Abstract
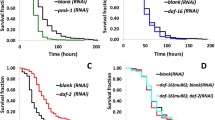
The c-Jun N-terminal kinase-mitogen-activated protein kinase (JNK-MAPK) pathway assists in modulating signals for growth, survival, and metabolism, thereby coordinating many cellular events during normal and stress conditions. To understand the role of the JNK-MAPK pathway during bacterial infection, an in vivo model organism Caenorhabditis elegans was used. In order to check the involvement of the JNK-MAPK pathway, the survival rate of C. elegans wild type (WT), and JNK-MAPK pathway mutant worms’ upon exposure to selective Gram-positive and Gram-negative pathogenic bacteria, was studied. Among the pathogens, Shigella flexneri M9OT was found to efficiently colonize inside the WT and JNK-MAPK pathway mutant worms. qPCR studies had suggested that the above pathway-specific genes kgb-2 and jnk-1 were prominently responsible for the immune response elicited by the host during the M9OT infection. In addition, daf-16, which is a major transcription factor of the insulin/insulin growth factor-1 signaling (IIS) pathway, was also found to be involved during the host response. Crosstalk between IIS and JNK-MAPK pathways has probably been involved in the activation of the host immune system, which consequently leads to lifespan extension. Furthermore, it is also observed that daf-16 activation by JNK-MAPK pathway leads to antimicrobial response, by activating lys-7 expression. These findings suggest that JNK-MAPK is not the sole pathway that enhances the immunity of the host. Nonetheless, the IIS pathway bridges the JNK-MAPK pathway that influences in protecting the host in counter to the M9OT infection.






Similar content being viewed by others
Abbreviations
- JNK:
-
C-Jun N-terminal kinase
- MAPK:
-
Mitogen-activated protein kinase
- IIS:
-
Insulin/insulin growth factor-1 signaling
- KGB-1:
-
Kinase, germline helicase binding
- MEK-1:
-
Mitogen-activated protein kinase kinase
- JKK-1:
-
C-Jun N-terminal kinase kinase
- DAF-16:
-
Abnormal dauer formation
References
Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124(4):783–801.
Balamurugan K, Sivamaruthi B, Durai S, Kesika P, Vigneshkumar B, JebaMercy G, et al. Understanding innate immune defenses against pathogens using model organism Caenorhabditis elegans. Spain: Formatex Publisher; 2013.
Kim DH, Feinbaum R, Alloing G, Emerson FE, Garsin DA, Inoue H, Tanaka-Hino M, Hisamoto N, Matsumoto K, Tan MW, Ausubel FM. A conserved p38 MAP kinase pathway in Caenorhabditis elegans innate immunity. Science. 2002;297(5581):623–6.
Troemel ER, Chu SW, Reinke V, Lee SS, Ausubel FM, Kim DH. p38 MAPK regulates expression of immune response genes and contributes to longevity in C. elegans. PLoS Genet. 2006;2(11):e183.
Van Der Hoeven R, McCallum KC, Cruz MR, Garsin DA. Ce-Duox1/BLI-3 generated reactive oxygen species trigger protective SKN-1 activity via p38 MAPK signaling during infection in C. elegans. PLoS Pathog. 2011;7(12):e1002453.
McMullan R, Anderson A, Nurrish S. Behavioral and immune responses to infection require Gαq-RhoA signaling in C. elegans. PLoS Pathog. 2012;8(2):e1002530.
Smith P, Leung-Chiu WM, Montgomery R, Orsborn A, Kuznicki K, Gressman-Coberly E, Mutapcic L, Bennett K. The GLH proteins, Caenorhabditis elegans P granule components, associate with CSN-5 and KGB-1, proteins necessary for fertility, and with ZYX-1, a predicted cytoskeletal protein. Dev Biol. 2002;251(2):333–47.
Li C, Hisamoto N, Nix P, Kanao S, Mizuno T, Bastiani M, Matsumoto K. The growth factor SVH-1 regulates axon regeneration in C. elegans via the JNK MAPK cascade. Nat Neurosci. 2012;15(4):551–7.
Gerke P, Keshet A, Mertenskötter A, Paul RJ. The JNK-like MAPK KGB-1 of Caenorhabditis elegans promotes reproduction, lifespan, and gene expressions for protein biosynthesis and germline homeostasis but interferes with hyperosmotic stress tolerance. Cell Physiol Biochem. 2014;34(6):1951–73.
Hattori A, Mizuno T, Akamatsu M, Hisamoto N, Matsumoto K. The Caenorhabditis elegans JNK signaling pathway activates expression of stress response genes by derepressing the Fos/HDAC repressor complex. PLoS Genet. 2013;9(2):e1003315.
Kao CY, Los FC, Huffman DL, Wachi S, Kloft N, Husmann M, Karabrahimi V, Schwartz JL, Bellier A, Ha C, Sagong Y. Global functional analyses of cellular responses to pore-forming toxins. PLoS Pathog. 2011;7(3):e1001314.
Villanueva A, Lozano J, Morales A, Lin X, Deng X, Hengartner MO, Kolesnick RN. jkk-1 and mek-1 regulate body movement coordination and response to heavy metals through jnk-1 in Caenorhabditis elegans. EMBO J. 2001;20(18):5114–28.
Kawasaki M, Hisamoto N, Iino Y, Yamamoto M, Ninomiya-Tsuji J, Matsumoto K. A Caenorhabditis elegans JNK signal transduction pathway regulates coordinated movement via type-D GABAergic motor neurons. EMBO J. 1999;18(13):3604–15.
Oh SW, Mukhopadhyay A, Svrzikapa N, Jiang F, Davis RJ, Tissenbaum HA. JNK regulates lifespan in Caenorhabditis elegans by modulating nuclear translocation of forkhead transcription factor/DAF-16. Proc Natl Acad Sci U S A. 2005;102(12):4494–9.
Neumann-Haefelin E, Qi W, Finkbeiner E, Walz G, Baumeister R, Hertweck M. SHC-1/p52Shc targets the insulin/IGF-1 and JNK signaling pathways to modulate life span and stress response in C. elegans. Genes Dev. 2008;22(19):2721–35.
Lee J, Kwon G, Lim YH. Elucidating the mechanism of Weissella-dependent lifespan extension in Caenorhabditis elegans. Sci Rep. 2015;5:17128.
Su S, Wink M. Natural lignans from Arctium lappa as antiaging agents in Caenorhabditis elegans. Phytochemistry. 2015;117:340–50.
Kong C, Tan MW, Nathan S. Orthosiphon stamineus protects Caenorhabditis elegans against Staphylococcus aureus infection through immunomodulation. Biol Open. 2014;3(7):644–55.
Mizuno T, Hisamoto N, Terada T, Kondo T, Adachi M, Nishida E, Kim DH, Ausubel FM, Matsumoto K. The Caenorhabditis elegans MAPK phosphatase VHP-1 mediates a novel JNK-like signaling pathway in stress response. EMBO J. 2004;23(11):2226–34.
Huffman DL, Abrami L, Sasik R, Corbeil J, van der Goot FG, Aroian RV. Mitogen-activated protein kinase pathways defend against bacterial pore-forming toxins. Proc Natl Acad Sci U S A. 2004;101(30):10995–1000.
Kim DH, Liberati NT, Mizuno T, Inoue H, Hisamoto N, Matsumoto K, Ausubel FM. Integration of Caenorhabditis elegans MAPK pathways mediating immunity and stress resistance by MEK-1 MAPK kinase and VHP-1 MAPK phosphatase. Proc Natl Acad Sci U S A. 2004;101(30):10990–4.
Visvikis O, Ihuegbu N, Labed SA, Luhachack LG, Alves AM, Wollenberg AC, Stuart LM, Stormo GD, Irazoqui JE. Innate host defense requires TFEB-mediated transcription of cytoprotective and antimicrobial genes. Immunity. 2014;40(6):896–909.
Twumasi-Boateng K, Wang TW, Tsai L, Lee KH, Salehpour A, Bhat S, Tan MW, Shapira M. An age-dependent reversal in the protective capacities of JNK signaling shortens Caenorhabditis elegans lifespan. Aging Cell. 2012;11(4):659–67.
Vadász I, Dada LA, Briva A, Helenius IT, Sharabi K, Welch LC, Kelly AM, Grzesik BA, Budinger GS, Liu J, Seeger W. Evolutionary conserved role of c-Jun-N-terminal kinase in CO2-induced epithelial dysfunction. PLoS One. 2012;7(10):e46696.
Uno M, Honjoh S, Matsuda M, Hoshikawa H, Kishimoto S, Yamamoto T, Ebisuya M, Yamamoto T, Matsumoto K, Nishida E. A fasting-responsive signaling pathway that extends life span in C. elegans. Cell Rep. 2013;3(1):79–91.
Melo JA, Ruvkun G. Inactivation of conserved genes induces microbial aversion, drug detoxification, and innate immunity in C. elegans. Cell. 2012;149(2):452.
Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974;77(1):71–94.
Stiernagle T. Maintenance of C. elegans. C elegans. 1999;2:51–67.
Kesika P, Balamurugan K. Studies on Shigella boydii infection in Caenorhabditis elegans and bioinformatics analysis of immune regulatory protein interactions. Biochim Biophys Acta. 2012;1824(12):1449–56.
Moy TI, Ball AR, Anklesaria Z, Casadei G, Lewis K, Ausubel FM. Identification of novel antimicrobials using a live-animal infection model. Proc Natl Acad Sci. 2006;103(27):10414–9.
Durai S, Singh N, Kundu S, Balamurugan K. Proteomic investigation of Vibrio alginolyticus challenged Caenorhabditis elegans revealed regulation of cellular homeostasis proteins and their role in supporting innate immune system. Proteomics. 2014;14(15):1820–32.
Burton EA, Pendergast AM, Aballay A. The Caenorhabditis elegans ABL-1 tyrosine kinase is required for Shigella flexneri pathogenesis. Appl Environ Microbiol. 2006;72(7):5043–51.
Irazoqui JE, Troemel ER, Feinbaum RL, Luhachack LG, Cezairliyan BO, Ausubel FM. Distinct pathogenesis and host responses during infection of C. elegans by P. aeruginosa and S. aureus. PLoS Pathog. 2010;6(7):e1000982.
JebaMercy G, Pandian SK, Balamurugan K. Changes in Caenorhabditis elegans life span and selective innate immune genes during Staphylococcus aureus infection. Folia Microbiol. 2011 Sep 1;56(5):373–80.
JebaMercy G, Vigneshwari L, Balamurugan K. A MAP kinase pathway in Caenorhabditis elegans is required for defense against infection by opportunistic Proteus species. Microbes Infect. 2013;15(8):550–68.
Sivamaruthi BS, Balamurugan K. Physiological and immunological regulations in Caenorhabditis elegans infected with Salmonella enterica serovar Typhi. Indian J Microbiol. 2014;54(1):52–8.
Wolf M, Nunes F, Henkel A, Heinick A, Paul RJ. The MAP kinase JNK-1 of Caenorhabditis elegans: location, activation, and influences over temperature-dependent insulin-like signaling, stress responses, and fitness. J Cell Physiol. 2008;214(3):721–9.
Evans EA, Kawli T, Tan MW. Pseudomonas aeruginosa suppresses host immunity by activating the DAF-2 insulin-like signaling pathway in Caenorhabditis elegans. PLoS Pathog. 2008;4(10):e1000175.
Portal-Celhay C, Bradley ER, Blaser MJ. Control of intestinal bacterial proliferation in regulation of lifespan in Caenorhabditis elegans. BMC Microbiol. 2012;12(1):1.
Durai S, Pandian SK, Balamurugan K. Establishment of a Caenorhabditis elegans infection model for Vibrio alginolyticus. J Basic Microbiol. 2011;51(3):243–52.
Zou CG, Ma YC, Dai LL, Zhang KQ. Autophagy protects C. elegans against necrosis during Pseudomonas aeruginosa infection. Proc Natl Acad Sci. 2014;111(34):12480–5.
Svensson C, Part K, Künnis-Beres K, Kaldmäe M, Fernaeus SZ, Land T. Pro-survival effects of JNK and p38 MAPK pathways in LPS-induced activation of BV-2 cells. Biochem Biophys Res Commun. 2011;406(3):488–92.
Kesika P, Prasanth MI, Balamurugan K. Modulation of Caenorhabditis elegans immune response and modification of Shigella endotoxin upon interaction. J Basic Microbiol. 2015;55(4):432–50.
Carayol N, Van Nhieu GT. Tips and tricks about Shigella invasion of epithelial cells. Curr Opin Microbiol. 2013;16(1):32–7.
Acknowledgements
We thank Caenorhabditis Genetics Center, which is funded by the National Institute of Health, and the National Center for Research Resources for providing C. elegans strains and E. coli OP50. SMP thanks the Department of Science and Technology (No. SR/FT/LS-83/2009(G)) for financial assistance through project JRF. SMP also would like to thank Udayakumar Prithika and Sekar Balaji for their constant support during the course of the experiment. The authors also thankfully acknowledge the computational facility provided by the Alagappa University Bioinformatics Infrastructure Facility (funded by DBT, Government of India; Grant No. BT/BI/25/001/2006). The Instrumentation Facility provided by the Department of Science and Technology, Government of India, through PURSE [Grant No.SR/S9Z-23/2010/42 (G)] and FIST (Grant No. SR-FST/LSI-087/2008) and University Grants Commission, New Delhi, through SAP-DRS1 [Grant No. F.3-28/2011 (SAP-II)] is greatly acknowledged. The authors also thank Dr. Maria Rescigno, European Institute of Oncology, Milano, Italy, for providing S. flexneri 5a M9OT:: DsRed strain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The study received partial financial support through the Extramural Research grants from DBT (BT/PR14932/MED/29/233/2010), ICMR (Sanction No: 5/3/3/13/2010-ECD-I), DST-SERB (No. SR/SO/AS-80/2010), UGC Major Research Project (No. 42-222/2013 (SR)), and CSIR (No. 37(1460)/11/EMR-II), New Delhi, India, by KB.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Marudhupandiyan, S., Balamurugan, K. Intrinsic JNK-MAPK pathway involvement requires daf-16-mediated immune response during Shigella flexneri infection in C. elegans . Immunol Res 65, 609–621 (2017). https://doi.org/10.1007/s12026-016-8879-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-016-8879-6