Abstract
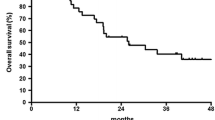
We performed a phase II trial to evaluate a docetaxel-based regimen in locoregionally advanced esophageal cancer. Untreated stage II–IVa esophageal cancer patients with performance status 0–2 were included. Tumor resectability was determined prior to initiation of study. Induction docetaxel (75 mg/m2) and cisplatin (75 mg/m2) day 1 with prophylactic filgrastim was delivered every 21 days for 3 cycles. Subsequent concomitant chemoradiotherapy (CRT) utilized weekly docetaxel (20 mg/m2) and concurrent radiotherapy (2 Gy/day) in resectable/resected patients (50 Gy) and in unresectable patients (66 Gy). A total of 78 patients (15 squamous cell carcinoma, 60 adenocarcinoma, 3 mixed/undifferentiated; 68 men, 10 women; median age 61 years) were accrued. The regimen was administered to 59 (76%) potentially resectable patients and 13 (17%) unresectable patients; 6 patients (8%) received the regimen post-operatively. Response rate in 66 evaluable patients following induction chemotherapy was 30%. Sixty-nine patients underwent CRT. Ten patients had disease progression during CRT. Forty-five out of 59 potentially resectable patients underwent esophagectomy after CRT, and 42 patients had complete tumor resection with negative margins. Eighteen out of 59 patients who were potentially resectable patients had pathologic complete response (pCR—31%). Grade 3/4 toxicity during induction chemotherapy included leucopenia, neutropenia, vomiting, and neuropathy. Esophagitis was the predominant toxicity during CRT. Median overall survival was 11.4 months for unresectable patients, 14.3 months for resectable patients and 10.4 months for patients who received the regimen post-operatively (log-rank P = 0.2492). Docetaxel-based CRT regimen is active and tolerable in esophageal cancer. The observed pCR in the potentially resectable group indicates good local control.



Similar content being viewed by others
References
Cooper JS, Guo MD, Herskovic A, Macdonald JS, Martenson JA Jr, Al-Sarraf M, et al. Chemoradiotherapy of locally advanced esophageal cancer: long-term follow-up of a prospective randomized trial (RTOG 85–01). Radiation Therapy Oncology Group. JAMA. 1999;281(17):1623–7.
Walsh TN, Noonan N, Hollywood D, Kelly A, Keeling N, Hennessy TP. A comparison of multimodal therapy and surgery for esophageal adenocarcinoma. N Engl J Med. 1996;335(7):462–7.
Bosset JF, Gignoux M, Triboulet JP, Tiret E, Mantion G, Elias D, et al. Chemoradiotherapy followed by surgery compared with surgery alone in squamous-cell cancer of the esophagus. N Engl J Med. 1997;337(3):161–7.
Macdonald JS, Smalley SR, Benedetti J, Hundahl SA, Estes NC, Stemmermann GN, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345(10):725–30.
Malthaner RA, Wong RK, Rumble RB, Zuraw L. Neoadjuvant or adjuvant therapy for resectable esophageal cancer: a systematic review and meta-analysis. BMC Med. 2004;2(1):35.
Graham AJ, Shrive FM, Ghali WA, Manns BJ, Grondin SC, Finley RJ, et al. Defining the optimal treatment of locally advanced esophageal cancer: a systematic review and decision analysis. Ann Thorac Surg. 2007;83(4):1257–64.
Gebski V, Burmeister B, Smithers BM, Foo K, Zalcberg J, Simes J. Survival benefits from neoadjuvant chemoradiotherapy or chemotherapy in oesophageal carcinoma: a meta-analysis. Lancet Oncol. 2007;8(3):226–34.
Das P, Fukami N, Ajani JA. Combined modality therapy of localized gastric and esophageal cancers. J Natl Compr Canc Netw. 2006;4(4):375–82.
Kavanagh B, Anscher M, Leopold K, Deutsch M, Gaydica E, Dodge R, et al. Patterns of failure following combined modality therapy for esophageal cancer, 1984–1990. Int J Radiat Oncol Biol Phys. 1992;24(4):633–42.
Medical Research Council Oesophageal Cancer Working Group. Surgical resection with or without preoperative chemotherapy in oesophageal cancer: a randomised controlled trial. Lancet. 2002;359(9319):1727–33.
Jin J, Liao Z, Zhang Z, Ajani J, Swisher S, Chang JY, et al. Induction chemotherapy improved outcomes of patients with resectable esophageal cancer who received chemoradiotherapy followed by surgery. Int J Radiat Oncol Biol Phys. 2004;60(2):427–36.
Kelsen DP, Ginsberg R, Pajak TF, Sheahan DG, Gunderson L, Mortimer J, et al. Chemotherapy followed by surgery compared with surgery alone for localized esophageal cancer. N Engl J Med. 1998;339(27):1979–84.
Law S, Fok M, Chow S, Chu KM, Wong J. Preoperative chemotherapy versus surgical therapy alone for squamous cell carcinoma of the esophagus: a prospective randomized trial. J Thorac Cardiovasc Surg. 1997;114(2):210–7.
Ancona E, Ruol A, Santi S, Merigliano S, Sileni VC, Koussis H, et al. Only pathologic complete response to neoadjuvant chemotherapy improves significantly the long term survival of patients with resectable esophageal squamous cell carcinoma: final report of a randomized, controlled trial of preoperative chemotherapy versus surgery alone. Cancer. 2001;91(11):2165–74.
Creane M, Seymour CB, Colucci S, Mothersill C. Radiobiological effects of docetaxel (Taxotere): a potential radiation sensitizer. Int J Radiat Biol. 1999;75(6):731–7.
Pradier O, Rave-Frank M, Lehmann J, Lucke E, Boghun O, Hess CF, et al. Effects of docetaxel in combination with radiation on human head and neck cancer cells (ZMK-1) and cervical squamous cell carcinoma cells (CaSki). Int J Cancer. 2001;91(6):840–5.
Einzig AI, Neuberg D, Remick SC, Karp DD, O’Dwyer PJ, Stewart JA, et al. Phase II trial of docetaxel (Taxotere) in patients with adenocarcinoma of the upper gastrointestinal tract previously untreated with cytotoxic chemotherapy: the Eastern Cooperative Oncology Group (ECOG) results of protocol E1293. Med Oncol. 1996;13(2):87–93.
Muro K, Hamaguchi T, Ohtsu A, Boku N, Chin K, Hyodo I, et al. A phase II study of single-agent docetaxel in patients with metastatic esophageal cancer. Ann Oncol. 2004;15(6):955–9.
Schull B, Kornek GV, Schmid K, Raderer M, Hejna M, Lenauer A, et al. Effective combination chemotherapy with bimonthly docetaxel and cisplatin with or without hematopoietic growth factor support in patients with advanced gastroesophageal cancer. Oncology. 2003;65(3):211–7.
Mauer AM, Masters GA, Haraf DJ, Hoffman PC, Watson SM, Golomb HM, et al. Phase I study of docetaxel with concomitant thoracic radiation therapy. J Clin Oncol. 1998;16(1):159–64.
Beahrs OH, American Joint Committee on Cancer, American Cancer Society, National Cancer Institute (U.S.). Manual for staging of cancer. 4th ed. Philadelphia: Lippincott; 1992.
Brunsvig PF, Hatlevoll R, Berg R, Lauvvang G, Owre K, Wang M, et al. Weekly docetaxel with concurrent radiotherapy in locally advanced non-small cell lung cancer: a phase I/II study with 5 years’ follow-up. Lung Cancer. 2005;50(1):97–105.
Koukourakis MI, Bahlitzanakis N, Froudarakis M, Giatromanolaki A, Georgoulias V, Koumiotaki S, et al. Concurrent conventionally fractionated radiotherapy and weekly docetaxel in the treatment of stage IIIb non-small-cell lung carcinoma. Br J Cancer. 1999;80(11):1792–6.
Font A, Arellano A, Fernandez-Llamazares J, Casas D, Boix J, Cardenal J, et al. Weekly docetaxel with concomitant radiotherapy in patients with inoperable oesophageal cancer. Clin Transl Oncol. 2007;9(3):177–82.
Ruhstaller T, Widmer L, Schuller JC, Roth A, Hess V, Mingrone W, et al. Multicenter phase II trial of preoperative induction chemotherapy followed by chemoradiation with docetaxel and cisplatin for locally advanced esophageal carcinoma (SAKK 75/02). Ann Oncol. 2009;20(9):1522–8.
Minsky BD, Pajak TF, Ginsberg RJ, Pisansky TM, Martenson J, Komaki R, et al. INT 0123 (Radiation Therapy Oncology Group 94-05) phase III trial of combined-modality therapy for esophageal cancer: high-dose versus standard-dose radiation therapy. J Clin Oncol. 2002;20(5):1167–74.
Onishi H, Kuriyama K, Yamaguchi M, Komiyama T, Tanaka S, Araki T, et al. Concurrent two-dimensional radiotherapy and weekly docetaxel in the treatment of stage III non-small cell lung cancer: a good local response but no good survival due to radiation pneumonitis. Lung Cancer. 2003;40(1):79–84.
Taghian AG, Assaad SI, Niemierko A, Kuter I, Younger J, Schoenthaler R, et al. Risk of pneumonitis in breast cancer patients treated with radiation therapy and combination chemotherapy with paclitaxel. J Natl Cancer Inst. 2001;93(23):1806–11.
Wright CD, Wain JC, Lynch TJ, Choi NC, Grossbard ML, Carey RW, et al. Induction therapy for esophageal cancer with paclitaxel and hyperfractionated radiotherapy: a phase I and II study. J Thorac Cardiovasc Surg. 1997;114(5):811–5. discussion 6.
Choi N, Park SD, Lynch T, Wright C, Ancukiewicz M, Wain J, et al. Twice-daily radiotherapy as concurrent boost technique during two chemotherapy cycles in neoadjuvant chemoradiotherapy for resectable esophageal carcinoma: mature results of phase II study. Int J Radiat Oncol Biol Phys. 2004;60(1):111–22.
Abou-Jawde RM, Mekhail T, Adelstein DJ, Rybicki LA, Mazzone PJ, Caroll MA, et al. Impact of induction concurrent chemoradiotherapy on pulmonary function and postoperative acute respiratory complications in esophageal cancer. Chest. 2005;128(1):250–5.
Geh JI, Crellin AM, Glynne-Jones R. Preoperative (neoadjuvant) chemoradiotherapy in oesophageal cancer. Br J Surg. 2001;88(3):338–56.
Flamen P, Lerut A, Van Cutsem E, De Wever W, Peeters M, Stroobants S, et al. Utility of positron emission tomography for the staging of patients with potentially operable esophageal carcinoma. J Clin Oncol. 2000;18(18):3202–10.
Heath EI, Burtness BA, Heitmiller RF, Salem R, Kleinberg L, Knisely JP, et al. Phase II evaluation of preoperative chemoradiation and postoperative adjuvant chemotherapy for squamous cell and adenocarcinoma of the esophagus. J Clin Oncol. 2000;18(4):868–76.
Meluch AA, Greco FA, Gray JR, Thomas M, Sutton VM, Davis JL, et al. Preoperative therapy with concurrent paclitaxel/carboplatin/infusional 5-FU and radiation therapy in locoregional esophageal cancer: final results of a Minnie Pearl Cancer Research Network phase II trial. Cancer J. 2003;9(4):251–60.
Kim DW, Blanke CD, Wu H, Shyr Y, Berlin J, Beauchamp RD, et al. Phase II study of preoperative paclitaxel/cisplatin with radiotherapy in locally advanced esophageal cancer. Int J Radiat Oncol Biol Phys. 2007;67(2):397–404.
Swisher SG, Ajani JA, Komaki R, Nesbitt JC, Correa AM, Cox JD, et al. Long-term outcome of phase II trial evaluating chemotherapy, chemoradiotherapy, and surgery for locoregionally advanced esophageal cancer. Int J Radiat Oncol Biol Phys. 2003;57(1):120–7.
Wang H, Ryu J, Gandara D, Bold RJ, Urayama S, Tanaka M, et al. A phase II study of paclitaxel, carboplatin, and radiation with or without surgery for esophageal cancer. J Thorac Oncol. 2007;2(2):153–7.
Herskovic A, Martz K, al-Sarraf M, Leichman L, Brindle J, Vaitkevicius V, et al. Combined chemotherapy and radiotherapy compared with radiotherapy alone in patients with cancer of the esophagus. N Engl J Med. 1992;326(24):1593–8.
al-Sarraf M, Martz K, Herskovic A, Leichman L, Brindle JS, Vaitkevicius VK, et al. Progress report of combined chemoradiotherapy versus radiotherapy alone in patients with esophageal cancer: an intergroup study. J Clin Oncol. 1997;15(1):277–84.
Suntharalingam M, Moughan J, Coia LR, Krasna MJ, Kachnic L, Haller DG, et al. The national practice for patients receiving radiation therapy for carcinoma of the esophagus: results of the 1996–1999 patterns of care study. Int J Radiat Oncol Biol Phys. 2003;56(4):981–7.
Coia LR, Minsky BD, Berkey BA, John MJ, Haller D, Landry J, et al. Outcome of patients receiving radiation for cancer of the esophagus: results of the 1992–1994 patterns of care study. J Clin Oncol. 2000;18(3):455–62.
Bedenne L, Michel P, Bouche O, Milan C, Mariette C, Conroy T, et al. Chemoradiation followed by surgery compared with chemoradiation alone in squamous cancer of the esophagus: FFCD 9102. J Clin Oncol. 2007;25(10):1160–8.
Stahl M, Stuschke M, Lehmann N, Meyer HJ, Walz MK, Seeber S, et al. Chemoradiation with and without surgery in patients with locally advanced squamous cell carcinoma of the esophagus. J Clin Oncol. 2005;23(10):2310–7.
Acknowledgments
This study was supported by Sanofi-Aventis, University of Chicago Cancer Center (CM107102-02), and Valda and Robert Svendsen Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choong, N.W., Mauer, A.M., Haraf, D.C. et al. Long-term outcome of a phase II study of docetaxel-based multimodality chemoradiotherapy for locally advanced carcinoma of the esophagus or gastroesophageal junction. Med Oncol 28 (Suppl 1), 152–161 (2011). https://doi.org/10.1007/s12032-010-9658-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-010-9658-1