Abstract
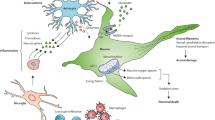
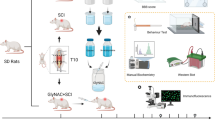
Spinal cord injury (SCI) usually introduces permanent or long-lasting neurological impairments. Maintaining the integrity of the limited number of white matter bundles (5–10%) preserves wholly or partially locomotor following SCI. Considering that the basic structure of white matter bundles is axon wrapped by oligodendrocytes, promoting oligodendrocytes survival might be a feasible strategy for reducing white matter injury (WMI) after SCI. Oligodendrocytes are rich in unsaturated fatty acid and susceptible to ferroptosis-induced damage. Hence, exploring method to reduce ferroptosis is supposed to expedite oligodendrocytes survival, thereafter mitigating WMI to facilitate functional recovery post-SCI. Here, the results indicated the administration of hepcidin reduced iron accumulation to promote oligodendrocytes survival and to decrease spinal cord atrophy, therefore facilitating functional recovery. Then, the WMI was evidently decreased owing to attenuating ferroptosis. Subsequently, the results revealed that the expression of divalent metal transporter 1 (DMT1) and transferrin receptor (TfR) was expressed in CC1+ cells. The expression level of DMT1 and TfR was significantly increased, while this phenomenon was obviously neutralized with the administration of hepcidin in the epicenter of spinal cord after SCI. Afterward, the application of hepcidin downregulated reactive oxygen species (ROS) overload, which was evidently increased with the treatment of 20 μM FeCl3, therefore increasing cell viability and reducing lactate dehydrogenase (LDH) activity through downregulating the expression of DMT1 and TfR to inhibit ferroptosis in oligodendrocyte progenitor cells (OPCs). The present study provides evidence that the application of hepcidin facilitates oligodendrocytes survival to alleviate WMI via reducing the expression of DMT1 and TfR.







Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- SCI:
-
Spinal cord injury
- WMI:
-
White matter injury
- DMT1:
-
Divalent metal transporter 1
- TfR:
-
Transferrin receptor
- ROS:
-
Reactive oxygen species
- LDH:
-
Lactate dehydrogenase
- OPCs:
-
Oligodendrocyte progenitor cells
- GPER1:
-
G-protein coupled estrogen receptor 1
- NSC:
-
Neural stem cell
- MSC:
-
Mesenchymal stem cell
- Fpn1:
-
Ferroportin 1
- PBS:
-
Phosphate-buffered saline
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- bFGF:
-
Basic fibroblast growth factor
- PDGF:
-
Platelet-derived growth factor
- HE:
-
Hematoxylin and eosin
- IHC:
-
Immunohistochemistry
- DAB:
-
3-Diaminobenzidine
- TEM:
-
Transmission electron microscopy
- BCA:
-
Bicinchoninic acid
- PVDF:
-
Polyvinylidene difluoride
- CCK8:
-
Cell counting kit-8
- dMBP:
-
Degraded myelin basic protein
- APP:
-
Amyloid precursor protein
- AD:
-
Alzheimer’s disease
- HRP:
-
Horseradish peroxidase
- RT-qPCR:
-
Reverse transcription-quantitative polymerase chain reaction
- FSP1:
-
Ferroptosis suppressor protein 1 (FSP1)
References
Ge H, Xue X, Xian J, Yuan L, Wang L, Zou Y, Zhong J, Jiang Z, et al (2022) Ferrostatin-1 alleviates white matter injury via decreasing ferroptosis following spinal cord injury. Mol Neurobiol 59(1):161–176. https://doi.org/10.1007/s12035-021-02571-y
Krupa P, Svobodova B, Dubisova J, Kubinova S, Jendelova P, Machova Urdzikova L (2019) Nano-formulated curcumin (Lipodisq™) modulates the local inflammatory response, reduces glial scar and preserves the white matter after spinal cord injury in rats. Neuropharmacology 155:54–64. https://doi.org/10.1016/j.neuropharm.2019.05.018
Yuan J, Liu W, Zhu H, Chen Y, Zhang X, Li L, Chu W, Wen Z, et al (2017) Curcumin inhibits glial scar formation by suppressing astrocyte-induced inflammation and fibrosis in vitro and in vivo. Brain Res 1655:90–103. https://doi.org/10.1016/j.brainres.2016.11.002
Xia Y, Zhao T, Li J, Li L, Hu R, Hu S, Feng H, Lin J (2008) Antisense vimentin cDNA combined with chondroitinase ABC reduces glial scar and cystic cavity formation following spinal cord injury in rats. Biochem Biophys Res Commun 377(2):562–566. https://doi.org/10.1016/j.bbrc.2008.10.024
Xia Y, Yan Y, Xia H, Zhao T, Chu W, Hu S, Feng H, Lin J (2015) Antisense vimentin cDNA combined with chondroitinase ABC promotes axon regeneration and functional recovery following spinal cord injury in rats. Neurosci Lett 590:74–79. https://doi.org/10.1016/j.neulet.2015.01.073
Hu R, Duan B, Wang D, Yu Y, Li W, Luo H, Lu P, Lin J, et al (2011) Role of acid-sensing ion channel 1a in the secondary damage of traumatic spinal cord injury. Ann Surg 254(2):353–362. https://doi.org/10.1097/SLA.0b013e31822645b4
Hu R, Sun H, Zhang Q, Chen J, Wu N, Meng H, Cui G, Hu S, et al (2012) G-protein coupled estrogen receptor 1 mediated estrogenic neuroprotection against spinal cord injury. Crit Care Med 40(12):3230–3237. https://doi.org/10.1097/CCM.0b013e3182657560
Chen J, Hu R, Ge H, Duanmu W, Li Y, Xue X, Hu S, Feng H (2015) G-protein-coupled receptor 30-mediated antiapoptotic effect of estrogen on spinal motor neurons following injury and its underlying mechanisms. Mol Med Rep 12(2):1733–1740. https://doi.org/10.3892/mmr.2015.3601
Li L, Xiong ZY, Qian ZM, Zhao TZ, Feng H, Hu S, Hu R, Ke Y, et al (2014) Complement C5a is detrimental to histological and functional locomotor recovery after spinal cord injury in mice. Neurobiol Dis 66:74–82. https://doi.org/10.1016/j.nbd.2014.02.008
Chu W, Yuan J, Huang L, Xiang X, Zhu H, Chen F, Chen Y, Lin J, et al (2015) Valproic acid arrests proliferation but promotes neuronal differentiation of adult spinal NSPCs from SCI rats. Neurochem Res 40(7):1472–1486. https://doi.org/10.1007/s11064-015-1618-x
Chen F, Wang H, Xiang X, Yuan J, Chu W, Xue X, Zhu H, Ge H, et al (2014) Curcumin increased the differentiation rate of neurons in neural stem cells via wnt signaling in vitro study. J Surg Res 192(2):298–304. https://doi.org/10.1016/j.jss.2014.06.026
Hu SL, Luo HS, Li JT, Xia YZ, Li L, Zhang LJ, Meng H, Cui GY, et al (2010) Functional recovery in acute traumatic spinal cord injury after transplantation of human umbilical cord mesenchymal stem cells. Crit Care Med 38(11):2181–2189. https://doi.org/10.1097/CCM.0b013e3181f17c0e
Liu S, Zhang W, Yang L, Zhou F, Liu P, Wang Y (2021) Overexpression of bone morphogenetic protein 7 reduces oligodendrocytes loss and promotes functional recovery after spinal cord injury. J Cell Mol Med 25(18):8764–8774. https://doi.org/10.1111/jcmm.16832
Duncan GJ, Manesh SB, Hilton BJ, Assinck P, Plemel JR, Tetzlaff W (2020) The fate and function of oligodendrocyte progenitor cells after traumatic spinal cord injury. Glia 68(2):227–245. https://doi.org/10.1002/glia.23706
Pukos N, Goodus MT, Sahinkaya FR, McTigue DM (2019) Myelin status and oligodendrocyte lineage cells over time after spinal cord injury: what do we know and what still needs to be unwrapped? Glia 67(11):2178–2202. https://doi.org/10.1002/glia.23702
Jhelum P, Santos-Nogueira E, Teo W, Haumont A, Lenoël I, Stys PK, David S (2020) Ferroptosis mediates cuprizone-induced loss of oligodendrocytes and demyelination. J Neurosci 40(48):9327–9341. https://doi.org/10.1523/jneurosci.1749-20.2020
Chen Y, Liu S, Li J, Li Z, Quan J, Liu X, Tang Y, Liu B (2020) The latest view on the mechanism of ferroptosis and its research progress in spinal cord injury. Oxidative Med Cell Longev 2020:6375938. https://doi.org/10.1155/2020/6375938
Li Q, Weiland A, Chen X, Lan X, Han X, Durham F, Liu X, Wan J, et al (2018) Ultrastructural characteristics of neuronal death and white matter injury in mouse brain tissues after intracerebral hemorrhage: coexistence of ferroptosis, autophagy, and necrosis. Front Neurol 9:581. https://doi.org/10.3389/fneur.2018.00581
Yao X, Zhang Y, Hao J, Duan HQ, Zhao CX, Sun C, Li B, Fan BY, et al (2019) Deferoxamine promotes recovery of traumatic spinal cord injury by inhibiting ferroptosis. Neural Regen Res 14(3):532–541. https://doi.org/10.4103/1673-5374.245480
Feng Z, Min L, Chen H, Deng W, Tan M, Liu H, Hou J (2021) Iron overload in the motor cortex induces neuronal ferroptosis following spinal cord injury. Redox Biol 43:101984. https://doi.org/10.1016/j.redox.2021.101984
Qian ZM, Ke Y (2020) Hepcidin and its therapeutic potential in neurodegenerative disorders. Med Res Rev 40(2):633–653. https://doi.org/10.1002/med.21631
Ganz T (2013) Systemic iron homeostasis. Physiol Rev 93(4):1721–1741. https://doi.org/10.1152/physrev.00008.2013
Ge XH, Wang Q, Qian ZM, Zhu L, Du F, Yung WH, Yang L, Ke Y (2009) The iron regulatory hormone hepcidin reduces ferroportin 1 content and iron release in H9C2 cardiomyocytes. J Nutr Biochem 20(11):860–865. https://doi.org/10.1016/j.jnutbio.2008.07.014
Du F, Qian ZM, Luo Q, Yung WH, Ke Y (2015) Hepcidin suppresses brain iron accumulation by downregulating iron transport proteins in iron-overloaded Rats. Mol Neurobiol 52(1):101–114. https://doi.org/10.1007/s12035-014-8847-x
Du F, Qian C, Qian ZM, Wu XM, Xie H, Yung WH, Ke Y (2011) Hepcidin directly inhibits transferrin receptor 1 expression in astrocytes via a cyclic AMP-protein kinase A pathway. Glia 59(6):936–945. https://doi.org/10.1002/glia.21166
Shi J, Tang R, Zhou Y, Xian J, Zuo C, Wang L, Wang J, Feng H, et al (2020) Attenuation of white matter damage following deferoxamine treatment in rats after spinal cord injury. World Neurosurg 137:e9–e17. https://doi.org/10.1016/j.wneu.2019.08.246
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21. https://doi.org/10.1089/neu.1995.12.1
Hu SL, Lu PG, Zhang LJ, Li F, Chen Z, Wu N, Meng H, Lin JK, et al (2012) In vivo magnetic resonance imaging tracking of SPIO-labeled human umbilical cord mesenchymal stem cells. J Cell Biochem 113(3):1005–1012. https://doi.org/10.1002/jcb.23432
Jiang X, Zhang J, Kou B, Zhang C, Zhong J, Fang X, Huang X, Zhang X, et al (2020) Ambroxol improves neuronal survival and reduces white matter damage through suppressing endoplasmic reticulum stress in microglia after intracerebral hemorrhage. Biomed Res Int 2020:8131286. https://doi.org/10.1155/2020/8131286
Cheli VT, Correale J, Paez PM, Pasquini JM (2020) Iron metabolism in oligodendrocytes and astrocytes, implications for myelination and remyelination. ASN neuro 12:1759091420962681. https://doi.org/10.1177/1759091420962681
Cheli VT, Santiago González DA, Marziali LN, Zamora NN, Guitart ME, Spreuer V, Pasquini JM, et al (2018) The divalent metal transporter 1 (DMT1) is required for iron uptake and normal development of oligodendrocyte progenitor cells. J Neurosci 38(43):9142–9159. https://doi.org/10.1523/jneurosci.1447-18.2018
Zhang L, Wang Z, Song C, Liu H, Li Y, Li J, Yu Y, Wang G, et al (2021) Spinal NR2B phosphorylation at Tyr1472 regulates IRE(-)DMT1-mediated iron accumulation and spine morphogenesis via kalirin-7 in tibial fracture-associated postoperative pain after orthopedic surgery in female mice. Reg Anesth Pain Med 46(4):363–373. https://doi.org/10.1136/rapm-2020-101883
Park E, Chung SW (2019) ROS-mediated autophagy increases intracellular iron levels and ferroptosis by ferritin and transferrin receptor regulation. Cell Death Dis 10(11):822. https://doi.org/10.1038/s41419-019-2064-5
Krause A, Neitz S, Mägert HJ, Schulz A, Forssmann WG, Schulz-Knappe P, Adermann K (2000) LEAP-1, a novel highly disulfide-bonded human peptide, exhibits antimicrobial activity. FEBS Lett 480(2-3):147–150. https://doi.org/10.1016/s0014-5793(00)01920-7
Park CH, Valore EV, Waring AJ, Ganz T (2001) Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J Biol Chem 276(11):7806–7810. https://doi.org/10.1074/jbc.M008922200
Raha AA, Vaishnav RA, Friedland RP, Bomford A, Raha-Chowdhury R (2013) The systemic iron-regulatory proteins hepcidin and ferroportin are reduced in the brain in Alzheimer's disease. Acta Neuropathologica Commun 1:55. https://doi.org/10.1186/2051-5960-1-55
Derry PJ, Hegde ML, Jackson GR, Kayed R, Tour JM, Tsai AL, Kent TA (2020) Revisiting the intersection of amyloid, pathologically modified tau and iron in Alzheimer's disease from a ferroptosis perspective. Prog Neurobiol 184:101716. https://doi.org/10.1016/j.pneurobio.2019.101716
Blomster LV, Cowin GJ, Kurniawan ND, Ruitenberg MJ (2013) Detection of endogenous iron deposits in the injured mouse spinal cord through high-resolution ex vivo and in vivo MRI. NMR Biomed 26(2):141–150. https://doi.org/10.1002/nbm.2829
Urrutia PJ, Hirsch EC, González-Billault C, Núñez MT (2017) Hepcidin attenuates amyloid beta-induced inflammatory and pro-oxidant responses in astrocytes and microglia. J Neurochem 142(1):140–152. https://doi.org/10.1111/jnc.14005
Li JZ, Fan BY, Sun T, Wang XX, Li JJ, Zhang JP, Gu GJ, Shen WY, et al (2023) Bioinformatics analysis of ferroptosis in spinal cord injury. Neural Regen Res 18(3):626–633. https://doi.org/10.4103/1673-5374.350209
Wang F, Li J, Zhao Y, Guo D, Liu D, Chang S, Qiao H, Li J, et al (2022) miR-672-3p promotes functional recovery in rats with contusive spinal cord injury by inhibiting ferroptosis suppressor protein 1. Oxidative Med Cell Longev 2022:6041612. https://doi.org/10.1155/2022/6041612
Shao C, Chen Y, Yang T, Zhao H, Li D (2022) Mesenchymal stem cell derived exosomes suppress neuronal cell ferroptosis via lncGm36569/miR-5627-5p/FSP1 axis in acute spinal cord injury. Stem Cell Rev Rep 18(3):1127–1142. https://doi.org/10.1007/s12015-022-10327-x
Funding
This work was supported by grants from the National Natural Science Foundation of China (approval no. 81471261) and Natural Science Foundation of Chongqing (approval no. cstc2018jcyjAX0080).
Author information
Authors and Affiliations
Contributions
Jiantao Shi, Xingsen Xue, and Linbo Yuan performed most of the experiments, with assistance from Guanjian He, Zhouyang Jiang, Long Wang, Jiali Zhang, and Hong Su. Zhouyang Jiang and Long Wang analyzed the results and edited figures. Jiantao Shi, Xingsen Xue, and Linbo Yuan performed SCI model and statistical analysis. Guanjian He and Jiali Zhang performed cell culture and treatments. Jiantao Shi, Xingsen Xue, and Jishu Xian performed immunoblotting and immunostaining. Jiantao Shi wrote preliminary draft of the manuscript. Chuhua Fu designed experiments and revised the manuscript. Shengli Hu made the hypothesis and revised the manuscript. All authors approved final version of the manuscript.
Corresponding authors
Ethics declarations
Research Involving Animals
The animal experiment procedures were implemented according to the China’s animal welfare legislation for the protection of animals used for scientific purposes. And, this work was supervised by the Ethics Committee of the Southwest Hospital, Third Military Medical University (Army Medical University) for the use of laboratory animals (approval no. AMUWEC20210017).
Informed Consent
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shi, J., Xue, X., Yuan, L. et al. Amelioration of White Matter Injury Through Mitigating Ferroptosis Following Hepcidin Treatment After Spinal Cord Injury. Mol Neurobiol 60, 3365–3378 (2023). https://doi.org/10.1007/s12035-023-03287-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-023-03287-x