Abstract
Backgrounds
Gastric cancer is a frequent cause of cancer in Brazil. The understanding of gastric carcinogenesis is not completely known but the progress of the molecular biology has provided that the initiation and progression of gastric cancer process is a consequence of a cumulative series of multiple gene alterations.
Aim
The aim of the study is to investigate the relationship among cytoplasmatic COX-1 and COX-2, Bcl-2 and nuclear P53 in chronic gastritis, metaplasia, and intestinal and gastric cancer.
Patients and Methods
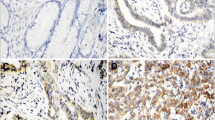
COX-1, COX-2, P53, and Bcl-2 were evaluated by immunohistochemistry in 34 gastric adenocarcinoma (GA) tissues obtained from gastric resection, 21 tissues of patients with chronic gastritis (CG), and 34 with intestinal metaplasia (IM) obtained from endoscopic biopsies.
Results
COX-1 and COX-2 were expressed in more than 85% of the tissues. A correlation between COX-1 and COX-2 were observed (r = 0.66). P53 was positive in 29% CG, 20% of IM and in 59 % of GA. Bcl-2 was negative in all the CG, in 88% of IM, and in 85% of GA. P53 staining was expressed more frequently in gastric cancer when compared to CG (p = 0.05) or IM (p = 0.003). The expression of Bcl-2 was also higher in gastric cancer (p = 0.002) and in intestinal metaplasia (p = 0.04) when compared to CG. There were no difference between metaplasia and chronic gastritis for P53 or Bcl-2. The imunoreactivity of COX-2 in gastric cancer was higher in the intestinal type (58%) than in diffuse type. A higher expression of COX-2 was found in advanced gastric cancer (p = 0.019). P53 was also more frequent in node positive cancer (p = 0.04).
Conclusion
COX-2 is probably involved in gastric carcinogenesis, being an early alteration in cancer. Although we observed in this study a correlation between COX-2 and depth of cancer, this association as a prognostic marker is not well defined. P53 and Bcl-2 was expressed mainly in gastric cancer, being probably a latest alteration in gastric development.




Similar content being viewed by others
References
INCA, Estimativas 2008: Incidência de Câncer no Brasil. 2008, Ministério da Saúde: Rio de Janeiro. http://www.inca.gov.br.
Arber N, et al. Celecoxib for the prevention of colorectal adenomatous polyps. N Engl J Med. 2006. 355(9):885–95. doi:10.1056/NEJMoa061652.
Miller EA, et al. Calcium, dietary, and lifestyle factors in the prevention of colorectal adenomas. Cancer 2007. 109(3):510–7. doi:10.1002/cncr.22453.
Vinogradova Y, et al. Risk of colorectal cancer in patients prescribed statins, nonsteroidal anti-inflammatory drugs, and cyclooxygenase-2 inhibitors: nested case-control study. Gastroenterology 2007. 133(2):393–402. doi:10.1053/j.gastro.2007.05.023.
Fujimura T, et al. Role of cyclooxygenase-2 in the carcinogenesis of gastrointestinal tract cancers: a review and report of personal experience. World J Gastroenterol. 2006;12(9):1336–45.
Futagami S, et al. Chemopreventive effect of celecoxib in gastric cancer. Inflammopharmacology 2007. 15(1):1–4. doi:10.1007/s10787-006-1541-5.
Lima-Rodrigues M, et al. A new model of laryngitis: neuropeptide, cyclooxygenase, and cytokine profile. Laryngoscope 2008;118(1):78–86.
Eblin KE, et al. Mitogenic signal transduction caused by monomethylarsonous acid in human bladder cells: role in arsenic-induced carcinogenesis. Toxicol Sci. 2007. 95(2):321–30. doi:10.1093/toxsci/kfl160.
Li GQ, et al. Effects of cyclooxygenase-1 and -2 gene disruption on Helicobacter pylori-induced gastric inflammation. J Infect Dis. 2006. 193(7):1037–46. doi:10.1086/500984.
Nasif WA, et al. Apoptosis deregulation in human gastric carcinomas: relationship to anti- and pro-apoptotic protein expression. Asian Pac J Cancer Prev. 2007;8(1):45–50.
Tzouvala M, et al. Potential role of apoptosis and apoptotic regulatory proteins in colorectal neoplasia: correlations with clinico-pathological parameters and survival. Dig Dis Sci. 2008. 53(2):451–60. doi:10.1007/s10620-007-9857-6.
Chen XL, et al. Relationship between expression and distribution of cyclooxygenase-2 and bcl-2 in human gastric adenocarcinoma. World J Gastroenterol. 2005;11(8):1228–31.
Shun CT, et al. Cyclooxygenase-2 expression correlates with nuclear p53 accumulation in gastric carcinoma. Hepatogastroenterology. 2003;50(52):988–92.
Sung JJ, et al. Cyclooxygenase-2 expression in Helicobacter pylori-associated premalignant and malignant gastric lesions. Am J Pathol. 2000;157(3):729–35.
Yamagata R, et al. Cyclooxygenase-2 expression is increased in early intestinal-type gastric cancer and gastric mucosa with intestinal metaplasia. Eur J Gastroenterol Hepatol. 2002. 14(4):359–63. doi:10.1097/00042737-200204000-00004.
Sun WH, et al. Roles of Helicobacter pylori infection and cyclooxygenase-2 expression in gastric carcinogenesis. World J Gastroenterol. 2004;10(19):2809–13.
Martins FP, et al. Over-expression of cyclooxygenase-2 in endoscopic biopsies of ectopic gastric mucosa. Braz J Med Biol Res. 2007. 40(11):1447–54. doi:10.1590/S0100-879X2007001100005.
Kawasaki T, et al. Cyclooxygenase-2 overexpression is common in serrated and non-serrated colorectal adenoma, but uncommon in hyperplastic polyp and sessile serrated polyp/adenoma. BMC Cancer. 2008. 8:33. doi:10.1186/1471-2407-8-33.
Ohno S, et al. Cyclooxygenase-2 expression correlates with apoptosis and angiogenesis in endometrial cancer tissue. Anticancer Res. 2007;27(6A):3765–70.
Sun Y, et al. Cyclooxygenase-2 overexpression reduces apoptotic susceptibility by inhibiting the cytochrome c-dependent apoptotic pathway in human colon cancer cells. Cancer Res. 2002;62(21):6323–8.
Yu HP, et al. Cyclooxygenase-2 expression in squamous dysplasia and squamous cell carcinoma of the esophagus. Cancer Lett. 2003. 198(2):193–201. doi:10.1016/S0304-3835(03)00340-9.
Kinoshita T, et al. Growth stimulation and induction of epidermal growth factor receptor by overexpression of cyclooxygenases 1 and 2 in human colon carcinoma cells. Biochim Biophys Acta. 1999;1438(1):120–30.
Joo YE, et al. Expression of cyclooxygenase-2, p53 and Ki-67 in gastric cancer. J Korean Med Sci. 2006;21(5):871–6.
Mao XY, et al. COX-2 expression in gastric cancer and its relationship with angiogenesis using tissue microarray. World J Gastroenterol. 2007;13(25):3466–71.
Li M, et al. Correlation of COX-2 and K-ras expression to clinical outcome in gastric cancer. Acta Oncol. 2006. 45(8):1115–9. doi:10.1080/02841860601043066.
Lim SC, et al. Expression of cyclooxygenase-2 and its relationship to p53 accumulation in colorectal cancers. Yonsei Med J. 2007. 48(3):495–501. doi:10.3349/ymj.2007.48.3.495.
Xiong B, et al. Expression of cyclooxygenase-2 in colorectal cancer and its clinical significance. World J Gastroenterol. 2005;11(8):1105–8.
Liang JT, et al. Microvessel density, cyclo-oxygenase 2 expression, K-ras mutation and p53 overexpression in colonic cancer. Br J Surg. 2004. 91(3):355–61. doi:10.1002/bjs.4447.
Mattioni M, et al. Serum anti-p53 antibodies as a useful marker for prognosis of gastric carcinoma. Int J Biol Markers. 2007;22(4):302–6.
Deveci MS, Deveci G. Prognostic value of p53 protein and MK-1 (a tumor-associated antigen) expression in gastric carcinoma. Gastric Cancer. 2007. 10(2):112–6. doi:10.1007/s10120-007-0418-7.
Potrc S, et al. Clinicopathological and immunohistochemical markers after radical gastrectomy for gastric cancer. Hepatogastroenterology 2007;54(73):308–14.
Milne AN, et al. Early-onset gastric cancers has a different molecular expression profile than conventional gastric cancers. Mod Pathol. 2006. 19(4):564–72. doi:10.1038/modpathol.3800563.
Leung WK, et al. Association between cyclo-oxygenase-2 overexpression and missense p53 mutations in gastric cancer. Br J Cancer. 2001. 84(3):335–9. doi:10.1054/bjoc.2000.1607.
Rajnakova A, et al. Expression of nitric oxide synthase, cyclooxygenase, and p53 in different stages of human gastric cancer. Cancer Lett. 2001. 172(2):177–85. doi:10.1016/S0304-3835(01)00645-0.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Forones, N.M., Kawamura, K.Y., Segreto, H.R.C. et al. Expression of COX-2 in Stomach Carcinogenesis. J Gastrointest Canc 39, 4–10 (2008). https://doi.org/10.1007/s12029-008-9039-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-008-9039-6