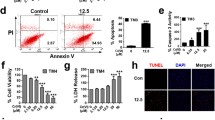
Abstract
Titanium dioxide nanoparticles (TiO2 NPs) can result in the reduction of sperm numbers, but the mechanisms have not been well elucidated. The purpose of this study was to investigate the effects of TiO2 NPs on cell cycle and apoptosis in spermatogonia and to explore the role of PI3K/AKT/mTOR signaling pathway in this process. The mouse spermatogonia cell line (GC-1) was treated with TiO2 NPs at different concentrations (0, 25, 50, 75 and 100 μg/mL) for 24 h to detect cell viability, cell cycle, apoptosis, and key proteins related to cell cycle and PI3K/AKT/mTOR signaling pathway. The agonist (IGF-1) and inhibitor (LY294002) of PI3K were used to verify the role of PI3K/AKT/mTOR signaling pathway in cell cycle and apoptosis. TiO2 NPs significantly inhibited cell proliferation, induced cell cycle arrest at G0/G1 phase and resulted in apoptosis. TiO2 NPs downregulated the levels of cyclin-dependent kinases (CDKs) and cyclins, including CDK4, CDK2, Cyclin D1 and Cyclin E1, while upregulated the levels of p21 and p53 proteins. Furthermore, TiO2 NPs inhibited the PI3K/AKT/mTOR signaling pathway by decreasing the levels of p-PI3K, p-AKT and p-mTOR. IGF-1 reversed the G0/G1 phase arrest and apoptosis caused by TiO2 NPs. However, LY294002 aggravated the G0/G1 phase arrest and apoptosis resulting from TiO2 NPs. Collectively, TiO2 NPs induced cell cycle arrest at G0/G1 phase and apoptosis through inhibiting the activation of PI3K/AKT/mTOR pathway, which could be the main reason for the reduction in sperm numbers caused by TiO2 NPs.







Similar content being viewed by others

Data Availability
The datasets supporting the conclusions of this article are included within the article and its supplementary material, and the original data of this study will be made available upon reasonable request.
References
Najahi-Missaoui W, Arnold RD, Cummings BS (2020) Safe nanoparticles: are we there yet? Int J Mol Sci 22(1):385–405. https://doi.org/10.3390/ijms22010385
Bevacqua E, Occhiuzzi MA, Grande F, Tucci P (2022) TiO2-NPs toxicity and safety: an update of the findings published over the last six years. Mini Rev Med Chem. https://doi.org/10.2174/1389557522666220929152403
Tsuji JS, Maynard AD, Howard PC et al (2006) Research strategies for safety evaluation of nanomaterials, part IV: risk assessment of nanoparticles. Toxicol Sci 89:42–50. https://doi.org/10.1093/toxsci/kfi339
Vance ME, Kuiken T, Vejerano EP et al (2015) Nanotechnology in the real world: Redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6:1769–1780. https://doi.org/10.3762/bjnano.6.181
Dhupal M, Oh JM, Tripathy DR, Kim SK, Koh SB, Park KS (2018) Immunotoxicity of titanium dioxide nanoparticles via simultaneous induction of apoptosis and multiple toll-like receptors signaling through ROS-dependent SAPK/JNK and p38 MAPK activation. Int J Nanomedicine 13:6735–6750. https://doi.org/10.2147/ijn.S176087
Kim H, Jeon D, Oh S et al (2019) Titanium dioxide nanoparticles induce apoptosis by interfering with EGFR signaling in human breast cancer cells. Environ Res 175:117–123. https://doi.org/10.1016/j.envres.2019.05.001
Lee J, Jeong JS, Kim SY et al (2019) Titanium dioxide nanoparticles oral exposure to pregnant rats and its distribution. Part Fibre Toxicol 16:31. https://doi.org/10.1186/s12989-019-0313-5
Racovita AD (2022) Titanium Dioxide: Structure, Impact, and Toxicity. Int J Environ Res Public Health 19:5681. https://doi.org/10.3390/ijerph19095681
García-Rodríguez A, Vila L, Cortés C, Hernández A, Marcos R (2018) Effects of differently shaped TiO(2)NPs (nanospheres, nanorods and nanowires) on the in vitro model (Caco-2/HT29) of the intestinal barrier. Part Fibre Toxicol 15:33. https://doi.org/10.1186/s12989-018-0269-x
Baranowska-Wójcik E, Szwajgier D, Winiarska-Mieczan A (2022) A review of research on the impact of E171/TiO(2) NPs on the digestive tract. J Trace Elem Med Biol 72:126988. https://doi.org/10.1016/j.jtemb.2022.126988
Vaccari F, Zhang L, Giuberti G et al (2023) The impact of metallic nanoparticles on gut fermentation processes: An integrated metabolomics and metagenomics approach following an in vitro digestion and fecal fermentation model. J Hazard Mater 453:131331. https://doi.org/10.1016/j.jhazmat.2023.131331
Virtanen HE, Jørgensen N, Toppari J (2017) Semen quality in the 21(st) century. Nat Rev Urol 14:120–130. https://doi.org/10.1038/nrurol.2016.261
Hong F, Si W, Zhao X et al (2015) TiO2 Nanoparticle Exposure Decreases Spermatogenesis via Biochemical Dysfunctions in the Testis of Male Mice. J Agric Food Chem 63:7084–7092. https://doi.org/10.1021/acs.jafc.5b02652
Hong F, Zhou Y (2020) Spermatogenic Apoptosis and the Involvement of the Nrf2 Pathway in Male Mice Following Exposure to Nano Titanium Dioxide. J Biomed Nanotechnol 16:373–381. https://doi.org/10.1166/jbn.2020.2895
Deng Y, Meng X, Ling C et al (2022) Nanosized Titanium Dioxide Induced Apoptosis and Abnormal Expression of Blood-Testis Barrier Junction Proteins Through JNK Signaling Pathway in TM4 Cells. Biol Trace Elem Res 200:5172–5187. https://doi.org/10.1007/s12011-022-03099-5
Mao Z, Yao M, Xu B et al (2017) Cytoskeletons of Two Reproductive Germ Cell Lines Response Differently to Titanium Dioxide Nanoparticles Mediating Vary Reproductive Toxicity. J Biomed Nanotechnol 13:409–416. https://doi.org/10.1166/jbn.2017.2360
Santonastaso M, Mottola F, Colacurci N et al (2019) In vitro genotoxic effects of titanium dioxide nanoparticles (n-TiO(2) ) in human sperm cells. Mol Reprod Dev 86:1369–1377. https://doi.org/10.1002/mrd.23134
Du X, Liu H, Tian Z et al (2023) PI3K/AKT/mTOR pathway mediated-cell cycle dysregulation contribute to malignant proliferation of mouse spermatogonia induced by microcystin-leucine arginine. Environ Toxicol 38:343–358. https://doi.org/10.1002/tox.23691
Liu H, Deng H, Jian Z et al (2021) Copper exposure induces hepatic G0/G1 cell-cycle arrest through suppressing the Ras/PI3K/Akt signaling pathway in mice. Ecotoxicol Environ Saf 222:112518. https://doi.org/10.1016/j.ecoenv.2021.112518
Dang F, Nie L, Wei W (2021) Ubiquitin signaling in cell cycle control and tumorigenesis. Cell Death Differ 28:427–438. https://doi.org/10.1038/s41418-020-00648-0
Pack LR, Daigh LH, Meyer T (2019) Putting the brakes on the cell cycle: mechanisms of cellular growth arrest. Curr Opin Cell Biol 60:106–113. https://doi.org/10.1016/j.ceb.2019.05.005
Gousias K, Theocharous T, Simon M (2022) Mechanisms of cell cycle arrest and apoptosis in glioblastoma. Biomedicines 10(3):564–582. https://doi.org/10.3390/biomedicines10030564
Liu Y, Li X, Xiao S et al (2020) The Effects of Gold Nanoparticles on Leydig Cells and Male Reproductive Function in Mice. Int J Nanomedicine 15:9499–9514. https://doi.org/10.2147/ijn.S276606
Tewari D, Patni P, Bishayee A, Sah AN, Bishayee A (2022) Natural products targeting the PI3K-Akt-mTOR signaling pathway in cancer: A novel therapeutic strategy. Semin Cancer Biol 80:1–17. https://doi.org/10.1016/j.semcancer.2019.12.008
Zhou C, Du J, Zhao L et al (2021) GLI1 reduces drug sensitivity by regulating cell cycle through PI3K/AKT/GSK3/CDK pathway in acute myeloid leukemia. Cell Death Dis 12:231. https://doi.org/10.1038/s41419-021-03504-2
Liu JS, Huo CY, Cao HH et al (2019) Aloperine induces apoptosis and G2/M cell cycle arrest in hepatocellular carcinoma cells through the PI3K/Akt signaling pathway. Phytomedicine 61:152843. https://doi.org/10.1016/j.phymed.2019.152843
Cornu R, Béduneau A, Martin H (2022) Ingestion of titanium dioxide nanoparticles: a definite health risk for consumers and their progeny. Arch Toxicol 96:2655–2686. https://doi.org/10.1007/s00204-022-03334-x
Khorsandi L, Orazizadeh M, Moradi-Gharibvand N, Hemadi M, Mansouri E (2017) Beneficial effects of quercetin on titanium dioxide nanoparticles induced spermatogenesis defects in mice. Environ Sci Pollut Res Int 24:5595–5606. https://doi.org/10.1007/s11356-016-8325-2
Liberal V, De Miguel MP, Henze M, Nistal M, Reed SI (2010) Reduced spermatogonial proliferation and decreased fertility in mice overexpressing cyclin E in spermatogonia. Cell Cycle 9:4222–4227. https://doi.org/10.4161/cc.9.20.13544
Baranowska-Wójcik E, Szwajgier D, Oleszczuk P, Winiarska-Mieczan A (2020) Effects of Titanium Dioxide Nanoparticles Exposure on Human Health-a Review. Biol Trace Elem Res 193:118–129. https://doi.org/10.1007/s12011-019-01706-6
Verma A, Stellacci F (2010) Effect of surface properties on nanoparticle-cell interactions. Small 6:12–21. https://doi.org/10.1002/smll.200901158
Wang J, Wang J, Liu Y et al (2019) Aging-independent and size-dependent genotoxic response induced by titanium dioxide nanoparticles in mammalian cells. J Environ Sci (China) 85:94–106. https://doi.org/10.1016/j.jes.2019.04.024
Bernier MC, El Kirat K, Besse M, Morandat S, Vayssade M (2012) Preosteoblasts and fibroblasts respond differently to anatase titanium dioxide nanoparticles: a cytotoxicity and inflammation study. Colloids Surf B Biointerfaces 90:68–74. https://doi.org/10.1016/j.colsurfb.2011.09.044
Yu Q, Wang H, Peng Q, Li Y, Liu Z, Li M (2017) Different toxicity of anatase and rutile TiO(2) nanoparticles on macrophages: Involvement of difference in affinity to proteins and phospholipids. J Hazard Mater 335:125–134. https://doi.org/10.1016/j.jhazmat.2017.04.026
Liu L, Michowski W, Kolodziejczyk A, Sicinski P (2019) The cell cycle in stem cell proliferation, pluripotency and differentiation. Nat Cell Biol 21:1060–1067. https://doi.org/10.1038/s41556-019-0384-4
Gupta VK, Chaudhuri O (2022) Mechanical regulation of cell-cycle progression and division. Trends Cell Biol 32:773–785. https://doi.org/10.1016/j.tcb.2022.03.010
Matthews HK, Bertoli C, de Bruin RAM (2022) Cell cycle control in cancer. Nat Rev Mol Cell Biol 23:74–88. https://doi.org/10.1038/s41580-021-00404-3
Wang Z (2021) Regulation of Cell Cycle Progression by Growth Factor-Induced Cell Signaling. Cells 10:3327. https://doi.org/10.3390/cells10123327
Verma SK, Jha E, Panda PK et al (2018) Mechanistic Insight into Size-Dependent Enhanced Cytotoxicity of Industrial Antibacterial Titanium Oxide Nanoparticles on Colon Cells Because of Reactive Oxygen Species Quenching and Neutral Lipid Alteration. ACS Omega 3:1244–1262. https://doi.org/10.1021/acsomega.7b01522
Yang J, Liu J, Wang P, Sun J, Lv X, Diao Y (2021) Toxic effect of titanium dioxide nanoparticles on corneas in vitro and in vivo. Aging (Albany NY) 13:5020–5033. https://doi.org/10.18632/aging.202412
Stallaert W, Kedziora KM, Taylor CD et al (2022) The structure of the human cell cycle. Cell Syst 13:230-240.e3. https://doi.org/10.1016/j.cels.2021.10.007
Lee Y, Lahens NF, Zhang S, Bedont J, Field JM, Sehgal A (2019) G1/S cell cycle regulators mediate effects of circadian dysregulation on tumor growth and provide targets for timed anticancer treatment. PLoS Biol 17:e3000228. https://doi.org/10.1371/journal.pbio.3000228
Huang B, Hao M, Li C, Luo KQ (2022) Acetyltanshinone IIA reduces the synthesis of cell cycle-related proteins by degrading p70S6K and subsequently inhibits drug-resistant lung cancer cell growth. Pharmacol Res 179:106209. https://doi.org/10.1016/j.phrs.2022.106209
Engeland K (2018) Cell cycle arrest through indirect transcriptional repression by p53: I have a DREAM. Cell Death Differ 25:114–132. https://doi.org/10.1038/cdd.2017.172
Bejarano I, Rodríguez AB, Pariente JA (2018) Apoptosis Is a Demanding Selective Tool During the Development of Fetal Male Germ Cells. Front Cell Dev Biol 6:65. https://doi.org/10.3389/fcell.2018.00065
Gupta S, Kumar M, Chaudhuri S, Kumar A (2022) The non-canonical nuclear functions of key players of the PI3K-AKT-MTOR pathway. J Cell Physiol 237:3181–3204. https://doi.org/10.1002/jcp.30782
Cai Z, Wang J, Li Y et al (2023) Overexpressed Cyclin D1 and CDK4 proteins are responsible for the resistance to CDK4/6 inhibitor in breast cancer that can be reversed by PI3K/mTOR inhibitors. Sci China Life Sci 66:94–109. https://doi.org/10.1007/s11427-021-2140-8
Zhang Y, Zhang C, Li J et al (2022) Inhibition of AKT induces p53/SIRT6/PARP1-dependent parthanatos to suppress tumor growth. Cell Commun Signal 20:93. https://doi.org/10.1186/s12964-022-00897-1
Pei XD, Yao HL, Shen LQ et al (2020) α-Cyperone inhibits the proliferation of human cervical cancer HeLa cells via ROS-mediated PI3K/Akt/mTOR signaling pathway. Eur J Pharmacol 883:173355. https://doi.org/10.1016/j.ejphar.2020.173355
Funding
This work was supported by the Corps science and technology planning project (2023AB049) and the National Natural Science Foundation of China (21966027, 81560536).
Author information
Authors and Affiliations
Contributions
Guanling Song and Xiaojia Meng contributed to the study conception and design. Experimental execution was performed by Xiaojia Meng, Ruoyun Dong and Hongmei Chang. Data analyses were performed by Yuzhu Lei, Yizhong Yan and Yuhua Hu. The first draft of the manuscript was written by Ruoyun Dong, Xiaojia Meng and Guanling Song, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dong, R., Meng, X., Chang, H. et al. Titanium Dioxide Nanoparticles Induce Cell Cycle Arrest and Apoptosis through Inhibiting PI3K/AKT/mTOR Pathway in Spermatogonia. Biol Trace Elem Res (2023). https://doi.org/10.1007/s12011-023-03984-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12011-023-03984-7