Abstract
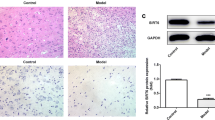
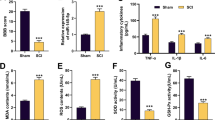
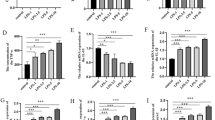
This study investigated the impact of low-dose lipopolysaccharide (LPS) on spinal cord injury (SCI) and the potential molecular mechanism. Rats were randomly assigned to four groups: Sham, SCI, SCI + LPS, and SCI + LPS + agomir. Allen’s weight-drop method was used to establish an in vivo SCI model. The Basso Bcattie Bresnahan rating scale was employed to monitor locomotor function. An in vitro SCI model was constructed by subjecting PC12 cells to oxygen and glucose deprivation/ reoxygenation (OGD/R). Enzyme-linked immunosorbent assay (ELISA) was applied for the determination interleukin (IL)-1β and IL-6. The dual luciferase reporter assay was used to validate the targeting of microRNA (miR)-429 with PI3K. Immunohistochemical staining was used to assess the expression of PI3K, phosphorylated AKT and Nrf2 proteins. The Nrf2-downstream anti-oxidative stress proteins, OH-1 and NQO1, were detected by western blot assay. MiR-429 expression was detected by fluorescence in situ hybridization and real-time quantitative reverse transcription PCR. In vitro, low-dose LPS decreased miR-429 expression, activated PI3K/AKT/Nrf2, inhibited oxidative stress and inflammation, and attenuated SCI. MiR-429 was found to target and negatively regulate PI3K. Inhibition of miR-429 suppressed low-dose LPS-mediated oxidative stress and inflammation via activation of the PI3K/AKT/Nrf2 pathway. In vivo, miR-429 was detectable in neurons. Inhibition of miR-429 blocked low-dose LPS-mediated oxidative stress and inflammation via activation of the PI3K/AKT/Nrf2 pathway. Overall, low-dose LPS was found to alleviate SCI-induced neuronal oxidative stress and inflammatory response by down-regulating miR-429 to activate the PI3K/AKT/Nrf2 pathway.
Graphical Abstract

Highlights
Low-dose LPS reduces the inflammatory response and alleviates SCI.
Low-dose LPS decreases miR-429 levels and activates PI3K/AKT/Nrf2 signaling in SCI.
MiR-429 targets PI3K in SCI.
Inhibition of miR-429 activates PI3K/AKT/Nrf2 signaling in SCI.







Similar content being viewed by others
Data Availability
Data will be made available on reasonable request.
Code Availability
Not applicable.
References
Eckert M, Martin M (2017) Trauma: spinal cord Injury. Surg Clin North Am 97(5):1031–1045
Wang T et al (2015) The role of the JAK-STAT pathway in neural stem cells, neural progenitor cells and reactive astrocytes after spinal cord injury. Biomedical Rep 3(2):141–146
Yang X et al (2018) Apolipoprotein E Deficiency exacerbates spinal cord Injury in mice: inflammatory response and oxidative stress mediated by NF-κB signaling pathway. Front Cell Neurosci 12:142
Khurana S, Garg D (2014) Spasticity and the use of intrathecal baclofen in patients with spinal cord injury. Phys Med Rehabil Clin North Am, 25(3): p. 655 – 69, ix.
Cardenas D et al (2013) A randomized trial of pregabalin in patients with neuropathic pain due to spinal cord injury. Neurology 80(6):533–539
Scholtes F, Brook G, Martin D (2012) Spinal cord injury and its treatment: current management and experimental perspectives. Adv Tech Stand Neurosurg 38:29–56
Hwang Y, Jeong H (2010) Ginsenoside Rb1 protects against 6-hydroxydopamine-induced oxidative stress by increasing heme oxygenase-1 expression through an estrogen receptor-related PI3K/Akt/Nrf2-dependent pathway in human dopaminergic cells. Toxicol Appl Pharmcol 242(1):18–28
Nguyen C, Kim H, Lee S (2013) Caffeoylserotonin protects human keratinocyte HaCaT cells against H2 O2 -induced oxidative stress and apoptosis through upregulation of HO-1 expression via activation of the PI3K/Akt/Nrf2 pathway Phytotherapy research: PTR, 27(12): p. 1810-8
Ma Y et al (2014) Atorvastatin represses the angiotensin 2-induced oxidative stress and inflammatory response in dendritic cells via the PI3K/Akt/Nrf 2 pathway Oxidative medicine and cellular longevity, 2014: p. 148798
Yuan Z et al (2020) Tribulus terrestris Ameliorates Oxidative Stress-Induced ARPE-19 Cell Injury through the PI3K/Akt-Nrf2 Signaling Pathway Oxidative medicine and cellular longevity, 2020. : p. 7962393
Fu C et al (2021) viaNeuroprotective effect of apigenin against hypoxic-ischemic brain injury in neonatal rats activation of the PI3K/Akt/Nrf2 signaling pathway, vol 12. Food & function, pp 2270–2281. 5
Xu S et al (2021) CD73 alleviates GSDMD-mediated microglia pyroptosis in spinal cord injury through PI3K/AKT/Foxo1 signaling. Clin Transl Med 11(1):e269
Xu S et al (2020) TLR4 promotes microglial pyroptosis via lncRNA-F630028O10Rik by activating PI3K/AKT pathway after spinal cord injury. Cell Death Dis 11(8):693
Ge MH et al (2021) Zinc attenuates ferroptosis and promotes functional recovery in contusion spinal cord injury by activating Nrf2/GPX4 defense pathway. CNS Neurosci Ther 27(9):1023–1040
Mohr AM, Mott JL (2015) Overview of microRNA biology. Semin Liver Dis 35(1):3–11
Liu W et al (2020) Exosome-shuttled miR-216a-5p from hypoxic preconditioned mesenchymal stem cells repair traumatic spinal cord injury by shifting microglial M1/M2 polarization. J Neuroinflammation 17(1):47
Chen T et al (2019) MiR-27a promotes insulin resistance and mediates glucose metabolism by targeting PPAR-γ-mediated PI3K/AKT signaling. Aging 11(18):7510–7524
Sun R et al (2020) MiR-429 regulates blood-spinal cord barrier permeability by targeting Krüppel-like factor 6. Biochem Biophys Res Commun 525(3):740–746
Zhu Q, Hu F (2019) Antagonism of miR-429 ameliorates anoxia/reoxygenation injury in cardiomyocytes by enhancing MO25/LKB1/AMPK mediated autophagy. Life Sci 235:116842
Xiao J, Kong R, Hu J (2018) Inhibition of microRNA-429 attenuates oxygen-glucose deprivation/reoxygenation-induced neuronal injury by promoting expression of GATA-binding protein 4. NeuroReport 29(9):723–730
Lv H, Zhang S, Hao X (2020) Swainsonine protects H9c2 cells against lipopolysaccharide-induced apoptosis and inflammatory injury via down-regulating miR-429. Cell Cycle 19(2):207–217
Li H et al (2019) Long non-coding RNA Mirt2 relieves lipopolysaccharide-induced injury in PC12 cells by suppressing miR-429. J Physiol Biochem 75(3):403–413
Wang X et al (2022) Ginsenoside Rg3 alleviates Cisplatin Resistance of Gastric Cancer cells through inhibiting SOX2 and the PI3K/Akt/mTOR Signaling Axis by Up-Regulating miR-429. Front Genet 13:823182
Yu Z et al (2020) Daidzein ameliorates LPS-induced hepatocyte injury by inhibiting inflammation and oxidative stress. Eur J Pharmacol 885:173399
Park BS et al (2009) The structural basis of lipopolysaccharide recognition by the TLR4-MD-2 complex. Nature 458(7242):1191–1195
Cochet F, Peri F (2017) The role of Carbohydrates in the lipopolysaccharide (LPS)/Toll-Like receptor 4 (TLR4) signalling. Int J Mol Sci, 18(11)
Ling Y et al (2020) The Protective Effect of Low Dose of Lipopolysaccharide Pretreatment on Endotoxin-Induced Uveitis in Rats Is Associated with Downregulation of CSF-1 and Upregulation of LRR-1 J Immunol Res, 2020: p. 9314756
Boribong BP et al (2019) Super-low dose Lipopolysaccharide Dysregulates Neutrophil Migratory decision-making. Front Immunol 10:359
Zhou XY et al (2020) Trained Innate Immunity by Repeated Low-Dose Lipopolysaccharide Injections Displays Long-Term Neuroprotective Effects Mediators Inflamm, 2020: p. 8191079
Chang R et al (2014) LPS preconditioning ameliorates intestinal injury in a rat model of hemorrhagic shock. Inflamm Res 63(8):675–682
Zhu Q et al (2015) [Low-dose lipopolysaccharide mediated up-regulation of Nrf2 attenuates inflammatory response in rats with spinal cord injury] Xi bao yu fen zi mian yi xue za zhi = Chinese journal of cellular and molecular immunology, 31(4): p. 437 – 42
Hayakawa K et al (2014) Lipopolysaccharide preconditioning facilitates M2 activation of resident microglia after spinal cord injury. J Neurosci Res 92(12):1647–1658
Popovich P et al (2012) A reassessment of a classic neuroprotective combination therapy for spinal cord injured rats: LPS/pregnenolone/indomethacin. Exp Neurol 233(2):677–685
Freria C et al (2020) Serial systemic injections of Endotoxin (LPS) elicit neuroprotective spinal cord Microglia through IL-1-Dependent Cross talk with endothelial cells. J neuroscience: official J Soc Neurosci 40(47):9103–9120
Nourbakhsh S, Yu L, Ha B (2021) Modeling the protective role of bacterial lipopolysaccharides against membrane-rupturing peptides. J Phys Chem,
Dong Z, Yuan Y (2018) Accelerated inflammation and oxidative stress induced by LPS in acute lung injury: Ιnhibition by ST1926. Int J Mol Med 41(6):3405–3421
Bastin A et al (2021) The effects of malvidin on oxidative stress parameters and inflammatory cytokines in LPS-induced human THP-1 cells. J Cell Physiol 236(4):2790–2799
Young B, Ildefonso C (2019) Systematic injection of low-dose LPS transiently improves the retina function and structure of a mouse model of Geographic Atrophy. Adv Exp Med Biol 1185:57–62
Wang D et al (2016) Low dose of lipopolysaccharide pretreatment can alleviate the inflammatory response in wound infection mouse model. Chin J traumatology = Zhonghua chuang shang za zhi 19(4):193–198
Lu Y et al (2018) Ketogenic diet attenuates oxidative stress and inflammation after spinal cord injury by activating Nrf2 and suppressing the NF-κB signaling pathways. Neurosci Lett 683:13–18
Li W et al (2021) Low-dose lipopolysaccharide protects nerve cells against spinal cord injury via regulating the PI3K-AKT-Nrf2 signaling pathway Biochemistry and cell biology = Biochimie et biologie cellulaire, : p. 1–9
Kim M et al (2021) Fucoxanthin inhibits lipopolysaccharide-induced inflammation and oxidative stress by activating nuclear factor E2-related factor 2 via the phosphatidylinositol 3-kinase/AKT pathway in macrophages European journal of nutrition,
Wu L et al (2020) Targeting oxidative stress and inflammation to prevent Ischemia-Reperfusion Injury. Front Mol Neurosci 13:28
Shang Y et al (2017) Airborne nitro-PAHs induce Nrf2/ARE defense system against oxidative stress and promote inflammatory process by activating PI3K/Akt pathway in A549 cells. Toxicol vitro: Int J published association BIBRA 44:66–73
Wang M, Xia L, Fu G (2019) Lipopolysaccharide pretreatment inhibits oxidative stress-induced endothelial progenitor cell apoptosis via a TLR4-mediated PI3K/Akt/ NF-κB p65 signaling pathway Cellular and molecular biology. France) 65(4):101–106(Noisy-le-Grand
Ye Z et al (2015) miR-429 inhibits migration and invasion of breast cancer cells in vitro. Int J Oncol 46(2):531–538
Niu H et al (2021) MiR-429 prohibited NF-κB signalling to alleviate contrast-induced acute kidney injury via targeting PDCD4. Autoimmunity 54(5):243–253
Zhang L et al (2020) MiR-429 suppresses proliferation and invasion of breast cancer via inhibiting the Wnt/β-catenin signaling pathway. Thorac cancer 11(11):3126–3138
Yang Q et al (2019) Down-regulation of microRNA-429 alleviates myocardial injury of rats with coronary heart disease. Cell cycle (Georgetown Tex) 18(19):2550–2565
Qi R, Wang X (2020) Inhibition of miR-429 improves neurological recovery of traumatic brain injury mice and attenuates microglial neuroinflammation. Int Immunopharmacol 79:106091
Liu X, Zhao W, Wang X (2020) Inhibition of long non-coding RNA MALAT1 elevates microRNA-429 to suppress the progression of hypopharyngeal squamous cell carcinoma by reducing ZEB1. Life Sci 262:118480
Tang J et al (2015) MiR-429 increases the metastatic capability of HCC via regulating classic wnt pathway rather than epithelial-mesenchymal transition. Cancer Lett 364(1):33–43
Li WC et al (2014) Lipopolysaccharide preconditioning attenuates apoptotic processes and improves neuropathologic changes after spinal cord injury in rats. Int J Neurosci 124(8):585–592
Li G et al (2018) MiR-103 alleviates autophagy and apoptosis by regulating SOX2 in LPS-injured PC12 cells and SCI rats. Iran J Basic Med Sci 21(3):292–300
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21
Bao G et al (2016) Tetrandrine protects against oxygen-glucose-serum deprivation/reoxygenation-induced injury via PI3K/AKT/NF-κB signaling pathway in rat spinal cord astrocytes. Biomed Pharmacother 84:925–930
Acknowledgements
No.
Funding
This study was supported by the National Natural Science Foundation of China (Grant No.: 82260257), Key Research Project of Yunnan Provincial Science and Technology (Grant No.: 202102AA310042; 202001AS070028), Key project of Yunnan clinical medicine research center (Grant No.: 2022YJZX-GK02; 2022YJZX-GK23).
Author information
Authors and Affiliations
Contributions
Weichao Li, Tao Tang, and Tiannan Zou performed the experiment; Shaoping Yao, Qianbo Fan, and Shixiao Zhong contributed significantly to analysis and manuscript preparation; Weichao Li, Tao Tang, and Tiannan Zou performed the data analyses and wrote the manuscript; Weichao Li, Tao Tang, and Tiannan Zou helped perform the analysis with constructive discussions. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics Approval
All animals were kept in a pathogen-free environment and fed ad lib. The procedures for care and use of animals were approved by the Ethics Committee of the First People’s Hospital of Yunnan province and all applicable institutional and governmental regulations concerning the ethical use of animals were followed.
Consent to Participate
Not applicable.
Consent for Publication
All authors have given final approval of the version and agreed with the publication of this study here.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, W., Tang, T., Yao, S. et al. Low-dose Lipopolysaccharide Alleviates Spinal Cord Injury-induced Neuronal Inflammation by Inhibiting microRNA-429-mediated Suppression of PI3K/AKT/Nrf2 Signaling. Mol Neurobiol 61, 294–307 (2024). https://doi.org/10.1007/s12035-023-03483-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03483-9