Abstract
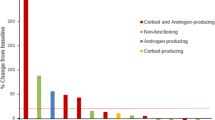
Adrenal insufficiency (AI) is an immune-related adverse event of immune checkpoint inhibitors and on occasion could be serious. There have been few reports of clinical information regarding AI associated with anti-programmed cell death protein-1 (anti-PD-1) antibody in non-small-cell lung cancer (NSCLC) patients. Patients with advanced NSCLC treated with anti-PD-1 antibodies between December 2015 and October 2017 were identified from the medical records of our institution. We investigated the frequency, symptoms, test results, and treatment outcomes of AI, and prognosis of patients who developed AI. In total, 282 NSCLC patients were treated with anti-PD-1 antibodies, and 4 patients (1.4%) were diagnosed as AI and 6 patients (2.1%) as suspicious of AI according to the laboratory data or clinical findings. The median follow-up period was 13.6 months. All patients with AI and suspicious of AI were treated with corticosteroid replacement, and performance status (PS) was improved in 50% of patients. Two of 10 patients were thought to have central AI. Six patients of 10 patients continued to receive anti-PD-1 antibodies with corticosteroid hormone replacement after diagnosis. The median progression-free survival and overall survival were 10.2 and 15.4 months, respectively. In conclusion, the incidence of AI among NSCLC patients treated with anti-PD-1 antibodies was similar to previous studies. Corticosteroid replacement improved PS, symptoms, and laboratory data of patients with AI and suspicious of AI. Corticosteroid replacement may contribute to continuation of anti-PD-1 antibodies and survival outcome was preferable in patients with AI and suspicious of AI receiving corticosteroid treatment.




Similar content being viewed by others
References
Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, et al. Gefitinib or chemotherapy for non–small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362(25):2380–8. https://doi.org/10.1056/NEJMoa0909530.
Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. 2015;373(17):1627–39.
Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WEE, Poddubskaya E, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. 2015;373(2):123–35.
Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375(19):1823–33.
Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016;387(10027):1540–50.
Brahmer JR, Lacchetti C, Schneider BJ, Atkins MB, Brassil KJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36(17):1714–68.
Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(4):iv119–iv142.
Byun DJ, Wolchok JD, Rosenberg LM, Girotra M. Cancer immunotherapy-immune checkpoint blockade and associated endocrinopathies. Nat Rev Endocrinol. 2017;13(4):195–207. https://doi.org/10.1038/nrendo.2016.205.
Wang PF, Chen Y, Song SY, Wang TJ, Ji WJ, Li SW, et al. Immune-related adverse events associated with anti-PD-1/PD-L1 treatment for malignancies: a meta-analysis. Front Pharmacol. 2017;8:730.
Higashiyama RI, Horinouchi H, Sekine K, Matsumoto Y, Murakami S, Goto Y, et al. Efficacy of nivolumab in patients treated by corticosteroid due to immune-related adverse events. J Clin Oncol. 2018. https://doi.org/10.1200/JCO.2018.36.5_suppl.163.
Faje A. Immunotherapy and hypophysitis: clinical presentation, treatment, and biologic insights. Pituitary. 2016;19(1):82–92.
Yanase T, Tajima T, Katabami T, Iwasaki Y, Tanahashi A, Sugawara A, et al. Diagnosis and treatment of adrenal insufficiency including adrenal crisis: a Japan Endocrine Society Clinical Practice Guideline. Endocr J. 2016;63(9):765–84.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48(3):452–8.
Sanlorenzo M, Vujic I, Daud A, Algazi A, Gubens M, Luna SA, et al. Pembrolizumab cutaneous adverse events and their association with disease progression. JAMA Dermatol. 2015;151(11):1206–12.
Nakamura Y, Tanaka R, Asami Y, Teramoto Y, Imamura T, Sato S, et al. Correlation between vitiligo occurrence and clinical benefit in advanced melanoma patients treated with nivolumab: a multi-institutional retrospective study. J Dermatol. 2017;44(2):117–22.
Hua C, Boussemart L, Mateus C, Routier E, Boutros C, Cazenave H, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152(1):45–51.
Freeman-Keller M, Kim Y, Cronin H, Richards A, Gibney G, Weber JS. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Cancer Res. 2016;22(4):886–94.
Teulings HE, Limpens J, Jansen SN, Zwinderman AH, Reitsma JB, Spuls PI, et al. Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J Clin Oncol. 2015;33(7):773–81.
Sato K, Akamatsu H, Murakami E, Sasaki S, Kanai K, Hayata A, et al. Correlation between immune-related adverse events and efficacy in non-small cell lung cancer treated with nivolumab. Lung Cancer. 2018;115:71–4.
Teraoka S, Fujimoto D, Morimoto T, Kawachi H, Ito M, Sato Y, et al. Early immune-related adverse events and association with outcome in advanced non-small cell lung cancer patients treated with nivolumab: a prospective cohort study. J Thorac Oncol. 2017;12(12):1798–805. https://doi.org/10.1016/j.jtho.2017.08.022.
Haratani K, Hayashi H, Chiba Y, Kudo K, Yonesaka K, Kato R, et al. Association of immune-related adverse events with nivolumab efficacy in non-small cell lung cancer. JAMA Oncol. 2018;4(3):374–8.
Weber JS, Kähler KC, Hauschild A. Management of immune-related adverse events and kinetics of response with ipilimumab. J Clin Oncol. 2012;30(21):2691–7.
Eigentler TK, Hassel JC, Berking C, Aberle J, Bachmann O, Grünwald V, et al. Diagnosis, monitoring and management of immune-related adverse drug reactions of anti-PD-1 antibody therapy. Cancer Treat Rev. 2016;45:7–18. https://doi.org/10.1016/j.ctrv.2016.02.003.
Motzer RJ, Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S, et al. Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. 2015;373(19):1803–13.
Downey SG, Klapper JA, Smith FO, Yang JC, Sherry RM, Royal RE, et al. Prognostic factors related to clinical response in patients with metastatic melanoma treated by CTL-associated antigen-4 blockade. Clin Cancer Res. 2007;13(22):6681–8.
Yang JC, Hughes M, Kammula U, Royal R, Sherry RM, Topalian SL, et al. Ipilimumab (anti-CTLA4 antibody) causes regression of metastatic renal cell cancer associated with enteritis and hypophysitis. J Immunother. 2007;30:825–30.
Arriola E, Wheater M, Krishnan R, Smart J, Foria V, Ottensmeier C. Immunosuppression for ipilimumab-related toxicity can cause pneumocystis pneumonia but spare antitumor immune control. Oncoimmunology. 2015;4(10):e1040218.
Faje AT, Lawrence D, Flaherty K, Freedman C, Fadden R, Rubin K, et al. High-dose glucocorticoids for the treatment of ipilimumab-induced hypophysitis is associated with reduced survival in patients with melanoma. Cancer. 2018;124(18):3706–14.
Japanese Ministry of Health, Labor and Welfare Ethical guidelines for medical and health research involving human subjects. https://www.mhlw.go.jp/file/06-Seisakujouhou-10600000-Daijinkanboukouseikagakuka/0000153339.pdf. https://www.mhlw.go.jp/file/06-Seisakujouhou-10600000-Daijinkanboukouseikagakuka/0000080278.pdf (2020). Accessed 12 Jan 2020
Funding
This work had no Grant support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Yasushi Goto received honoraria from AstraZeneca, Pfizer; Shintaro Kanda received honoraria from AstraZeneca, Bristol-Myers Squibb, Ono Pharmaceutical and grants from AstraZeneca, Bristol-Myers Squibb, Ono Pharmaceutical; Hidehito Horinouchi received honoraria from AstraZeneca, Bristol-Myers Squibb, MSD, Ono Pharmaceutical and grants from Chugai Pharma, MSD, Ono Pharmaceutical, Taiho Pharmaceutical; Yutaka Fujiwara received honoraria from AstraZeneca, Bristol-Myers Squibb, Ono Pharmaceutical and grants from AstraZeneca, Bristol-Myers Squibb, Chugai Pharma, MSD; Noboru Yamamoto received honoraria from AstraZeneca, Bristol-Myers Squibb, Chugai Pharma, Eli Lilly, Ono Pharmaceutical, Pfizer and grants from Astellas, AvvVie, Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai, Daiichi-Sankyo, Eisai, Janssen Pharma, Kyowa-Hakko Kirin, MERCK, MSD, Novartis, Ono Pharmaceutical, Pfizer, Taiho Pharmaceutical, Takeda; Yuichiro Ohe received honoraria from AstraZeneca, Chugai Pharma, Ono Pharmaceutical and grants from AstraZeneca, Bristol-Myers Squibb, Chugai Pharma, MSD, Ono Pharmaceutical, Taiho Pharmaceutical. The other authors declare no conflict of interest for this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This retrospective study was approved by the Institutional Review Board of NCCH (2015–355).
Informed consent
Since this study was not utilizing human biological specimens, not involving invasiveness, and not involving intervention, formal consent was not required. The study was conducted in compliance with the “Ethical guidelines for medical and health research involving human subjects” [29].
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ida, H., Goto, Y., Sato, J. et al. Clinical characteristics of adrenal insufficiency as an immune-related adverse event in non-small-cell lung cancer. Med Oncol 37, 30 (2020). https://doi.org/10.1007/s12032-020-01357-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-020-01357-x