Abstract
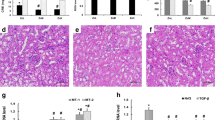
Zinc (Zn) is an essential element that regulates not only cellular immunity but also antioxidant and anti-inflammatory agents. The present study investigated the effect of Zn deficiency on renal cell apoptosis and its mechanism. A Zn-deficient kidney model in mice was created by a Zn-deficient diet. Mice were fed diets with different Zn levels for 41 days as follows: normal-Zn group (NG, 34 mg Zn/kg), low-Zn group (LG, 2 mg Zn/kg), and high-Zn group (HG, 100 mg Zn/kg). H&E staining showed that inflammatory cells and many erythrocytes exuded in the renal tissue space of the low-Zn group, and TUNEL staining indicated massive death of kidney cells in the low-Zn group. In the low-Zn group, the levels of oxygen free radicals (ROS) were significantly increased, the antioxidants were significantly decreased, and the total antioxidant capacity was decreased. Moreover, RT–qPCR and ELISA results showed that inflammatory factors (TNF-α, IL-1β, and IL-6) were significantly increased in the low-Zn group. In addition, the levels of p-IκBα, p-NF-κB p65, p-ERK, p-JNK, and p-p38 were significantly increased in the low-Zn group, indicating that zinc deficiency activates NF-κB and MAPK signalling as well as increases its expression. RT–qPCR analysis of apoptosis-related genes, including Bcl-2 Bax, Caspa8, Caspa6, and Caspa3, demonstrated that the expression levels of proapoptotic genes in mouse kidneys were significantly increased. Importantly, the in vitro results were consistent with the in vivo results. Together, these data suggested that zinc deficiency induces renal oxidative stress to activate NF-κB and MAPK signalling, thereby inducing renal cell apoptosis.








Similar content being viewed by others
Data Availability
The data of this study will be made available on reasonable request.
Code Availability
Not applicable.
References
Prasad AS (2013) Discovery of human zinc deficiency: its impact on human health and disease. Advances in Nutrition (Bethesda, Md) 4(2):176–190
Calesnick B, Dinan AM (1988) Zinc deficiency and zinc toxicity. Am Fam Phys 37:267–270
Choi S, Liu X, Pan Z (2018) Zinc deficiency and cellular oxidative stress: prognostic implications in cardiovascular diseases. Acta Pharmacol Sin 39(7):1120–1132
Ho E (2004) Zinc deficiency, DNA damage and cancer risk. J Nutr Biochem 15(10):572–578
Jarosz M, Olbert M, Wyszogrodzka G, Młyniec K, Librowski T (2017) Antioxidant and anti-inflammatory effects of zinc. Zinc-dependent NF-κB signaling Inflammopharmacology 25(1):11–24
Doboszewska U, Szewczyk B, Sowa-Kućma M, Noworyta-Sokołowska K, Misztak P, Gołębiowska J, Młyniec K, Ostachowicz B, Krośniak M, Wojtanowska-Krośniak A, Gołembiowska K, Lankosz M, Piekoszewski W, Nowak G (2016) Alterations of bio-elements, oxidative, and inflammatory status in the zinc deficiency model in rats. Neurotox Res 29(1):143–154
Samarghandian, S., Azimi-Nezhad, M., Farkhondeh, T., & Samini, F. (2017). Anti-oxidative effects of curcumin on immobilization-induced oxidative stress in rat brain, liver and kidney. Biomedicine & pharmacotherapy=Biomedecine&pharmacotherapie,87,223–229.
Jing H, Wang S, Wang Y, Shen N, Gao X-J (2020) Environmental contaminant ammonia triggers epithelial-to-mesenchymal transition-mediated jejunal fibrosis with the disassembly of epithelial cell-cell contacts in chicken. Sci Total Environ 726:138686. https://doi.org/10.1016/j.scitotenv.2020.138686
Totsuka K, Ueta T, Uchida T, Roggia MF, Nakagawa S, Vavvas DG, Honjo M, Aihara M (2019) Oxidative stress induces ferroptotic cell death in retinal pigment epithelial cells. Exp Eye Res 181:316–324
Morgan MJ, Kim YS, Liu Z (2007) Lipid rafts and oxidative stress-induced cell death. Antioxid Redox Signal 9(9):1471–1483
Abdelmegeed M, Mukhopadhyay P (2019) Understanding the roles and mechanisms of oxidative stress in diseases, tissue injury, and cell death in vivo and in vitro: therapeutic possibilities of antioxidants. Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association 127:70–71
Liu Xiao-Jing, Wang Yu-Qi, Shang Shao-Qian, Shiwen Xu, Guo Mengyao (2022) TMT induces apoptosis and necroptosis in mouse kidneys through oxidative stress-induced activation of the NLRP3 inflammasome. Ecotoxicology and Environmental Safety. 230:113167
Zhang T-Y, Chen T, Wan-Ying Hu, Li J-C, Guo M-Y (2021) Ammonia induces autophagy via circ-IFNLR1/miR-2188-5p/RNF182 axis in tracheas of chickens. BioFactors. https://doi.org/10.1002/biof.1795
Jing H, Wang F, Gao X-j (2022) Lithium intoxication induced pyroptosis via ROS/NF-κB/NLRP3 inflammasome regulatory networks in kidney of mice. Environmental Toxicology. 1–11. https://doi.org/10.1002/tox.23446.
Chen Y, Jing H, Chen M, Liang W, Yang J, Deng G, Guo M (2021) Transcriptional profiling of exosomes derived from Staphylococcus aureus-infected bovine mammary epithelial cell line MAC-T by RNA-Seq analysis. Oxidative Medicine and Cellular Longevity. 8460355. https://doi.org/10.1155/2021/8460355.
Chen Y, Yang J, Huang Z, Jing H, Yin B, Guo S, Deng G, Guo M (2021) Exosomal lnc-AFTR as a novel translation regulator of FAS ameliorates Staphylococcus aureus-induced mastitis. Biofactors. https://doi.org/10.1002/biof.1806
Li R, Jia Z, Trush MA (2016). Defining ROS in biology and medicine. Reactive oxygen species (Apex, N.C.), 1(1), 9–21
Zhang Q, Xue Y, Fu Y, Bao B, Guo M-y (2021) Zinc deficiency aggravates oxidative stress leading to inflammation and fibrosis in lung of mice. Biological Trace Element Research. https://doi.org/10.1007/s12011-021-03011-7.
Gammoh NZ, Rink L (2017) Zinc in infection and inflammation. Nutrients 9(6):624
Morgan MJ, Liu ZG (2011) Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res 21(1):103–115
Shen H, Oesterling E, Stromberg A, Toborek M, MacDonald R, Hennig B (2008) Zinc deficiency induces vascular pro-inflammatory parameters associated with NF-kappaB and PPAR signaling. J Am Coll Nutr 27(5):577–587
Prasad AS (2007) Zinc: mechanisms of host defense. J Nutr 137(5):1345–1349
Zhang Y, Xu Y, Chen B, Zhao B, Gao X-j (2021) Selenium deficiency promotes oxidative stress-induced mastitis via activating the NF-kappa B and MAPK pathways in dairy cow. Biological Trace Element Research. https://doi.org/10.1007/s12011-021-02882-0.
Guo YJ, Pan WW, Liu SB, Shen ZF, Xu Y, Hu LL (2020) ERK/MAPK signalling pathway and tumorigenesis. Exp Ther Med 19(3):1997–2007
Wang S, Gu J, Xu Z, Zhang Z, Bai T, Xu J, Cai J, Barnes G, Liu QJ, Freedman JH, Wang Y, Liu Q, Zheng Y, Cai L (2017) Zinc rescues obesity-induced cardiac hypertrophy via stimulating metallothionein to suppress oxidative stress-activated BCL10/CARD9/p38 MAPK pathway. J Cell Mol Med 21(6):1182–1192
Song N, Wang W, Wang Y, Guan Y, Shiwen Xu, Guo M-Y (2021) Hydrogen sulfide of air induces macrophage extracellular traps to aggravate inflammatory injury via the regulation of miR-15b-5p on MAPK and insulin signals in trachea of chickens. Sci Total Environ 771:145407. https://doi.org/10.1016/j.scitotenv.2021.145407
Luo M, Luo P, Zhang Z, Payne K, Watson S, Wu H, Tan Y, Ding Y, Sun W, Yin X, Zhang X, Liu G, Wintergerst K, Miao L, Cai L (2016) Zinc delays the progression of obesity-related glomerulopathy in mice via down-regulating P38 MAPK-mediated inflammation. Obesity (Silver Spring, Md.) 24(6):1244–1256
Zago MP, Mackenzie GG, Adamo AM, Keen CL, Oteiza PI (2005) Differential modulation of MAP kinases by zinc deficiency in IMR-32 cells: role of H(2)O(2). Antioxid Redox Signal 7(11–12):1773–1782
Li MS, Adesina SE, Ellis CL, Gooch JL, Hoover RS, Williams CR (2017) NADPH oxidase-2 mediates zinc deficiency-induced oxidative stress and kidney damage. Am J Physiol Cell Physiol 312(1):C47–C55
Sun W, Yang J, Wang W, Hou J, Cheng Y, Fu Y, Xu Z, Cai L (2018) The beneficial effects of Zn on Akt-mediated insulin and cell survival signaling pathways in diabetes. Journal of trace elements in medicine and biology : organ of the Society for Minerals and Trace Elements (GMS) 46:117–127
Yang F, Li B, Dong X, Cui W, Luo P (2017) The beneficial effects of zinc on diabetes-induced kidney damage in murine rodent model of type 1 diabetes mellitus. Journal of trace elements in medicine and biology : organ of the Society for Minerals and Trace Elements (GMS) 42:1–10
Chen Y, Yang J, Wang Y, Yang M, Guo M (2020) Zinc deficiency promotes testicular cell apoptosis in mice. Biol Trace Elem Res 195(1):142–149
Herbein G, Varin A, Fulop T (2006) NF-kappaB, AP-1, Zinc-deficiency and aging. Biogerontology 7(5–6):409–419
Ben-Neriah Y, Karin M (2011) Inflammation meets cancer, with NF-κB as the matchmaker. Nat Immunol 12(8):715–723
Zhang Q, Lenardo MJ, Baltimore D (2017) 30 years of NF-κB: a blossoming of relevance to human pathobiology. Cell 168(1–2):37–57
Mitchell S, Vargas J, Hoffmann A (2016) Signaling via the NFκB system. Wiley interdisciplinary reviews. Systems biology and medicine 8(3):227–241
Rink L, Gabriel P (2000) Zinc and the immune system. Proc Nutr Soc 59(4):541–552
Chen LF, Greene WC (2004) Shaping the nuclear action of NF-kappaB. Nat Rev Mol Cell Biol 5(5):392–401
Sun J, Nan G (2016) The mitogen-activated protein kinase (MAPK) signaling pathway as a discovery target in stroke. Journal of molecular neuroscience : MN 59(1):90–98
Author information
Authors and Affiliations
Contributions
XG and YX conceived and designed the experiments. YX, AL, and XL carried out the experiments. YX and XD processed the data. YX and XG wrote the paper. The manuscript was read and approved by all authors.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
All the animal experiments involved in this paper have been approved by the Animal Research Ethics Committee of Northeast Agricultural University (NAUMO-2021–0119).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, Y., Li, A., Li, X. et al. Zinc Deficiency Induces Inflammation and Apoptosis via Oxidative Stress in the Kidneys of Mice. Biol Trace Elem Res 201, 739–750 (2023). https://doi.org/10.1007/s12011-022-03166-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-022-03166-x