry, solid state chemistry: Crystal engineering (Oxford: Pergamon Press)
Malek N, Maris T, Perron M-E and Wuest J D 2005 Angew. Chem., Int. Ed. 44 4021
Maly K E, Gagnon E, Maris T and Wuest J D 2007 J. Am. Chem. Soc. 129 4306
Holman K T, Pivovar A M, Swift J A and Ward M D 2001 Acc. Chem. Res. 34 107
Moorthy J N, Natarajan P and Venugopalan P 2009 J. Org. Chem. 74 8566
Moorthy J N, Natarajan P and Venugopalan P 2010 Chem. Commun. 46 3574
Nangia A 2008 Acc. Chem. Res. 41 595
Moorthy J N, Natarajan P, Venkatakrishnan P, Huang D-F and Chow T J 2007 Org. Lett. 9 5215
Sheldrick, G M 1997 SHELX-97 Program for the refinement and solution of crystal structures (University of Gottingen: Gottingen, Germany)
Desiraju G R and Steiner T 1999 The weak hydrogen bond in structural chemistry and biology (Oxford: Oxford University Press)
Nishio M 2005 Cryst. Eng. Commun. 6 130
Miyata M, Tohnai N and Hisaki I 2007 Molecules 12 1973
Moorthy J N and Natarajan P 2008 J. Mol. Struct. 885 139
Rebek J 1987 Science 235 1478
Rebek J, Askew B, Islam N, Killoran M, Nemeth D and Wolak R 1985 J. Am. Chem. Soc. 107 6736
Pascal R A and Ho D M 1993 J. Am. Chem. Soc. 115 8507
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Natarajan, P., Venugopalan, P. & Moorthy, J.N. A novel tetraarylpyrene host: Conformation-dependent inclusion of guest molecules in the crystal lattice. J Chem Sci 122, 697–706 (2010). https://doi.org/10.1007/s12039-010-0058-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-010-0058-z