Abstract
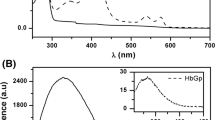
A comparative denaturation of HbA and HbS in the R states using sodium n-dodecyl sulfate (SDS) was carried out at pH 7.20 in the presence and absence of Calcium (0–40 μM) and monitored by UV–Vis spectrophotometry in the range of 250–650 nm. In the HbS spectra, the calcium alone caused little or no perturbation of the aromatic region but caused a decrease in oxygen affinity when compared to the HbA. The combinations of [SDS] and [Ca] perturbed the HbS the most, relative to the individual spectra of the [SDS] and [Ca]. However, the presence of Ca appeared to diminish the adverse effects of the SDS on HbA. The denaturation pathway of the HbA involved mainly the formation of heme dimers and some ferryl heme species. For the HbS, heme monomers and a large amount of ferryl species were formed. It is suggested that the greater monomer species formed by the HbS denaturation pathway would result in both Fenton and enhanced enzymatic reactions, compared to the dimer. This could lead ultimately to the formation of ferryl radicals. Thus, at physiological pH for the HbS, the Ca–SDS interaction increases the tendency for protein denaturation in comparison to the HbA.




Similar content being viewed by others
References
Lew, V. L., Daw, N., Perdomo, D., Etzion, Z., Bookchin, R. M., & Tiffert, T. (2003). Distribution of plasma membrane Ca2+ pump activity in normal human red blood cells. Blood, 102, 4206–4213.
Lew, V. L., Tiffert, T., & Ginsburg, H. (2003). Excess hemoglobin digestion and the osmotic stability of Plasmodium falciparum-infected red blood cells. Blood, 101, 4189–4194.
Miller, L. H., Good, M. F., & Milon, G. (1994). Malaria pathogenesis. Science, 264, 1878–1883.
McGilvray, I. D., Serghides, L., Kapus, A., Rotstein, O. D., & Kain, K. C. (2000). Nonopsonic monocyte/macrophage phagocytosis of Plasmodium falciparum-parasitized erythrocytes: a role for CD36 in malarial clearance. Blood, 96, 3231–3240.
Lopez-Estraño, C., Bhattacharjee, S., Harrison, T., & Haldar, K. (2003). Cooperative domains define a unique host cell-targeting signal in Plasmodium falciparum-infected erythrocytes. Proceedings of the National Academy of Sciences of the United States of America, 100, 12402–12407.
Banerjee, R., Liu, J., Beatty, W., Pelosof, L., Klemba, M., & Goldberg, D. E. (2002). Four plasmepsins are active in the Plasmodium falciparum food vacuole, including a protease with an active-site histidine. Proceedings of the National Academy of Sciences of the United States of America, 99, 990–995.
Hanspal, M., Dua, M., Takakuwa, Y., Chishti, A. H., & Mizuno, A. (2002). Plasmodium falciparum cysteine protease falcipain-2 cleaves erythrocyte membrane skeletal proteins at late stages of parasite development. Blood, 100, 1048–1054.
Nielsen, K. M., Kasper, J., Choi, M., Bedford, T., Kristiansen, K., Wirth, D. F., et al. (2003). Gene conversion as a source of nucleotide diversity in Plasmodium falciparum. Molecular Biology of Evolution, 20, 726–734.
Kadota, K., Ishino, T., Matsuyama, T., Chinzei, Y., & Yuda, M. (2004). Essential role of membrane-attack protein in malarial transmission to mosquito host. Proceedings of the National Academy of Sciences of the United States of America, 101, 16310–16315.
Allison, A. C. (2002). The discovery of resistance to malaria of sickle-cell heterozygotes. In mini-series: significant contributions to biological chemistry over the past 125 years. Biochemistry and Molecular Biology of Education, 30, 279–287.
Chotivanich, K., Udomsangpetch, R., Pattanapanyasat, K., Chierakul, W., Simpson, J., Looareesuwan, S., et al. (2002). Hemoglobin E: a balanced polymorphism protective against high parasitemias and thus severe P. falciparum malaria. Blood, 100, 1172–1176.
Kelemen, C., Chien, S., & Artmann, G. M. (2001). Temperature transition of human hemoglobin at body temperature: effects of calcium. Biophysical Journal, 80, 2622–2630.
Wasserman, M., Alarcón, C., & Mendoza, P. M. (1982). Effects of Ca2+ depletion on the asexual cell cycle of Plasmodium falciparum. American Journal of Tropical Medicine and Hygiene, 31, 711–717.
Tanabe, K. (1990). Ion metabolism in malaria infected erythrocytes. Blood Cells, 16, 437–449.
Hubber, S. M., Uhlemann, A.-C., Gamper, N. L., Duranton, C., Kremsner, P. G., & Lang, F. (2002). Plasmodium falciparum activates endogenous Cl− channels of human erythrocytes by membrane oxidation. European Molecular Biology Organization Journal, 21, 22–30.
Gazarini, M. L., Thomas, A. P., Pozzan, T., & Garcia, C. R. S. (2003). Calcium signalling in a low calcium environment: how the intracellular malaria parasite solves the problem. Journal of Cell Biology, 161, 103–110.
Ward, G. E., Miller, L. H., & Dvorak, J. A. (1993). The origin of parasitophorous vacuole membrane lipids in malaria-infected erythrocytes. Journal of Cell Science, 106, 237–248.
Dluzewski, A. R., Zicha, D., Dunn, G. A., & Gratzer, W. B. (1995). Origins of the parasitophorous vacuole membrane of the malaria parasite: surface area of the parasitized red cell. European Journal of Cell Biology, 68, 446–449.
Murphy, S. C., Samuel, B. U., Harrison, T., Speicher, K. D., Speicher, D. W., Reid, M. E., et al. (2004). Erythrocyte detergent-resistant membrane proteins: their characterization and selective uptake during malarial infection. Blood, 103, 1920–1928.
Sein, K. K., & Aikawa, M. (1998). The prime role of plasma membrane cholesterol in the pathogenesis of immune evasion and clinical manifestations of falciparum malaria. Medical Hypotheses, 51, 105–110.
Samuel, B. U., Mohandas, N., Harrison, T., McManus, H., Rosse, W., Reid, M., et al. (2001). The role of cholesterol and glycosylphosphatidylinositol-anchored proteins of erythrocyte rafts in regulating raft protein content and malarial infection. Journal of Biological Chemistry, 276, 29319–29329.
Tekwani, B. L., & Walker, L. A. (2005). Targeting the hemozoin synthesis pathway for new antimalarial drug discovery: technologies for in vitro β-hematin formation assay. Combinatorial Chemistry and High Throughput Screening, 8, 63–79.
Aprelev, A., Rotter, M. A., Etzion, Z., Bookchin, R. M., Briehl, R. W., & Ferrone, F. A. (2005). The effects of erythrocyte membranes on the nucleation of sickle hemoglobin. Biophysical Journal, 88, 2815–2822.
Orjih, A. U. (2001). On the mechanism of hemozoin production in malaria parasites: activated erythrocyte membranes promote β-hematin synthesis. Experimental Biology and Medicine, 226, 746–752.
Decker, H., Ryan, M., Jaenicke, E., & Terwilliger, N. (2001). SDS-induced phenoloxidase activity of hemocyanins from Limulus polyphemus, Eurypelma californicum, and Cancer magister. Journal of Biological Chemistry, 276, 17796–17799.
Tanaka, A., & Hoshino, E. (2003). Similarities between the thermal inactivation kinetics of Bacillus amyloliquefaciens α-amylase in an aqueous solution of sodium dodecyl sulphate and the kinetics in the solution of anionic phospholipid vesicles. Biotechnology and Applied Biochemistry, 38, 175–181.
Moosavi-Movahedi, A. A., Nazari, K., & Saboury, A. O. A. (1997). Thermodynamics of denaturation of horseradish peroxidase with sodium n-dodecyl sulphate and n-dodecyl trimethylammonium bromide. Colloids and Surfaces B: Biointerfaces, 9, 123–130.
Ajloo, D., Moosavi-Movahedi, A. A., Sadeghi, M., & Gharibi, H. (2002). Comparative, structural and functional studies of avian and mammalian haemoglobins. Acta Biochimica Polonica, 49, 459–470.
Bordbar, A. K., Moosavi-Movahedi, A. A., & Amini, M. K. (2003). A microcalorimetry and binding study on interaction of dodecyl trimethylammonium bromide with wigeon haemoglobin. Thermochimica Acta, 400, 95–100.
William, R. C., Jr., & Tsay, K. Y. (1973). A convenient chromatographic method for the preparation of human hemoglobin. Analytical Biochemistry, 54, 137–145.
Riggs, A. (1981). Preparation of blood hemoglobin of vertebrates. Methods in Enzymology, 76, 5–29.
La Brake, C. C., & Fung, L. W.-M. (1998). Sickle hemoglobin is more fusogenic than normal hemoglobin at physiological pH and ionic strength conditions. Biochemica et Biophysica Acta, 1406, 152–161.
Antonini, E., & Brunori, M. (1971). The derivatives of ferrous hemoglobin and myoglobin. In A. Neuberger & E. L. Tatum (Eds.), Hemoglobin and myoglobin in their reactions with ligands, vol 21 (pp. 13–39). Amsterdam: North-Holland Publishing Co.
Coletta, M., Ascenzi, P., Santucci, R., Bertollini, A., & Amconi, G. (1993). Interaction of inositol hexakisphosphate with liganded ferrous human hemoglobin: direct evidence for two functionally operative binding sites. Biochemica et Biophysica Acta, 1162, 309–314.
Winterbourn, C. C. (1990). Oxidative reactions of hemoglobins. Methods in Enzymology, 186, 265–272.
Heitz, F., Van, & Mau, N. (2002). Protein structural changes induced by their uptake at interfaces. Biochemica et Biophysica Acta, 1597, 1–11.
Polet, H., & Steinhardt, J. (1969). Sequential stages in the acid denaturation of horse and human ferrihemoglobins. Biochemistry, 8, 857–864.
Jarolim, P., Lahav, M., Liu, S.-C., & Palek, J. (1990). Effect of hemoglobin oxidation products on the stability of red cell membrane skeletons and the associations of skeletal proteins: correlation with a release of hemin. Blood, 76, 2125–2131.
Egawa, T., Hishiki, T., Ichikawa, Y., Kanamori, Y., Shimada, H., Takahashi, S., et al. (2004). Refolding processes of cytochrome P450cam from ferric and ferrous acid forms to the native conformation: formations of folding intermediates with non-native heme coordination state. Journal of Biological Chemistry, 279, 32008–32017.
Asakura, T., Minakata, K., Adachi, K., Russell, M. O., & Schwartz, E. (1977). Denatured hemoglobin in sickle erythrocytes. Journal of Clinical Investigation, 59, 633–640.
Rifkind, J. M., Abugo, O., Levy, A., & Heim, J. (1994). Detection, formation, and relevance of hemichromes and hemochromes. Methods in Enzymology, 231, 449–480.
Asakura, T., Ohnishi, T., Friedman, S., & Schwartz, E. (1974). Abnormal precipitation of oxyhemoglobin S by mechanical shaking. Proceedings of the National Academy of Sciences of the United States of America, 71, 1594–1598.
Giulivi, C., & Davies, K. J. A. (1990). A novel antioxidant role of hemoglobin: the comproportionation of ferrylhemoglobin with oxyhemoglobin. Journal of Biological Chemistry, 265, 19453–19460.
Tsuruga, M., & Shikama, K. (1997). Biphasic nature in the autoxidation reaction of human oxyhemoglobin. Biochemica et Biophysica Acta, 1337, 96–104.
Tsuruga, M., Matsuoka, A., Hachimori, A., Sugawara, Y., & Shikama, K. (1998). The molecular mechanism of autoxidation for human oxyhemoglobin: tilting of the distal histidine causes nonequivalent oxidation in the β chain. Journal of Biological Chemistry, 273, 8607–8615.
Macdonald, V. (1994). Measuring relative rates of hemoglobin oxidation and denaturation. Methods in Enzymology, 231, 480–490.
Tofani, L., Feis, A., Snoke, R. E., Berti, D., Baglioni, P., & Smulevich, G. (2004). Spectroscopic and interfacial properties of myoglobin/surfactant complexes. Biophysical Journal, 87, 1186–1195.
Yusa, K., & Shikama, K. (1987). Oxidation of oxymyoglobin to metmyoglobin with hydrogen peroxide: involvement of ferryl intermediate. Biochemistry, 26, 6684–6688.
Exner, M., & Herold, S. (2000). Kinetic and mechanistic studies of the peroxynitrite-mediated oxidation of oxymyoglobin and oxyhemoglobin. Chemical Research and Toxicology, 12, 287–293.
Lardinois, O. M., Ortiz, & de Montellano, P. R. (2003). Intra- and intermolecular transfers of protein radicals in the reactions of sperm whale myoglobin and hydrogen peroxide. Journal of Biological Chemistry, 278, 36214–36226.
Everse, J., Johnson, M. C., & Marini, M. A. (1994). Peroxidative activities of hemoglobin and hemoglobin derivatives. Methods in Enzymology, 231, 547–561.
Fitch, C. D., & Kanjananggulpan, P. (1987). The state of ferriprotoporphyrin IX in malaria pigment. Journal of Biological Chemistry, 262, 15552–15555.
Pasternack, R. F., Huber, P. R., Boyd, P., Engasser, G., Francesconi, L., Gibbs, E., et al. (1972). On the aggregation of meso-substituted water-soluble porphyrins. Journal of American Chemical Society, 94, 4511–4517.
Das, R. R., Pasternack, R. F., & Plane, R. A. (1970). Fast reaction kinetics of porphyrin dimerization in aqueous solution. Journal of American Chemical Society, 92, 3312–3316.
Brown, S. B., Shillcock, M., & Jones, P. (1976). Equilibrium and kinetic studies of the aggregation of porphyrins in aqueous solution. Biochemical Journal, 153, 279–285.
Brown, S. B., Dean, T. C., & Jones, P. (1970). Catalatic activity of iron (III)-centred catalysts: role of dimerization in the catalytic action of ferrihaems. Biochemical Journal, 117, 741–744.
Jones, P., Robson, T., & Brown, S. B. (1973). The catalase activity of ferrihaems. Biochemical Journal, 135, 353–359.
Jones, P., Prudhoe, K., & Robson, T. (1973). Oxidation of deuteroferrihaem by hydrogen peroxide. Biochemical Journal, 135, 361–365.
Brown, N. A., King, R. F. G. J., Shillcock, M. E., & Brown, S. B. (1974). Haemoglobin catabolism: the role of ferrihaems in studies of the degradation pathway. Biochemical Journal, 137, 135–137.
Poillon, W. N., & Kim, B. C. (1990). 2,3-Diphosphoglycerate and intracellular pH as interdependent determinants of the physiologic solubility of deoxyhemoglobin S. Blood, 76, 1028–1036.
Acknowledgment
We thank Prof. I. Francis Cheng of the Department of Chemistry, the University of Idaho—Moscow, Idaho-USA for providing some of the helpful materials used in the discussion.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chilaka, F.C., Nwamba, C.O. & Moosavi-Movahedi, A.A. Cation Modulation of Hemoglobin Interaction with Sodium n-Dodecyl Sulfate (SDS). I: Calcium Modulation at pH 7.20. Cell Biochem Biophys 60, 187–197 (2011). https://doi.org/10.1007/s12013-010-9139-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-010-9139-3