Abstract
Purpose
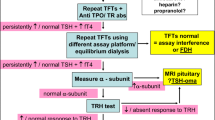
The polyethylene glycol (PEG) methodology is used for investigating incongruities in laboratory assays, such as thyroid-stimulating hormone (TSH) measurements. The aim of the study is to investigate the practical application of PEG-TSH testing in cases of discrepancies between elevated TSH and normal free thyroxine (FT4) levels.
Methods
A real-life observational study conducted in a tertiary medical center. The hospital’s electronic database was queried for TSH tests performed in pediatric patients between 2015 and 2023. Of those, PEG-TSH were identified. Patients’ clinical and biochemical characteristics and PEG-TSH-guided management were assessed.
Results
In total, 2949 TSH tests were performed in 891 children and adolescents for various indications. Among them were 61 (2.1%) PEG-TSH results, mean age 7.1 ± 5.3 years, of 38 patients (4.3%), comprised of 16 with congenital hypothyroidism, 16 with subclinical hypothyroidism, and 6 with Hashimoto thyroiditis. Both the TSH and the PEG-TSH levels of patients with congenital hypothyroidism were higher than those of the other two groups (P = 0.021 and P = 0.009, respectively), with no group differences in FT4 levels. Spearman’s correlation analysis revealed a strong association between TSH and PEG-TSH levels: r = 0.871, P < 0.001. In nearly one-half of the cases, clinical decisions made by clinicians (decreasing the dose or not initiating L-thyroxine treatment) were affected by the PEG-TSH results.
Conclusion
Our findings support PEG-TSH testing for determining appropriate TSH levels and avoid unnecessary thyroid hormone treatment among children and adolescents. We propose the suitability of managing their clinical condition based upon age-appropriate clinical parameters and FT4 levels when their PEG-TSH levels are within the normal range.



Similar content being viewed by others
References
C.M. Dayan, Interpretation of thyroid function tests. Lancet 357, 619–624 (2001). https://doi.org/10.1016/S0140-6736(00)04060-5
K. Kapelari, C. Kirchlechner, W. Högler, K. Schweitzer, I. Virgolini, R. Moncayo, Pediatric reference intervals for thyroid hormone levels from birth to adulthood: a retrospective study. BMC Endocr. Disord. 8, 15 (2008). https://doi.org/10.1186/1472-6823-8-15
R.P. Peeters, Subclinical hypothyroidism. N. Engl. J. Med. 376(26), 2556–2565 (2017). https://doi.org/10.1056/NEJMcp1611144
B. Biondi, A.R. Cappola, D.S. Cooper, Subclinical hypothyroidism: a review. JAMA 322, 153–160 (2019). https://doi.org/10.1001/jama.2019.9052
M. Salerno, N. Improda, D. Capalbo, management of endocrine disease subclinical hypothyroidism in children. Eur. J. Endocrinol. 183, R13–R28 (2020). https://doi.org/10.1530/EJE-20-0051
C.B. Larsen, E.R.B. Petersen, M. Overgaard, S.J. Bonnema, Macro-TSH: a diagnostic challenge. Eur. Thyroid J. 10, 93–97 (2021). https://doi.org/10.1159/000509184
D. Pluta, A. Bedkowska-Szczepanska, P. Madej, B. Zubelewicz-Szkodzinska, Macro-TSH - tips and tricks for gynecologists. Ginekol. Pol. 93, 601–602 (2022). https://doi.org/10.5603/GP.a2022.0003
R. D’Arcy, S. Hunter, K. Spence, M. McDonnell, A case of macro-TSH masquerading as subclinical hypothyroidism. BMJ Case Rep. 14, e243436 (2021). https://doi.org/10.1136/bcr-2021-243436
N. Hattori, T. Ishihara, K. Yamagami, A. Shimatsu, Macro TSH in patients with subclinical hypothyroidism. Clin. Endocrinol. 83, 923–930 (2015). https://doi.org/10.1111/cen.12643
T.P. Loh, S.L. Kao, D.J. Halsall, S.A.E.S. Toh, E. Chan, S.C. Ho, E.S. Tai, C.M. Khoo, Macro-thyrotropin: a case report and review of literature. J. Clin. Endocrinol. Metab. 97, 1823–1828 (2012). https://doi.org/10.1210/jc.2011-3490
A.A.A. Ismail, P.L. Walker, J.H. Barth, K.C. Lewandowski, R. Jones, W.A. Burr, Wrong biochemistry results: two case reports and observational study in 5310 patients on potentially misleading thyroid-stimulating hormone and gonadotropin immunoassay results. Clin. Chem. 48, 2023–2029 (2002)
V. Marks, False-positive immunoassay results: a multicenter survey of erroneous immunoassay results from assays of 74 analytes in 10 donors from 66 laboratories in seven countries. Clin. Chem. 48, 2008–2016 (2002)
O. Koulouri, M. Gurnell, How to interpret thyroid function tests. Clin. Med. 13, 282–286 (2013). https://doi.org/10.7861/clinmedicine.13-3-282
J. Tate, G. Ward, Interferences in immunoassay. Clin. Biochem. Rev. 25, 105–120 (2004)
K.A. Metwalley, H.S. Farghaly, Subclinical hypothyroidism in children: updates for pediatricians. Ann. Pediatr. Endocrinol. Metab. 26, 80–85 (2021). https://doi.org/10.6065/apem.2040242.121
A. Yamada, N. Hattori, T. Matsuda, N. Nishiyama, A. Shimatsu, Clearance of macro-TSH from the circulation is slower than TSH. Clin. Chem. Lab. Med. 60, E132–E135 (2022). https://doi.org/10.1515/cclm-2022-0131
J. Favresse, M.C. Burlacu, D. Maiter, D. Gruson, Interferences with thyroid function immunoassays: clinical implications and detection algorithm. Endocr. Rev. 39, 830–850 (2018). https://doi.org/10.1210/er.2018-00119
N. Hattori, T. Ishihara, Y. Saiki, Macroprolactinaemia: prevalence and aetiologies in a large group of hospital workers. Clin. Endocrinol. 71, 702–708 (2009). https://doi.org/10.1111/j.1365-2265.2009.03570.x
W.A. Marshall, J.M. Tanner, Variations in the pattern of pubertal changes in boys. Arch. Dis. Child. 45, 13–23 (1970). https://doi.org/10.1136/adc.45.239.13
W.A. Marshall, J.M. Tanner, Variations in pattern of pubertal changes in girls. Arch. Dis. Child. 44, 291–303 (1969). https://doi.org/10.1136/adc.44.235.291
A.E. Brown, M. Walker, Genetics of insulin resistance and the metabolic syndrome. Curr. Cardiol. Rep. 18 (2016). https://doi.org/10.1007/s11886-016-0755-4
J.H. Chou, S. Roumiantsev, R. Singh, PediTools electronic growth chart calculators: applications in clinical care, research, and quality improvement. J. Med. Internet Res. 22, e16204 (2020). https://doi.org/10.2196/16204
C.L. Ogden, R.J. Kuczmarski, K.M. Flegal, Z. Mei, S. Guo, R. Wei, L.M. Grummer-Strawn, L.R. Curtin, A.F. Roche, C.L. Johnson, Centers for Disease Control and Prevention 2000 growth charts for the United States: improvements to the 1977 National Center for Health Statistics version. Pediatrics 109, 45–60 (2002). https://doi.org/10.1542/peds.109.1.45
J.M. Tanner, H. Goldstein, R.H. Whitehouse, Standards for children’s height at ages 2-9 years allowing for heights of parents. Arch. Dis. Child. 45, 755–762 (1970). https://doi.org/10.1136/adc.45.244.755
P. Van Trotsenburg, A. Stoupa, J. Léger, T. Rohrer, C. Peters, L. Fugazzola, A. Cassio, C. Heinrichs, V. Beauloye, J. Pohlenz, P. Rodien, R. Coutant, G. Szinnai, P. Murray, B. Bartés, D. Luton, M. Salerno, L. De Sanctis, M. Vigone, H. Krude, L. Persani, and M. Polak, Congenital hypothyroidism: a 2020–2021 consensus guidelines update—an ENDO-European Reference Network initiative endorsed by the European Society for Pediatric Endocrinology and the European Society for Endocrinology. 31, 387–419 (2021). https://doi.org/10.1089/thy.2020.0333
J.G. Hollowell, N.W. Staehling, W.Dana Flanders, W.Harry Hannon, E.W. Gunter, C.A. Spencer, L.E. Braverman, Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J. Clin. Endocrinol. Metab. 87, 489–499 (2002). https://doi.org/10.1210/jcem.87.2.8182
D. Strich, S. Edri, D. Gillis, Current normal values for TSH and FT3 in children are too low: evidence from over 11,000 samples. J. Pediatr. Endocrinol. Metab. 25, 245–248 (2012). https://doi.org/10.1515/jpem-2011-0494
A.J. Lem, Y.B. De Rijke, H. Van Toor, M.A.J. De Ridder, T.J. Visser, A.C.S. Hokken-Koelega, Serum thyroid hormone levels in healthy children from birth to adulthood and in short children born small for gestational age. J. Clin. Endocrinol. Metab. 97, 3170–3178 (2012). https://doi.org/10.1210/jc.2012-1759
D.S. Cooper, B. Biondi, Subclinical thyroid disease. Lancet 379, 1142–1154 (2012). https://doi.org/10.1016/S0140-6736(11)60276-6
D.S. Cooper, Subclinical hypothyroidism. N. Engl. J. Med. 345, 260–265 (2001). https://doi.org/10.1056/NEJM200107263450406
J. Léger, A. Olivieri, M. Donaldson, T. Torresani, H. Krude, G. Van Vliet, M. Polak, G. Butler, European Society for Paediatric Endocrinology consensus guidelines on screening, diagnosis, and management of congenital hypothyroidism. Horm. Res. Paediatr. 81, 80–103 (2014). https://doi.org/10.1159/000358198
D. Bailey, D. Colantonio, L. Kyriakopoulou, A.H. Cohen, M.K. Chan, D. Armbruster, K. Adeli, Marked biological variance in endocrine and biochemical markers in childhood: establishment of pediatric reference intervals using healthy community children from the CALIPER cohort. Clin. Chem. 59, 1393–1405 (2013). https://doi.org/10.1373/clinchem.2013.204222
J.J. Bongers-Schokking, W.C.M. Resing, Y.B. De Rijke, M.A.J. De Ridder, S.M. De Muinck Keizer-Schrama, Cognitive development in congenital hypothyroidism: is overtreatment a greater threat than undertreatment? J. Clin. Endocrinol. Metab. 98, 4499–4506 (2013). https://doi.org/10.1210/jc.2013-2175
M. Álvarez, C. Iglesias Fernández, A. Rodríguez Sánchez, E. Dulín Íñiguez, M.D. Rodríguez Arnao, Episodes of overtreatment during the first six months in children with congenital hypothyroidism and their relationships with sustained attention and inhibitory control at school age. Horm. Res. Paediatr. 74, 114–120 (2010). https://doi.org/10.1159/000313370
J. Leger, A. Tar, M. Schlumberger, P. Czernichow, Control of thyroglobulin secretion in patients with ectopic thyroid gland. Pediatr. Res. 23, 266–269 (1988). https://doi.org/10.1203/00006450-198803000-00007
M.C. Vigone, D. Capalbo, G. Weber, M. Salerno, Mild hypothyroidism in childhood: who, when, and how should be treated? J. Endocr. Soc. 2, 1024–1039 (2018). https://doi.org/10.1210/js.2017-00471
J.R. Garber, R.H. Cobin, H. Gharib, J.V. Hennessey, I. Klein, J.I. Mechanick, R. Pessah-Pollack, P.A. Singer, K.A. Woeber, Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of clinical endocrinologists and the American Thyroid Association. Thyroid 22, 1200–1235 (2012). https://doi.org/10.1089/thy.2012.0205
S.H.S. Pearce, G. Brabant, L.H. Duntas, F. Monzani, R.P. Peeters, S. Razvi, J.-L. Wemeau, 2013 ETA guideline: management of subclinical hypothyroidism. Eur. Thyroid J. 2, 215–228 (2013). https://doi.org/10.1159/000356507
T. Wu, J.W. Flowers, F. Tudiver, J.L. Wilson, N. Punyasavatsut, Subclinical thyroid disorders and cognitive performance among adolescents in the United States. BMC Pediatr. 6, 12 (2006). https://doi.org/10.1186/1471-2431-6-12
L. Lazar, R.B.D. Frumkin, E. Battat, Y. Lebenthal, M. Phillip, J. Meyerovitch, Natural history of thyroid function tests over 5 years in a large pediatric cohort. J. Clin. Endocrinol. Metab. 94, 1678–1682 (2009). https://doi.org/10.1210/jc.2008-2615
A.A. Fryer, W.S.A. Smellie, Managing demand for laboratory tests: a laboratory toolkit. J. Clin. Pathol. 66, 62–67 (2013). https://doi.org/10.1136/jclinpath-2011-200524
B. Yeshoua, C. Bowman, J. Dullea, J. Ditkowsky, M. Shyu, H. Lam, W. Zhao, J.Y. Shin, A. Dunn, S. Tsega, A.S. Linker, M. Shah, Interventions to reduce repetitive ordering of low-value inpatient laboratory tests: a systematic review. BMJ Open Qual. 12, e002128 (2023). https://doi.org/10.1136/BMJOQ-2022-002128
M. Salinas, M. López-Garrigós, E. Flores, C. Leiva-Salinas, Uncritical request of thyroid laboratory tests may result in a major societal economic burden: results from a large population study in Spain. Clin. Lab. 63, 1139–1145 (2017). https://doi.org/10.7754/Clin.Lab.2017.170101
J.K. Kluesner, D.J. Beckman, J.M. Tate, A.A. Beauvais, M.I. Kravchenko, J.L. Wardian, S.D. Graybill, J.A. Colburn, I. Folaron, M.W. True, Analysis of current thyroid function test ordering practices. J. Eval. Clin. Pract. 24, 347–352 (2018). https://doi.org/10.1111/jep.12846
Acknowledgements
The authors thank Esther Eshkol for editorial assistance. A portion of this research has been accepted in abstract form for presentation at ESPE 2023, taking place from September 21st to 23rd, 2023 in The Hague, Netherlands.
Author information
Authors and Affiliations
Contributions
H.Z. and A.B. designed the study, contributed to the data used in the study, searched the literature, interpreted the data, and wrote the manuscript. G.S., A.S.-B., O.E., and Y.L. contributed to the data used in this article, to the discussion, reviewed, and edited the manuscript. All authors approved the final version. A.B. is the guarantor of this work, and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to participate
The Tel Aviv Sourasky Medical Center review board waived informed parental consent.
Ethical approval
This study protocol was reviewed and approved by the Tel Aviv Sourasky Medical Center review board, approval number TLV-0086-23.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zaitoon, H., Shefer, G., Segev-Becker, A. et al. Polyethylene glycol thyroid-stimulating hormone (PEG-TSH) testing in the management of pediatric thyroid dysfunction. Endocrine 84, 524–532 (2024). https://doi.org/10.1007/s12020-023-03575-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03575-5