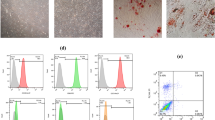
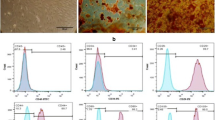
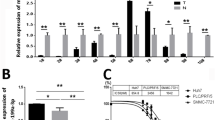
Abstract
Recent investigations have emphasized the role of aberrant expression of microRNAs (miRNAs) in progression of almost all types of cancers. Exosomes, membrane-enclosed natural nanovesicles, transport cellular contents, including proteins, mRNAs, and miRNAs, between cells. Unique features of exosomes make them an appropriate carrier for drug delivery. miRNA-381 is one of the downregulated miRNAs in several cancers including triple-negative breast cancer (TNBC) and restoration of its expression in TNBC cells can restrict their migratory ability through targeting several signaling pathways. In current study, we exploited the exosomes isolated from adipose-derived mesenchymal stem cells (ADMSC-exosomes) to deliver miR-381 mimic to MDA-MB-231 cells to elucidate their effects on TNBC cells. The effects of miR-381 loaded ADMSC-exosomes on proliferation, apoptosis, migration, and invasion of MDA-MB-231 cells were analyzed. Our results indicated that ADMSC-exosomes were successfully isolated and internalized by MDA-MB-231 cells. miR-381 mimic was efficiently delivered to MDA-MB-231 cells by ADMSC-exosomes. miR-381 loaded ADMSC-exosomes significantly downregulated the expression of epithelial to mesenchymal transition (EMT) related genes and proteins. Notably, miR-381 loaded ADMSC-exosomes inhibited proliferation, migration, and invasion capacity of MDA-MB-231 and promoted their apoptosis in vitro. Taken together, we showed that ADMSC-exosomes could be used as efficient nanocarriers for RNA-based therapies.

Graphical abstract






Similar content being viewed by others
References
Adams, B. D., Kasinski, A. L., & Slack, F. J. (2014). Aberrant regulation and function of microRNAs in cancer. Current biology : CB, 24(16), R762–R776. https://doi.org/10.1016/j.cub.2014.06.043.
Peng, Y., & Croce, C. M. (2016). The role of MicroRNAs in human cancer. Signal Transduction and Targeted Therapy, 1(1), 15004. https://doi.org/10.1038/sigtrans.2015.4.
Marote, A., Teixeira, F. G., Mendes-Pinheiro, B., & Salgado, A. J. (2016). MSCs-derived Exosomes: Cell-secreted Nanovesicles with regenerative potential. Frontiers in Pharmacology, 7, 231. https://doi.org/10.3389/fphar.2016.00231.
Thery, C. (2011). Exosomes: Secreted vesicles and intercellular communications. F1000 biology reports, 3, 15. https://doi.org/10.3410/b3-15.
Batrakova, E. V., & Kim, M. S. (2015). Using exosomes, naturally-equipped nanocarriers, for drug delivery. Journal of controlled release : official journal of the Controlled Release Society, 219, 396–405. https://doi.org/10.1016/j.jconrel.2015.07.030.
Greco, S. J., & Rameshwar, P. (2012). Mesenchymal stem cells in drug/gene delivery: Implications for cell therapy. Therapeutic Delivery, 3(8), 997–1004.
Yeo, R. W., Lai, R. C., Zhang, B., Tan, S. S., Yin, Y., Teh, B. J., et al. (2013). Mesenchymal stem cell: An efficient mass producer of exosomes for drug delivery. Advanced Drug Delivery Reviews, 65(3), 336–341. https://doi.org/10.1016/j.addr.2012.07.001.
Melzer, C., Rehn, V., Yang, Y., Bahre, H., von der Ohe, J., & Hass, R. (2019). Taxol-loaded MSC-derived Exosomes provide a therapeutic vehicle to target metastatic breast Cancer and other carcinoma cells. Cancers, 11(6). https://doi.org/10.3390/cancers11060798.
Wu, S., Ju, G. Q., Du, T., Zhu, Y. J., & Liu, G. H. (2013). Microvesicles derived from human umbilical cord Wharton's jelly mesenchymal stem cells attenuate bladder tumor cell growth in vitro and in vivo. PLoS One, 8(4), e61366. https://doi.org/10.1371/journal.pone.0061366.
Bu, S., Wang, Q., Zhang, Q., Sun, J., He, B., Xiang, C., Liu, Z., & Lai, D. (2016). Human endometrial mesenchymal stem cells exhibit intrinsic anti-tumor properties on human epithelial ovarian cancer cells. Scientific Reports, 6, 37019. https://doi.org/10.1038/srep37019.
Reza, A., Choi, Y. J., Yasuda, H., & Kim, J. H. (2016). Human adipose mesenchymal stem cell-derived exosomal-miRNAs are critical factors for inducing anti-proliferation signalling to A2780 and SKOV-3 ovarian cancer cells. Scientific Reports, 6, 38498. https://doi.org/10.1038/srep38498.
Liang, Y., Zhao, Q., Fan, L., Zhang, Z., Tan, B., Liu, Y., et al. (2015). Down-regulation of MicroRNA-381 promotes cell proliferation and invasion in colon cancer through up-regulation of LRH-1. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 75, 137–141. https://doi.org/10.1016/j.biopha.2015.07.020.
Cao, Q., Liu, F., Ji, K., Liu, N., He, Y., Zhang, W., & Wang, L. (2017). MicroRNA-381 inhibits the metastasis of gastric cancer by targeting TMEM16A expression. Journal of experimental & clinical cancer research : CR, 36(1), 29. https://doi.org/10.1186/s13046-017-0499-z.
Xue, Y., Xu, W., Zhao, W., Wang, W., Zhang, D., & Wu, P. (2017). miR-381 inhibited breast cancer cells proliferation, epithelial-to-mesenchymal transition and metastasis by targeting CXCR4. Biomedicine & pharmacotherapy = Biomedecine & pharmacotherapie, 86, 426–433. https://doi.org/10.1016/j.biopha.2016.12.051.
Rui, X., Gu, T. T., Pan, H. F., Shao, S. L., & Shao, H. X. (2019). MicroRNA-381 suppresses proliferation and invasion of prostate cancer cells through downregulation of the androgen receptor. Oncology Letters, 18(2), 2066–2072. https://doi.org/10.3892/ol.2019.10471.
Mohammadi-Yeganeh, S., Hosseini, V., & Paryan, M. (2019). Wnt pathway targeting reduces triple-negative breast cancer aggressiveness through miRNA regulation in vitro and in vivo. Journal of Cellular Physiology, 234(10), 18317–18328. https://doi.org/10.1002/jcp.28465.
Mi, H., Wang, X., Wang, F., Li, L., Zhu, M., Wang, N., Xiong, Y., & Gu, Y. (2018). miR-381 induces sensitivity of breast cancer cells to doxorubicin by inactivation of MAPK signaling via FYN. European Journal of Pharmacology, 839, 66–75. https://doi.org/10.1016/j.ejphar.2018.09.024.
Yi, D., Xu, L., Wang, R., Lu, X., & Sang, J. (2019). miR-381 overcomes cisplatin resistance in breast cancer by targeting MDR1. Cell Biology International, 43(1), 12–21. https://doi.org/10.1002/cbin.11071.
Schlimgen, R., Howard, J., Wooley, D., Thompson, M., Baden, L. R., Yang, O. O., Christiani, D. C., Mostoslavsky, G., Diamond, D. V., Duane, E. G., Byers, K., Winters, T., Gelfand, J. A., Fujimoto, G., Hudson, T. W., & Vyas, J. M. (2016). Risks associated with Lentiviral vector exposures and prevention strategies. Journal of Occupational and Environmental Medicine, 58(12), 1159–1166. https://doi.org/10.1097/jom.0000000000000879.
Kia, V., Paryan, M., Mortazavi, Y., Biglari, A., & Mohammadi-Yeganeh, S. (2019). Evaluation of exosomal miR-9 and miR-155 targeting PTEN and DUSP14 in highly metastatic breast cancer and their effect on low metastatic cells. Journal of Cellular Biochemistry, 120(4), 5666–5676. https://doi.org/10.1002/jcb.27850.
Kong, W., Yang, L., Li, P. P., Kong, Q. Q., Wang, H. Y., Han, G. X., et al. (2018). MiR-381-3p inhibits proliferation, migration and invasion by targeting LRP6 in papillary thyroid carcinoma. European review for medical and pharmacological sciences, 22(12), 3804–3811. https://doi.org/10.26355/eurrev_201806_15264.
Hu, W. W., Chen, P. C., Chen, J. M., Wu, Y. M., Liu, P. Y., Lu, C. H., et al. (2017). Periostin promotes epithelial-mesenchymal transition via the MAPK/miR-381 axis in lung cancer. Oncotarget, 8(37), 62248–62260. https://doi.org/10.18632/oncotarget.19273.
Bader, A. G., Brown, D., & Winkler, M. (2010). The promise of microRNA replacement therapy. Cancer Research, 70(18), 7027–7030. https://doi.org/10.1158/0008-5472.can-10-2010.
Zhang, D., Lee, H., Zhu, Z., Minhas, J. K., & Jin, Y. (2017). Enrichment of selective miRNAs in exosomes and delivery of exosomal miRNAs in vitro and in vivo. American journal of physiology Lung cellular and molecular physiology, 312(1), L110–Ll21. https://doi.org/10.1152/ajplung.00423.2016.
Shang, S., Wang, J., Chen, S., Tian, R., Zeng, H., Wang, L., Xia, M., Zhu, H., & Zuo, C. (2019). Exosomal miRNA-1231 derived from bone marrow mesenchymal stem cells inhibits the activity of pancreatic cancer. Cancer medicine., 8, 7728–7740. https://doi.org/10.1002/cam4.2633.
Che, Y., Shi, X., Shi, Y., Jiang, X., Ai, Q., Shi, Y., Gong, F., & Jiang, W. (2019). Exosomes derived from miR-143-overexpressing MSCs inhibit cell migration and invasion in human prostate Cancer by Downregulating TFF3. Molecular therapy Nucleic acids, 18, 232–244. https://doi.org/10.1016/j.omtn.2019.08.010.
Xie, C., Du, L. Y., Guo, F., Li, X., & Cheng, B. (2019). Exosomes derived from microRNA-101-3p-overexpressing human bone marrow mesenchymal stem cells suppress oral cancer cell proliferation, invasion, and migration. Molecular and Cellular Biochemistry, 458(1–2), 11–26. https://doi.org/10.1007/s11010-019-03526-7.
Sharif, S., Ghahremani, M. H., & Soleimani, M. (2018). Delivery of exogenous miR-124 to Glioblastoma multiform cells by Wharton's jelly Mesenchymal stem cells decreases cell proliferation and migration, and confers Chemosensitivity. Stem Cell Reviews and Reports, 14(2), 236–246. https://doi.org/10.1007/s12015-017-9788-3.
Naseri, Z., Oskuee, R. K., Jaafari, M. R., & Forouzandeh, M. M. (2018). Exosome-mediated delivery of functionally active miRNA-142-3p inhibitor reduces tumorigenicity of breast cancer in vitro and in vivo. International Journal of Nanomedicine, 13, 7727–7747. https://doi.org/10.2147/ijn.s182384.
Naseri, Z., Oskuee, R. K., Forouzandeh-Moghadam, M., & Jaafari, M. R. (2020). Delivery of LNA-antimiR-142-3p by Mesenchymal stem cells-derived Exosomes to breast Cancer stem cells reduces Tumorigenicity. Stem cell reviews and reports., 16, 541–556. https://doi.org/10.1007/s12015-019-09944-w.
Lou, G., Song, X., Yang, F., Wu, S., Wang, J., Chen, Z., & Liu, Y. (2015). Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. Journal of Hematology & Oncology, 8, 122. https://doi.org/10.1186/s13045-015-0220-7.
Lou, G., Chen, L., Xia, C., Wang, W., Qi, J., Li, A., Zhao, L., Chen, Z., Zheng, M., & Liu, Y. (2020). MiR-199a-modified exosomes from adipose tissue-derived mesenchymal stem cells improve hepatocellular carcinoma chemosensitivity through mTOR pathway. Journal of experimental & clinical cancer research : CR, 39(1), 4. https://doi.org/10.1186/s13046-019-1512-5.
Strioga, M., Viswanathan, S., Darinskas, A., Slaby, O., & Michalek, J. (2012). Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells and Development, 21(14), 2724–2752. https://doi.org/10.1089/scd.2011.0722.
Lotvall, J., Hill, A. F., Hochberg, F., Buzas, E. I., Di Vizio, D., Gardiner, C., et al. (2014). Minimal experimental requirements for definition of extracellular vesicles and their functions: A position statement from the International Society for Extracellular Vesicles. Journal of extracellular vesicles, 3, 26913. https://doi.org/10.3402/jev.v3.26913.
Yuana, Y., Böing, A. N., Grootemaat, A. E., van der Pol, E., Hau, C. M., Cizmar, P., Buhr, E., Sturk, A., & Nieuwland, R. (2015). Handling and storage of human body fluids for analysis of extracellular vesicles. Journal of extracellular vesicles, 4, 29260. https://doi.org/10.3402/jev.v4.29260.
Asadirad, A., Hashemi, S. M., Baghaei, K., Ghanbarian, H., Mortaz, E., Zali, M. R., & Amani, D. (2019). Phenotypical and functional evaluation of dendritic cells after exosomal delivery of miRNA-155. Life Sciences, 219, 152–162. https://doi.org/10.1016/j.lfs.2019.01.005.
Dey, N., Barwick, B. G., Moreno, C. S., Ordanic-Kodani, M., Chen, Z., Oprea-Ilies, G., Tang, W., Catzavelos, C., Kerstann, K. F., Sledge Jr., G. W., Abramovitz, M., Bouzyk, M., de, P., & Leyland-Jones, B. R. (2013). Wnt signaling in triple negative breast cancer is associated with metastasis. BMC Cancer, 13, 537. https://doi.org/10.1186/1471-2407-13-537.
Xu, J., Prosperi, J. R., Choudhury, N., Olopade, O. I., & Goss, K. H. (2015). Beta-catenin is required for the tumorigenic behavior of triple-negative breast cancer cells. PloS one, 10(2), e0117097. https://doi.org/10.1371/journal.pone.0117097.
Zhan, T., Rindtorff, N., & Boutros, M. (2017). Wnt signaling in cancer. Oncogene, 36(11), 1461–1473. https://doi.org/10.1038/onc.2016.304.
Ma, J., Lu, W., Chen, D., Xu, B., & Li, Y. (2017). Role of Wnt co-receptor LRP6 in triple negative breast Cancer cell migration and invasion. Journal of Cellular Biochemistry, 118(9), 2968–2976. https://doi.org/10.1002/jcb.25956.
He, X., Wei, Y., Wang, Y., Liu, L., Wang, W., & Li, N. (2016). MiR-381 functions as a tumor suppressor in colorectal cancer by targeting Twist1. Oncotargets and Therapy, 9, 1231–1239. https://doi.org/10.2147/ott.s99228.
Kashiwagi, S., Yashiro, M., Takashima, T., Nomura, S., Noda, S., Kawajiri, H., Ishikawa, T., Wakasa, K., & Hirakawa, K. (2010). Significance of E-cadherin expression in triple-negative breast cancer. British Journal of Cancer, 103(2), 249–255. https://doi.org/10.1038/sj.bjc.6605735.
Lamouille, S., Xu, J., & Derynck, R. (2014). Molecular mechanisms of epithelial-mesenchymal transition. Nature Reviews Molecular Cell Biology, 15(3), 178–196. https://doi.org/10.1038/nrm3758.
Wang, Y., Liu, J., Ying, X., Lin, P. C., & Zhou, B. P. (2016). Twist-mediated epithelial-mesenchymal transition promotes breast tumor cell invasion via inhibition of hippo pathway. Scientific Reports, 6, 24606. https://doi.org/10.1038/srep24606.
Zhang, Y. Q., Wei, X. L., Liang, Y. K., Chen, W. L., Zhang, F., Bai, J. W., Qiu, S. Q., du, C. W., Huang, W. H., & Zhang, G. J. (2015). Over-expressed twist associates with markers of epithelial Mesenchymal transition and predicts poor prognosis in breast cancers via ERK and Akt activation. PLoS One, 10(8), e0135851. https://doi.org/10.1371/journal.pone.0135851.
Xiao, B., Shi, X., & Bai, J. (2019). miR-30a regulates the proliferation and invasion of breast cancer cells by targeting snail. Oncology Letters, 17(1), 406–413. https://doi.org/10.3892/ol.2018.9552.
Lin, R., Wang, S., & Zhao, R. C. (2013). Exosomes from human adipose-derived mesenchymal stem cells promote migration through Wnt signaling pathway in a breast cancer cell model. Molecular and Cellular Biochemistry, 383(1–2), 13–20. https://doi.org/10.1007/s11010-013-1746-z.
Wegmeyer, H., Broske, A. M., Leddin, M., Kuentzer, K., Nisslbeck, A. K., Hupfeld, J., et al. (2013). Mesenchymal stromal cell characteristics vary depending on their origin. Stem Cells and Development, 22(19), 2606–2618. https://doi.org/10.1089/scd.2013.0016.
Acknowledgements
The authors should thank Cellular and Molecular Biology Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran for providing technical support.
Funding
This project funded by School of Advanced Technologies in Medicine, Shahid Beheshti University of Medical Sciences,Tehran, Iran (Contact grant No: 11985).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was executed under supervision of ethical committee of shahid Beheshti University of Medical Sciences, Tehran, Iran (ethical code: IR.SBMU.REC.1397.004).
Conflict of Interest
Authors declare there are no competing conflict of interests in relation to the work described.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shojaei, S., Hashemi, S.M., Ghanbarian, H. et al. Delivery of miR-381-3p Mimic by Mesenchymal Stem Cell-Derived Exosomes Inhibits Triple Negative Breast Cancer Aggressiveness; an In Vitro Study. Stem Cell Rev and Rep 17, 1027–1038 (2021). https://doi.org/10.1007/s12015-020-10089-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-020-10089-4