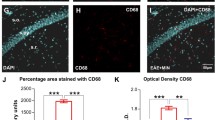
Abstract
The immune system shapes synaptic transmission and plasticity in experimental autoimmune encephalomyelitis (EAE), the mouse model of multiple sclerosis (MS). These synaptic adaptations are believed to drive recovery of function after brain lesions, and also learning and memory deficits and excitotoxic neurodegeneration; whether inflammation influences synaptic plasticity in MS patients is less clear. In a cohort of 59 patients with MS, we found that continuous theta-burst transcranial magnetic stimulation did not induce the expected long-term depression (LTD)-like synaptic phenomenon, but caused persisting enhancement of brain cortical excitability. The amplitude of this long-term potentiation (LTP)-like synaptic phenomenon correlated with the concentration of the pro-inflammatory cytokine interleukin-1β (IL-1β) in the cerebrospinal fluid. In MS and EAE, the brain and spinal cord are typically enriched of CD3+ T lymphocyte infiltrates, which are, along with activated microglia and astroglia, a major cause of inflammation. Here, we found a correlation between the presence of infiltrating T lymphocytes in the hippocampus of EAE mice and synaptic plasticity alterations. We observed that T lymphocytes from EAE, but not from control mice, release IL-1β and promote LTP appearance over LTD, thereby mimicking the facilitated LTP induction observed in the cortex of MS patients. EAE-specific T lymphocytes were able to suppress GABAergic transmission in an IL-1β-dependent manner, providing a possible synaptic mechanism able to lower the threshold of LTP induction in MS brains. Moreover, in vivo blockade of IL-1β signaling resulted in inflammation and synaptopathy recovery in EAE hippocampus. These data provide novel insights into the pathophysiology of MS.






Similar content being viewed by others
References
Aktas, O., & Zipp, F. (2003). Regulation of self-reactive T cells by human immunoglobulins-implications for multiple sclerosis therapy. Current Pharmaceutical Design, 9(3), 245–256.
Balducci, C., Mehdawy, B., Mare, L., Giuliani, A., Lorenzini, L., Sivilia, S., et al. (2011). The γ-secretase modulator CHF5074 restores memory and hippocampal synaptic plasticity in plaque-free Tg2576 mice. Journal of Alzheimers Disease, 24(4), 799–816.
Barbizan, R., & Oliveira, A. L. (2010). Impact of acute inflammation on spinal motoneuron synaptic plasticity following ventral root avulsion. Journal of Neuroinflammation, 7, 29.
Bienenstock, E. L., Cooper, L. N., & Munro, P. W. (1982). Theory for the development of neuron selectivity: Orientation specificity and binocular interaction in visual cortex. The Journal of Neuroscience, 2(1), 32–48.
Caramia, M. D., Palmieri, M. G., Desiato, M. T., Boffa, L., Galizia, P., Rossini, P. M., et al. (2004). Brain excitability changes in the relapsing and remitting phases of multiple sclerosis: A study with transcranial magnetic stimulation. Clinical Neurophysiology, 115(4), 956–965.
Centonze, D., Muzio, L., Rossi, S., Cavasinni, F., De Chiara, V., Bergami, A., et al. (2009). Inflammation triggers synaptic alteration and degeneration in experimental autoimmune encephalomyelitis. The Journal of Neuroscience, 29(11), 3442–3452.
Chapman, C. A., Perez, Y., & Lacaille, J. C. (1998). Effects of GABA(A) inhibition on the expression of long-term potentiation in CA1 pyramidal cells are dependent on tetanization parameters. Hippocampus, 8(3), 289–298.
Coogan, A., & O’Connor, J. J. (1997). Inhibition of NMDA receptor-mediated synaptic transmission in the rat dentate gyrus in vitro by IL-1β. Neuroreport, 8(9–10), 2107–2110.
Cose, S. (2007). T-cell migration: A naive paradigm? Immunology, 120(1), 1–7.
Di Filippo, M., Chiasserini, D., Gardoni, F., Viviani, B., Tozzi, A., Giampà, C., et al. (2013). Effects of central and peripheral inflammation on hippocampal synaptic plasticity. Neurobiology of Disease,. doi:10.1016/j.nbd.2012.12.009.
Di Lazzaro, V., Pilato, F., Saturno, E., Oliviero, A., Dileone, M., Mazzone, P., et al. (2005). Theta-burst repetitive transcranial magnetic stimulation suppresses specific excitatory circuits in the human motor cortex. The Journal of Physiology, 565(Pt3), 945–950.
Errico, F., Nisticò, R., Napolitano, F., Oliva, A. B., Romano, R., Barbieri, F., et al. (2011). Persistent increase of D-aspartate in D-aspartate oxidase mutant mice induces a precocious hippocampal age-dependent synaptic plasticity and spatial memory decay. Neurobiology of Aging, 32(11), 2061–2074.
Furlan, R., Bergami, A., Brambilla, E., Butti, E., De Simoni, M. G., Campagnoli, M., et al. (2007). HSV-1-mediated IL-1 receptor antagonist gene therapy ameliorates MOG(35–55)-induced experimental autoimmune encephalomyelitis in C57BL/6 mice. Gene Therapy, 14(1), 93–98.
Gambrill, A. C., Storey, G. P., & Barria, A. (2011). Dynamic regulation of NMDA receptor transmission. Journal of Neurophysiology, 105(1), 162–171.
Gentner, R., Wankerl, K., Reinsberger, C., Zeller, D., & Classen, J. (2008). Depression of human corticospinal excitability induced by magnetic theta-burst stimulation: Evidence of rapid polarity-reversing metaplasticity. Cerebral Cortex, 18(9), 2046–2053.
Grasselli, G., Rossi, S., Musella, A., Gentile, A., Loizzo, S., Muzio, L., et al. (2013). Abnormal NMDA receptor function exacerbates experimental autoimmune encephalomyelitis. Britsh Journal of Pharmacology, 168(2), 502–517.
Grover, L. M., & Yan, C. (1999). Blockade of GABAA receptors facilitates induction of NMDA receptor-independent long-term potentiation. Journal of Neurophysiology, 81(6), 2814–2822.
Haji, N., Mandolesi, G., Gentile, A., Sacchetti, L., Fresegna, D., Rossi, S., et al. (2012). TNF-α-mediated anxiety in a mouse model of multiple sclerosis. Experimental Neurology, 237(2), 296–303.
Herz, J., Paterka, M., Niesner, R. A., Brandt, A. U., Siffrin, V., Leuenberger, T., et al. (2011). In vivo imaging of lymphocytes in the CNS reveals different behaviour of naïve T cells in health and autoimmunity. Journal of Neuroinflammation, 8, 131.
Hess, G., & Donoghue, J. P. (1996). Long-term depression of horizontal connections in rat motor cortex. The European Journal of Neuroscience, 8(4), 658–665.
Huang, Y. Z., Edwards, M. J., Rounis, E., Bhatia, K. P., & Rothwell, J. C. (2005). Theta burst stimulation of the human motor cortex. Neuron, 45(2), 201–206.
Huang, Y. Z., Rothwell, J. C., Chen, R. S., Lu, C. S., & Chuang, W. L. (2011). The theoretical model of theta burst form of repetitive transcranial magnetic stimulation. Clinical Neurophysiology, 122(5), 1011–1018.
Iezzi, E., Conte, A., Suppa, A., Agostino, R., Dinapoli, L., Scontrini, A., et al. (2008). Phasic voluntary movements reverse the aftereffects of subsequent theta-burst stimulation in humans. Journal of Neurophysiology, 100(4), 2070–2076.
Jenkins, T. M., Toosy, A. T., Ciccarelli, O., Miszkiel, K. A., Wheeler-Kingshott, C. A., Henderson, A. P., et al. (2010). Neuroplasticity predicts outcome of optic neuritis independent of tissue damage. Annals of Neurology, 67(1), 99–113.
Kemp, N., McQueen, J., Faulkes, S., & Bashir, Z. I. (2000). Different forms of LTD in the CA1 region of the hippocampus: Role of age and stimulus protocol. The European Journal of Neuroscience, 12(1), 360–366.
Kim do, Y., Hao, J., Liu, R., Turner, G., Shi, F. D., & Rho, J. M. (2012). Inflammation-mediated memory dysfunction and effects of a ketogenic diet in a murine model of multiple sclerosis. PLoS One, 7(5), e35476.
Kirkwood, A., Rioult, M. C., & Bear, M. F. (1996). Experience-dependent modification of synaptic plasticity in visual cortex. Nature, 381(6582), 526–528.
Komaki, A., Shahidi, S., Lashgari, R., Haghparast, A., Malakouti, S. M., & Noorbakhsh, S. M. (2007). Effects of GABAergic inhibition on neocortical long-term potentiation in the chronically prepared rat. Neuroscience Letters, 422, 181–186.
Korn, T., Reddy, J., Gao, W., Betelli, E., Awasthi, A., Petersen, T. R., et al. (2007). Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nature Medicine, 13, 423–431.
Lai, A. Y., Swayze, R. D., El-Husseini, A., & Song, C. (2006). Interleukin-1 beta modulates AMPA receptor expression and phosphorylation in hippocampal neurons. Journal of Neuroimmunology, 175(1–2), 97–106.
Levkovitz, Y., Avignone, E., Groner, Y., & Segal, M. (1999). Upregulation of GABA neurotransmission suppresses hippocampal excitability and prevents long-term potentiation in transgenic superoxide dismutase-overexpressing mice. The Journal of Neuroscience, 19(24), 10977–10984.
Lotan, M., & Schwartz, M. (1994). Cross talk between the immune system and the nervous system in response to injury: Implications for regeneration. The FASEB Journal, 8, 1026–1033.
Lu, Y. M., Mansuym, I. M., Kandel, E. R., & Roder, J. (2000). Calcineurin-mediated LTD of GABAergic inhibition underlies the increased excitability of CA1 neurons associated with LTP. Neuron, 26(1), 197–205.
Lycke, J. N., Karlsson, J. E., Andersen, O., & Rosengren, L. E. (1998). Neurofilament protein in cerebrospinal fluid: A potential marker of activity in multiple sclerosis. Journal of Neurology, Neurosurgery and Psychiatry, 64, 402–404.
Malenka, R. C., & Bear, M. F. (2004). LTP and LTD: An embarrassment of riches. Neuron, 44(1), 5–21.
Mandolesi, G., Grasselli, G., Musella, A., Gentile, A., Musumeci, G., Sepman, H., et al. (2012). GABAergic signaling and connectivity on Purkinje cells are impaired in experimental autoimmune encephalomyelitis. Neurobiology of Disease, 46(2), 414–424.
Mandolesi, G., Grasselli, G., Musumeci, G., & Centonze, D. (2010). Cognitive deficits in experimental autoimmune encephalomyelitis: Neuroinflammation and synaptic degeneration. Neurological Sciences, 31(Suppl 2), S255–S259.
Mandolesi, G., Musella, A., Gentile, A., Grasselli, G., Haji, N., Sepman, H., et al. (2013). Interleukin-1β alters glutamate transmission at Purkinje cell synapses in a mouse model for multiple sclerosis. The Journal of Neuroscience, 33(29), 12105–12121.
Martin, L. J., Zurek, A. A., MacDonald, J. F., Roder, J. C., Jackson, M. F., & Orser, B. A. (2010). Alpha5GABAA receptor activity sets the threshold for long-term potentiation and constrains hippocampus-dependent memory. The Journal of Neuroscience, 30(15), 5269–5282.
Mori, F., Rossi, S., Sancesario, G., Codecà, C., Mataluni, G., Monteleone, F., et al. (2011). Cognitive and cortical plasticity deficits correlate with altered amyloid-β CSF levels in multiple sclerosis. Neuropsychopharmacology, 36(3), 559–568.
Musumeci, G., Grasselli, G., Rossi, S., De Chiara, V., Musella, A., Motta, C., et al. (2011). Transient receptor potential vanilloid 1 channels modulate the synaptic effects of TNF-α and of IL-1β in experimental autoimmune encephalomyelitis. Neurobiology of Disease, 43(3), 669–677.
Nisticò, R., Mango, D., Mandolesi, G., Piccinin, S., Berretta, N., Pignatelli, M., et al. (2013). Inflammation subverts hippocampal synaptic plasticity in experimental multiple sclerosis. PLoS One, 8(1), e54666.
Nisticò, R., Pignatelli, M., Piccinin, S., Mercuri, N. B., & Collingridge, G. (2012). Targeting synaptic dysfunction in Alzheimer’s disease therapy. Molecular Neurobiology, 46(3), 572–587.
Polman, C. H., Reingold, S. C., Banwell, B., Clanet, M., Cohen, J. A., Filippi, M., et al. (2011). Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Annals of Neurology, 69(2), 292–302.
Reddy, H., Narayanan, S., Arnoutelis, R., Jenkinson, M., Antel, J., Matthews, P. M., et al. (2000). Evidence for adaptive functional changes in the cerebral cortex with axonal injury from multiple sclerosis. Brain, 123(11), 2314–2320.
Rocca, M. A., Falini, A., Colombo, B., Scotti, G., Comi, G., & Filippi, M. (2002). Adaptive functional changes in the cerebral cortex of patients with non disabling multiple sclerosis correlate with the extent of brain structural damage. Annals Neurology, 51(3), 330–339.
Rossi, S., De Chiara, V., Furlan, R., Musella, A., Cavasinni, F., Muzio, L., et al. (2010). Abnormal activity of the Na/Ca exchanger enhances glutamate transmission in experimental autoimmune encephalomyelitis. Brain, Behavior, and Immunity, 24(8), 1379–1385.
Rossi, S., Furlan, R., De Chiara, V., Motta, C., Studer, V., Mori, F., et al. (2012a). Interleukin-1β causes synaptic hyperexcitability in multiple sclerosis. Annals of Neurology, 71(1), 76–83.
Rossi, S., Studer, V., Motta, C., De Chiara, V., Barbieri, F., Bernardi, G., et al. (2012b). Inflammation inhibits GABA transmission in multiple sclerosis. Multiple Sclerosis, 18(11), 1633–1635.
Rothwell, J. C., Hallett, M., Berardelli, A., Eisen, A., Rossini, P., & Paulus, W. (1999). Magnetic stimulation: motor evoked potentials. The International Federation of Clinical Neurophysiology. Electroencephalography and Clinical Neurophysiology. Supplement, 52, 97–103.
Schäfers, M., & Sorkin, L. (2008). Effect of cytokines on neuronal excitability. Neuroscience Letters, 437(3):188–193. Erratum in: Neuroscience Letters. 2010 481(3):191.
Schirmer, L., Merkler, D., König, F. B., Brück, W., & Stadelmann, C. (2013). Neuroaxonal regeneration is more pronounced in early multiple sclerosis than in traumatic brain injury lesions. Brain Pathology, 23(1), 2–12.
Siebner, H. R., Lang, N., Rizzo, V., Nitsche, M. A., Paulus, W., Lemon, R. N., et al. (2004). Preconditioning of low-frequency repetitive transcranial magnetic stimulation with transcranial direct current stimulation: Evidence for homeostatic plasticity in the human motor cortex. The Journal of Neuroscience, 24(13), 3379–3385.
Stagg, C. J., Wylezinska, M., Matthews, P. M., Johansen-Berg, H., Jezzard, P., Rothwell, J. C., et al. (2009). Neurochemical effects of theta burst stimulation as assessed by magnetic resonance spectroscopy. Journal of Neurophysiology, 101(6), 2872–2877.
Stäubli, U., Scafidi, J., & Chun, D. (1999). GABAB receptor antagonism: Facilitatory effects on memory parallel those on LTP induced by TBS but not HFS. The Journal of Neuroscience, 19(11), 4609–4615.
Trepel, C., & Racine, R. J. (2000). GABAergic modulation of neocortical long-term potentiation in the freely moving rat. Synapse (New York, N.Y.), 35(2), 120–128.
Viviani, B., Gardoni, F., Bartesaghi, S., Corsini, E., Facchi, A., Galli, C. L., et al. (2006). Interleukin-1 beta released by gp120 drives neural death through tyrosine phosphorylation and trafficking of NMDA receptors. The Journal of Biological Chemistry, 281(40), 30212–30222.
Wigström, H., & Gustafsson, B. (1983). Heterosynaptic modulation of homosynaptic long-lasting potentiation in the hippocampal slice. Acta Physiologica Scandinavica, 119(4), 455–458.
Yoshiike, Y., Kimura, T., Yamashita, S., Furudate, H., Mizoroki, T., Murayama, M., et al. (2008). GABA(A) receptor-mediated acceleration of aging-associated memory decline in APP/PS1 mice and its pharmacological treatment by picrotoxin. PLoS One, 3(8), e3029.
Zeller, D., Dang, S. Y., Weise, D., Rieckmann, P., Toyka, K. V., & Classen, J. (2012). Excitability decreasing central motor plasticity is retained in multiple sclerosis patients. BMC Neurology, 12, 92.
Zhang, D., Hu, X., Qian, L., O’Callaghan, J. P., & Hong, J. S. (2010). Astrogliosis in CNS pathologies: Is there a role for microglia? Molecular Neurobiology, 41(2–3), 232–241.
Ziehn, M. O., Avedisian, A. A., Dervin, S. M., Umeda, E. A., O’Dell, T. J., & Voskuhl, R. R. (2012). Therapeutic testosterone administration preserves excitatory synaptic transmission in the hippocampus during autoimmune demyelinating disease. The Journal of Neuroscience, 32(36), 12312–12324.
Ziehn, M. O., Avedisian, A. A., Tiwari-Woodruff, S., & Voskuhl, R. R. (2010). Hippocampal CA1 atrophy and synaptic loss during experimental autoimmune encephalomyelitis, EAE. Laboratory Investigation, 90(5), 774–786.
Acknowledgments
This investigation was supported by Fondazione Italiana Sclerosi Multipla (Progetto Speciale FISM), by the Italian National Ministry of Health and by the Italian National Ministry of University to DC. We are indebted to Dr. Mauro Federici and Vladimiro Batocchi for excellent technical assistance.
Conflict of interest
Diego Centonze acted as an Advisory Board member of Merck Serono, Teva, Bayer Schering, Biogen Idec, Novartis, and received funding for traveling and honoraria for speaking or consultation fees from Merck Serono, Teva, Novartis, Bayer Schering, Sanofi-aventis, Biogen Idec. He is also the principal investigator in clinical trials for Novartis, Merck Serono, Teva, Bayer Schering, Sanofi-aventis, Biogen Idec, Roche. The other authors have no disclosures to report.
Author information
Authors and Affiliations
Corresponding author
Additional information
Francesco Mori and Robert Nisticò are equally contributing first authors.
Rights and permissions
About this article
Cite this article
Mori, F., Nisticò, R., Mandolesi, G. et al. Interleukin-1β Promotes Long-Term Potentiation in Patients with Multiple Sclerosis. Neuromol Med 16, 38–51 (2014). https://doi.org/10.1007/s12017-013-8249-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-013-8249-7