Abstract
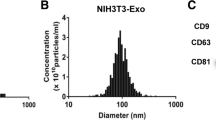
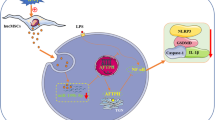
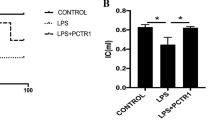
Microvascular hyperpermeability is a leading mechanism responsible for occurrence of edema in remote organs and tissues in patients with burn injury. Accumulated evidence has shown that exosomes can be transported into target cells, where they are capable of regulating biological functions and physiology. Of exosomal proteins contributing to enhanced inflammation and vascular permeability, S100 calcium binding protein A9 (S100A9) has received increasing attention. Here we hypothesized that S100A9-containing serum exosomes of patients with burn injury contribute to pathogenesis of hyperpermeability of microvascular structure in lung by transferring signaling molecules into it and activating downstream signaling pathways, ultimately leading to disruption of the tight junctions (TJs) and endothelial barrier. A use of enzyme-linked immunosorbent assay revealed that total serum concentrations of S100A9 were significantly augmented in burn injury patients in comparison to normal controls. With use of human pulmonary microvascular endothelial cells (HPMECs) as an in vitro model, we found that patients’ serum exosomes were effectively internalized by HPMECs. We further found that serum exosomes of stage II/II burn patients inhibited zonula occludens (ZO-1) and occludin protein levels, which are essential for TJs integrity and endothelial barrier function, but activated p38 MAPK signaling pathway in HPMECs. As expected, such exosomes-mediated effects on HPMECs were reversed by a simultaneous treatment of anti-S100A9 neutralizing antibody. Finally, we found that a recombinant human S100A9 treatment led to inhibition of expression of occludin and ZO-1 but an activation of p38 signaling in HPMECs, and that such effects were reversed when p38 activity was repressed, implying that S100A9 may stimulate p38 activity to inhibit ZO-1 and occludin in HPMECs. Collectively, these data suggest that S100A9-containing serum exosomes may play a critical role in contributing to pulmonary microvascular hyperpermeability, thus supporting that blocking exosomes’ access to HPMECs could hold a promise strategy for treatment of lung edema resulting from burn injuries.





Similar content being viewed by others
References
Bauer H, Zweimueller-Mayer J, Steinbacher P, Lametschwandtner A and Bauer HC 2010 The dual role of zonula occludens (ZO) proteins. J. Biomed. Biotechnol. 2010 402593
Bittner EA, Shank E, Woodson L and Martyn JA 2015 Acute and perioperative care of the burn-injured patient. Anesthesiology 122 448–464
Boldeanu L, Boldeanu MV, Bogdan M, Meca AD, Coman CG et al. 2020 Immunological approaches and therapy in burns (Review). Exp. Ther. Med. 20 2361–2367
Chakraborty RK and Burns B 2020 Systemic inflammatory response syndrome; in StatPearls (Treasure Island (FL): StatPearls Publishing)
Chen LW, Chang WJ, Wang JS and Hsu CM 2006 Thermal injury-induced peroxynitrite production and pulmonary inducible nitric oxide synthase expression depend on JNK/AP-1 signaling. Crit. Care Med. 34 142–150
Colohan SM 2010 Predicting prognosis in thermal burns with associated inhalational injury: a systematic review of prognostic factors in adult burn victims. J. Burn Care Res. 31 529–539
Ehlermann P, Eggers K, Bierhaus A, Most P, Weichenhan D et al. 2006 Increased proinflammatory endothelial response to S100A8/A9 after preactivation through advanced glycation end products. Cardiovasc. Diabetol. 5 6
Goyette J and Geczy CL 2011 Inflammation-associated S100 proteins: new mechanisms that regulate function. Amino Acids 41 821–842
Johnstone RM 2006 Exosomes biological significance: A concise review. Blood Cells Mol. Dis. 36 315–321
Liu J, Liu J, Wang H and Bai M 2019 Protective effect of celastrol for burn-induced acute lung injury in rats. Int. J. Clin. Exp. Pathol. 12 576–583
Miyazaki K, Hashimoto K, Sato M, Watanabe M, Tomikawa N et al. 2017 Establishment of a method for evaluating endothelial cell injury by TNF-alpha in vitro for clarifying the pathophysiology of virus-associated acute encephalopathy. Pediatr. Res. 81 942–947
Nielson CB, Duethman NC, Howard JM, Moncure M and Wood JG 2017 Burns: Pathophysiology of systemic complications and current management. J. Burn Care Res. 38 e469-e481
Qian F, Deng J, Wang G, Ye RD and Christman JW 2016 Pivotal Role of mitogen-activated protein kinase-activated protein kinase 2 in inflammatory pulmonary diseases. Curr. Protein Pept. Sci. 17 332–342
Qin D, Yang W, Pan Z, Zhang Y, Li X et al. 2020 Differential proteomics analysis of serum exosomein burn patients. Saudi J. Biol. Sci. 27 2215–2220
Rose LF and Chan RK 2016 The burn wound microenvironment. Adv. Wound Care 5 106–118
Rowan MP, Cancio LC, Elster EA, Burmeister DM, Rose LF et al. 2015 Burn wound healing and treatment: review and advancements. Crit. Care 19 243
Schaefer TJ and Szymanski KD 2020 Burn evaluation and management; in StatPearls (Treasure Island (FL): StatPearls Publishing)
Wang L, Luo H, Chen X, Jiang Y and Huang Q 2014 Functional characterization of S100A8 and S100A9 in altering monolayer permeability of human umbilical endothelial cells. PLoS One 9 e90472
Wang W, Weng J, Yu L, Huang Q, Jiang Y et al. 2018 Role of TLR4-p38 MAPK-Hsp27 signal pathway in LPS-induced pulmonary epithelial hyperpermeability. BMC Pulm. Med. 18 178
Williams FN, Herndon DN, Hawkins HK, Lee JO, Cox RA et al. 2009 The leading causes of death after burn injury in a single pediatric burn center. Crit. Care 13 R183
Wittekindt OH 2017 Tight junctions in pulmonary epithelia during lung inflammation. Pflugers Arch. 469 135–147
Youn YK, Lalonde C and Demling R 1992 Oxidants and the pathophysiology of burn and smoke inhalation injury. Free Radic. Biol. Med. 12 409–415
Zhang Y, Liu Y, Liu H and Tang WH 2019 Exosomes: biogenesis, biologic function and clinical potential. Cell Biosci. 9 19
Zheng B, Yin WN, Suzuki T, Zhang XH, Zhang Y et al. 2017 Exosome-mediated miR-155 transfer from smooth muscle cells to endothelial cells induces endothelial injury and promotes atherosclerosis. Mol. Ther. 25 1279–1294
Zu YL, Qi J, Gilchrist A, Fernandez GA, Vazquez-Abad D et al. 1998 p38 mitogen-activated protein kinase activation is required for human neutrophil function triggered by TNF-alpha or FMLP stimulation. J. Immunol. 160 1982–1989
Acknowledgements
This study was supported by Shanghai Health and Family Planning Commission, Shanghai Chinese and Western Medicine Clinical Cooperation Pilot Construction Project-Water Burning Injury (Burn) (ZY (2018–2020)-FWTX-1106), Training Program for Discipline Leaders of Shanghai Pudong New Area Health Planning Commission (Burn Surgery) (PWRd2017–09) and Study on the Effect of Burn Analgesic Ointment on Pain and Inflammatory Factors in Early Burn Stage, a General Project of Shanghai Pudong New Area Health Planning (PW2017–18).
Funding
Funding was provided by Shanghai Health and Family Planning Commission, Shanghai Chinese and Western medicine clinical cooperation pilot construction project-water burning injury (burn) (Grant No. ZY(2018-2020)-FWTX-1106), Training Program for Discipline Leaders of Shanghai Pudong New Area Health Planning Commission (Burn Surgery) (Grant No. PWRd2017-09) and a General Project of Shanghai Pudong New Area Health Planning (Grant No. PW2017-18).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Our research was permitted by the Ethics Committee of Seventh People’s Hospital Affiliated to Shanghai University of Traditional Chinese Medicine.
Additional information
Corresponding editor: BJ RAO
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, A., Guo, B., Jia, Q. et al. S100A9-containing serum exosomes of burn injury patients promote permeability of pulmonary microvascular endothelial cells. J Biosci 46, 33 (2021). https://doi.org/10.1007/s12038-021-00151-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12038-021-00151-9