Abstract
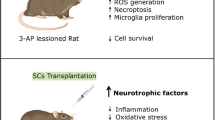
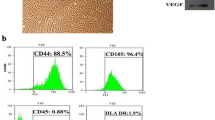
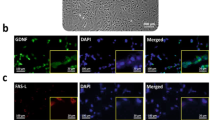
Huntington’s disease (HD) is a genetically heritable disorder, linked with continuing cell loss and degeneration mostly in the striatum. Currently, cell therapy approaches in HD have essentially been focused on replenishing or shielding cells lost over the period of the disease. Herein, we sought to explore the in vitro and in vivo efficacy of primary rat Sertoli cells (SCs) and their paracrine effect against oxidative stress with emphasis on HD. Initially, SCs were isolated and immunophenotypically characterized by positive expression of GATA4. Besides, synthesis of neurotrophic factors of glial cell-derived neurotrophic factor and VEGF by SCs were proved. Next, PC12 cells were exposed to hydrogen peroxide in the presence of conditioned media (CM) collected from SC (SC-CM) and cell viability and neuritogenesis were determined. Bilateral striatal implantation of SC in 3-nitropropionic acid (3-NP)-lesioned rat models was performed, and 1 month later, post-graft analysis was done. According to our in vitro results, the CM of SC protected PC12 cells against oxidative stress and remarkably augmented cell viability and neurite outgrowth. Moreover, grafted SCs survived, exhibited decreases in both gliosis and inflammatory cytokine levels, and ameliorated motor coordination and muscle activity, together with an increase in striatal volume as well as in dendritic length of the striatum in HD rats. In conclusion, our results indicate that SCs provide a supportive environment, with potential therapeutic benefits aimed at HD.











Similar content being viewed by others
References
Aslani F, Sebastian T, Keidel M, Fröhlich S, Elsässer HP, Schuppe HC, Klug J, Mahavadi P, Fijak M, Bergmann M, Meinhardt A, Bhushan S (2017) Resistance to apoptosis and autophagy leads to enhanced survival in Sertoli cells. Mol Hum Rep 23:370–380
Bates G, Tabrizi S, Jones L (eds) (2014). Huntington's disease (No. 64). Oxford University Press, UK
Borlongan CV, Koutouzis TK, Freeman TB, Cahill DW, Sanberg PR (1995) Behavioral pathology induced by repeated systemic injections of 3-nitropropionic acid mimics the motoric symptoms of Huntington’s disease. Brain Res 697:254–257
Borlongan CV, Cameron DF, Saporta S, Sanberg PR (1997) Intracerebral transplantation of testis-derived Sertoli cells promotes functional recovery in female rats with 6-hydroxydopamine-induced hemiparkinsonism. Exp Neurol 148:388–392
Boroujeni ME, Gardaneh M (2017) Umbilical cord: an unlimited source of cells differentiable towards dopaminergic neurons. Neur Reg Res 12:1186
Boroujeni ME, Gardaneh M (2018) The superiority of sucrose cushion centrifugation to ultrafiltration and PEGylation in generating high-titer lentivirus particles and transducing stem cells with enhanced efficiency. Mol Biotechnol 60:185. https://doi.org/10.1007/s12033-017-0044-5
Buytaert-Hoefen KA, Alvarez E, Freed CR (2004) Generation of tyrosine hydroxylase positive neurons from human embryonic stem cells after coculture with cellular substrates and exposure to GDNF. Stem Cells 22:669–674
Chiappalupi S, Luca G, Mancuso F, Madaro L, Fallarino F, Nicoletti C, Calvitti M, Arato I, Falabella G, Salvadori L, di Meo A, Bufalari A, Giovagnoli S, Calafiore R, Donato R, Sorci G (2016) Intraperitoneal injection of microencapsulated Sertoli cells restores muscle morphology and performance in dystrophic mice. Biomaterials 75:313–326
Clelland CD, Barker RA, Watts C (2008) Cell therapy in Huntington disease. Neurosurg Focus 24:E9
Cudicini C, Kercret H, Touzalin AM, Ballet F, Jégou B (1997) Vectorial production of interleukin 1 and interleukin 6 by rat Sertoli cells cultured in a dual culture compartment system. Endocrinology 138:2863–2870
Doyle TJ, Kaur G, Putrevu SM, Dyson EL, Dyson M, McCunniff WT et al (2012) Immunoprotective properties of primary Sertoli cells in mice: potential functional pathways that confer immune privilege. Biol Reprod 86(6):1–14
Dufour JM, Dass B, Halley KR, Korbutt GS, Dixon DE, Rajotte RV (2008) Sertoli cell line lacks the immunoprotective properties associated with primary Sertoli cells. Cell Transplant 17:525–534
Emerich DF (2003) Sertoli cell grafts for Huntington’s disease. An opinion. Neurotox Res 5:567
Emerich DF, Hemendinger R, Halberstadt CR (2003) The testicular-derived Sertoli cell: cellular immunoscience to enable transplantation. Cell trans 12:335–349
French LE, Hahne M, Viard I, Radlgruber G, Zanone R, Becker K, Müller C, Tschopp J (1996) Fas and Fas ligand in embryos and adult mice: ligand expression in several immune-privileged tissues and coexpression in adult tissues characterized by apoptotic cell turnover. J Cell Biol 133:335–343
Gnessi L, Emidi A, Jannini EA, Carosa E, Maroder M, Arizzi M, Ulisse S, Spera G (1995) Testicular development involves the spatiotemporal control of PDGFs and PDGF receptors gene expression and action. J Cell Biol 131:1105–1121
Griswold MD (1993) The Sertoli cell. Cache River, Clearwater 801p
Hemendinger R, Wang J, Malik S, Persinski R, Copeland J, Emerich D, Gores P, Halberstadt C, Rosenfeld J (2005) Sertoli cells improve survival of motor neurons in SOD1 transgenic mice, a model of amyotrophic lateral sclerosis. Exp Neurol 196:235–243
Hynes M, Rosenthal A (1999) Specification of dopaminergic and serotonergic neurons in the vertebrate CNS. Curr Opin Neurobiol 9:26–36
Johasson M, Friedemann M, Hopper B, Strömberg I (1995) Effects of glial cell line-derived neurotrophic factor on developing and mature ventral mesencephalic grafts in oculo. Exp Neurol 134:25–34
Johnston DS, Olivas E, DiCandeloro P, Wright WW (2011) Stage-specific changes in GDNF expression by rat Sertoli cells: a possible regulator of the replication and differentiation of stem spermatogonia. Biol Reprod 85:763–769
Kirik D, Georgievska B, Rosenblad C, Björklund A (2001) Delayed infusion of GDNF promotes recovery of motor function in the partial lesion model of Parkinson’s disease. Eur J Neurosci 13:1589–1599
Korbutt GS, Elliott JF, Rajotte RV (1997) Cotransplantation of allogeneic islets with allogeneic testicular cell aggregates allows long-term graft survival without systemic immunosuppression. Diabetes 46:317–322
Kumar P, Kalonia H, Kumar A (2010) Huntington’s disease: pathogenesis to animal models. Pharmacol Rep 62:1–14
Leegwater-Kim J, Cha JH (2004) The paradigm of Huntington’s disease: therapeutic opportunities in neurodegeneration. NeuroRx 1:128–138
Lin LF, Doherty DH, Lile JD, Bektesh S, Collins F (1993) GDNF: a glial cell line-derived neurotrophic factor for midbrain dopaminergic neurons. Science 260:1130–1132
MacDonald ME, Christine M, Ambrose MP, Duyao RH, Myers CL et al (1993) A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 72:971–983
Mital P, Kaur G, Dufour JM (2010) Immunoprotective Sertoli cells: making allogeneic and xenogeneic transplantation feasible. Reproduction 139:495–504
O’Bryan MK, Gerdprasert O, Nikolic-Paterson DJ, Meinhardt A, Muir JA, Foulds LM, Phillips DJ, de Kretser DM, Hedger MP (2005) Cytokine profiles in the testes of rats treated with lipopolysaccharide reveal localized suppression of inflammatory responses. Am J Physiol Regul Integr Comp Physiol 288:R1744–R1R55
Piccirillo CA, Chang Y, Prud’homme GJ (1998) TGF-β1 somatic gene therapy prevents autoimmune disease in nonobese diabetic mice. J Immun 161:3950–3956
Rodriguez AI, Willing AE, Saporta S, Cameron DF, Sanberg PR (2003) Effects of Sertoli cell transplants in a 3-nitropropionic acid model of early Huntington’s disease: a preliminary study. Neurotox Res 5:443–450
Rosenblad C, Kirik D, Björklund A (2000) Sequential administration of GDNF into the substantia nigra and striatum promotes dopamine neuron survival and axonal sprouting but not striatal reinnervation or functional recovery in the partial 6-OHDA lesion model. Exp Neurol 161:503–516
Sanberg PR, Borlongan CV, Saporta S, Cameron DF (1996) Testis-derived Sertoli cells survive and provide localized immunoprotection for xenografts in rat brain. Nat Biotech 14:1692–1695
Sanberg PR, Borlongan CV, Othberg AI, Saporta S, Freeman TB, Cameron DF (1997a) Testis-derived Sertoli cells have a trophic effect on dopamine neurons and alleviate hemiparkinsonism in rats. Nat Med 3:1129–1132
Sanberg PR, Saporta S, Borlongan CV, Othberg AI, Allen RC, Cameron DF (1997b) The testis-derived cultured Sertoli cell as a natural Fas-L secreting cell for immunosuppressive cellular therapy. Cell trans 6:191–193
Shamekh R, Mallery J, Newcomb J, Hushen J, Saporta S, Cameron DF, Sanberg CD, Sanberg PR, Willing AE (2006) Enhancing tyrosine hydroxylase expression and survival of fetal ventral mesencephalon neurons with rat or porcine Sertoli cells in vitro. Brain Res 1096:1–10
Shamekh R, Samuel S, Cameron DF, Willing AE, Sanberg CD, Johe K, Sanberg PR (2008) Effects of Sertoli cell-conditioned medium on ventral midbrain neural stem cells: a preliminary report. Neurotox Res 13:241–246
Skinner MK (1993) Secretion of growth factors and other regulatory factors. In: Russell LD, Griswold MD (eds) The Sertoli cell. Cache River, Clearwater, pp 237–248
Suarez-Pinzon W, Korbutt GS, Power R, Hooton J, Rajotte RV, Rabinovitch A (2000) Testicular Sertoli cells protect islet beta-cells from autoimmune destruction in NOD mice by a transforming growth factor-beta1-dependent mechanism. Diabetes 49:1810–1818
Trupp M, Rydén M, Jörnvall H, Funakoshi H, Timmusk T, Arenas E, Ibáñez CF (1995) Peripheral expression and biological activities of GDNF, a new neurotrophic factor for avian and mammalian peripheral neurons. J Cell Biol 130:137–148
Widenfalk J, Nosrat C, Tomac A, Westphal H, Hoffer B, Olson L (1997) Neurturin and glial cell line-derived neurotrophic factor receptor-β (GDNFR-β), novel proteins related to GDNF and GDNFR-α with specific cellular patterns of expression suggesting roles in the developing and adult nervous system and in peripheral organs. J Neurosci 17:8506–8519
Willing AE, Othberg AI, Samuel S, Anton A, Sinibaldi S, Poulos SG et al (1999a) Sertoli cells enhance the survival of co-transplanted dopamine neurons. Brain Res 822:246–250
Willing AE, Sudberry JJ, Othberg AI, Saporta S, Poulos SG, Cameron DF, Freeman TB, Sanberg PR (1999b) Sertoli cells decrease microglial response and increase engraftment of human hNT neurons in the hemiparkinsonian rat striatum. Brain Res Bull 48:441–444
Yang H, Al-Jazaeri A, Wright JR Jr (2002) The immunoprotective effect of Sertoli cells coencapsulated with islet xenografts is not dependent upon Fas ligand expression. Cell Transplant 11:799–801
Yue F, Cui L, Johkura K, Naoko Ogiwara N, Sasaki K (2006) Induction of midbrain dopaminergic neurons from primate embryonic stem cells by coculture with Sertoli cells. Stem Cells 24:1695–1706
Acknowledgements
This project is part of the M.S.C thesis of H. Ahmadi.
Funding
This study was funded by the Vice Chancellor of Research of the Shahid Beheshti University of Medical Sciences (grant number 11621).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Approval
All procedures performed in this study involving animals were in accordance with the ethical standards of the ethics committee of Shahid Beheshti University of Medical Sciences and under approval no. IR.SBMU.MSP.REC.1396.604.
Rights and permissions
About this article
Cite this article
Ahmadi, H., Boroujeni, M.E., Sadeghi, Y. et al. Sertoli Cells Avert Neuroinflammation-Induced Cell Death and Improve Motor Function and Striatal Atrophy in Rat Model of Huntington Disease. J Mol Neurosci 65, 17–27 (2018). https://doi.org/10.1007/s12031-018-1062-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1062-x