Abstract
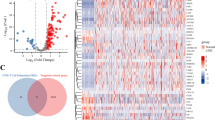
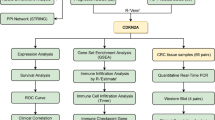
CD27 as a marker of memory B cells is belong to the tumor necrosis factor receptor (TNFR) superfamily, CD27 is ligated by CD70, they can co-stimulate T-cell growth and differentiation through their interaction. Uncertainty surrounds CD27's function in esophageal cancer (EC). This study investigated the role of CD27 in the prognosis of EC using the TCGA, cbioportal, linkedomics and GEPIA databases as well as the proliferation assay was applied. CD27 differential expression may be a key factor in the development of EC. different level of CD27 expression in EC has profound impacts on TOR complex, and many kinds of kinase (KIT proto-oncogene receptor tyrosine kinase, transforming growth factor beta receptor 1, and G protein-coupled receptor kinase 3.), as well as the cell membrane, and survival analysis revealed that it had a significant impact on both the overall survival and disease-free survival of EC. CD27 overexpression will suppress the viability of the KYSE150 and TE3 cells. Our findings suggested that the degree of CD27 expression could serve as an esophageal cancer prognosis biomarker.







Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Uhlenhopp, D. J., Then, E. O., Sunkara, T., & Gaduputi, V. (2020). Epidemiology of esophageal cancer: Update in global trends, etiology and risk factors. Clinical Journal of Gastroenterology, 13(6), 1010–1021.
Watanabe, M., Otake, R., Kozuki, R., et al. (2020). Recent progress in multidisciplinary treatment for patients with esophageal cancer. Surgery Today, 50(1), 12–20.
Short, M. W., Burgers, K. G., & Fry, V. T. (2017). Esophageal Cancer. American family physician., 95(1), 22–28.
Sanmamed, M. F., Pastor, F., Rodriguez, A., et al. (2015). Agonists of Co-stimulation in Cancer Immunotherapy Directed Against CD137, OX40, GITR, CD27, CD28, and ICOS. Seminars in Oncology, 42(4), 640–655.
Wajant, H. (2016). Therapeutic targeting of CD70 and CD27. Expert Opinion on Therapeutic Targets, 20(8), 959–973.
Stetler-Stevenson, M., Paiva, B., Stoolman, L., et al. (2016). Consensus guidelines for myeloma minimal residual disease sample staining and data acquisition. Cytometry. Part B, Clinical Cytometry, 90(1), 26–30.
Riether, C., Pabst, T., Hopner, S., et al. (2020). Targeting CD70 with cusatuzumab eliminates acute myeloid leukemia stem cells in patients treated with hypomethylating agents. Nature Medicine, 26(9), 1459–1467.
Tanaka Y, Takeda R, Fukushima T, et al. Eliminating chronic myeloid leukemia stem cells by IRAK1/4 inhibitors. Nat Commun. Jan 12 2022;13(1):271.
Reddy, V., Klein, C., Isenberg, D. A., et al. (2017). Obinutuzumab induces superior B-cell cytotoxicity to rituximab in rheumatoid arthritis and systemic lupus erythematosus patient samples. Rheumatology (Oxford), 56(7), 1227–1237.
Yang, Z. Z., Novak, A. J., Ziesmer, S. C., Witzig, T. E., & Ansell, S. M. (2007). CD70+ non-Hodgkin lymphoma B cells induce Foxp3 expression and regulatory function in intratumoral CD4+CD25 T cells. Blood, 110(7), 2537–2544.
Schizas, D., Charalampakis, N., Kole, C., et al. (2020). Immunotherapy for esophageal cancer: A 2019 update. Immunotherapy, 12(3), 203–218.
Buchan, S. L., Rogel, A., & Al-Shamkhani, A. (2018). The immunobiology of CD27 and OX40 and their potential as targets for cancer immunotherapy. Blood, 131(1), 39–48.
Starzer, A. M., & Berghoff, A. S. (2020). New emerging targets in cancer immunotherapy: CD27 (TNFRSF7). ESMO Open., 4(Suppl 3), e000629.
Beauchamp, E. M., & Platanias, L. C. (2013). The evolution of the TOR pathway and its role in cancer. Oncogene, 32(34), 3923–3932.
Paquette M, El-Houjeiri L, Pause A. mTOR Pathways in Cancer and Autophagy. Cancers (Basel). 2018;10(1).
Kamińska, K., Szczylik, C., Bielecka, Z. F., et al. (2015). The role of the cell-cell interactions in cancer progression. Journal of cellular and molecular medicine., 19(2), 283–296.
Yousefi SR, Alshamsi HA, Amiri O, Salavati-Niasari M. Synthesis, characterization and application of Co/Co3O4 nanocomposites as an effective photocatalyst for discoloration of organic dye contaminants in wastewater and antibacterial properties. J Mol Liq 2021;337.
Kose, D., Guzelcicek, A., Oz, O., et al. (2022). The mutation of CD27 deficiency presented with familial hodgkin lymphoma and a review of the literature. Journal of Pediatric Hematology/Oncology, 44(4), e833–e843.
Remedios, K. A., Meyer, L., Zirak, B., et al. (2019). CD27 Promotes CD4(+) Effector T Cell Survival in Response to Tissue Self-Antigen. The Journal of Immunology, 203(3), 639–646.
Gantner, P., Pagliuzza, A., Pardons, M., et al. (2020). Single-cell TCR sequencing reveals phenotypically diverse clonally expanded cells harboring inducible HIV proviruses during ART. Nat Commun, 11(1), 4089.
Flieswasser, T., Van den Eynde, A., Van Audenaerde, J., et al. (2022). The CD70-CD27 axis in oncology: The new kids on the block. J Exp Clin Cancer Res., 41(1), 12.
He, L. Z., Prostak, N., Thomas, L. J., et al. (2013). Agonist anti-human CD27 monoclonal antibody induces T cell activation and tumor immunity in human CD27-transgenic mice. The Journal of Immunology, 191(8), 4174–4183.
Buchan, S. L., Fallatah, M., Thirdborough, S. M., et al. (2018). PD-1 Blockade and CD27 Stimulation Activate Distinct Transcriptional Programs That Synergize for CD8(+) T-Cell-Driven Antitumor Immunity. Clinical Cancer Research, 24(10), 2383–2394.
McGray, A. J. R., Eppolito, C., Miliotto, A., et al. (2021). A prime/boost vaccine platform efficiently identifies CD27 agonism and depletion of myeloid-derived suppressor cells as therapies that rationally combine with checkpoint blockade in ovarian cancer. Cancer Immunology, Immunotherapy, 70(12), 3451–3460.
Katayama, Y., Sakai, A., Oue, N., et al. (2003). A possible role for the loss of CD27-CD70 interaction in myelomagenesis. British journal of haematology., 120(2), 223–234.
Kong F, Ye Q, Xiong Y. Comprehensive analysis of prognosis and immune function of CD70-CD27 signaling axis in pan-cancer. Functional & integrative genomics. Jan 26 2023;23(1):48.
Nilsson, A., de Milito, A., Mowafi, F., et al. (2005). Expression of CD27-CD70 on early B cell progenitors in the bone marrow: Implication for diagnosis and therapy of childhood ALL. Experimental Hematology, 33(12), 1500–1507.
Riether, C., Schurch, C. M., Buhrer, E. D., et al. (2017). CD70/CD27 signaling promotes blast stemness and is a viable therapeutic target in acute myeloid leukemia. Journal of Experimental Medicine, 214(2), 359–380.
Schurch, C., Riether, C., Matter, M. S., Tzankov, A., & Ochsenbein, A. F. (2012). CD27 signaling on chronic myelogenous leukemia stem cells activates Wnt target genes and promotes disease progression. The Journal of Clinical Investigation, 122(2), 624–638.
Funding
This project was funded by Youth Foundation of He’nan Scientific Committee (202300410416) and Henan province medical science, technology breakthrough plan project (LHGJ20190033).
Author information
Authors and Affiliations
Contributions
FX conceived and designed the study. LW and FX wrote the manuscript. LW and FX performed the data analysis, FX revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Research involving human and animal participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiao, Fk., Wang, L. Comprehensive Analysis of Expression and Pathway for CD27 in Esophageal Cancer. Mol Biotechnol (2023). https://doi.org/10.1007/s12033-023-00850-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-023-00850-8