Abstract
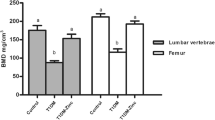
Insulin plays an important role in bone prevention of diabetic osteoporosis, but little is known about the relation between the bone mineral density (BMD) increase and the change of mineral element content after treated with insulin. To address this problem, male Wistar rats were randomly divided into three groups: normal group (n = 6), streptozotocin-induced diabetic group (n = 5), and streptozotocin-induced diabetic group with insulin treatment (n = 5). The femoral BMD was measured by dual energy X-ray absorptiometry, and the element content was determined by inductively coupled plasma atomic emission spectrometry (ICP-AES). The results showed that the femoral BMD in diabetic group was significantly lower than that in normal group (P < 0.01) but restored by insulin treatment (P < 0.01 vs diabetic group). ICP-AES analysis revealed that the element content of calcium (Ca), phosphorous (P), magnesium (Mg), strontium (Sr), and potassium (K) in diabetic group were remarkably lower than those in normal group (P < 0.01) but only Ca, P, and Mg content were significantly increased compared with diabetic group (P < 0.05) after insulin treatment. However, no significant differences were observed in element zinc (Zn) content among three groups. Our findings suggested that the loss of Ca, P, Mg, Sr, and K content accounted for the lower BMD in streptozotocin-induced diabetes rats, insulin treatment could restore BMD by increasing the content of Ca, P, and Mg.




Similar content being viewed by others
References
Seeman E (2003) Invited Review: pathogenesis of osteoporosis. J Appl Physiol 95:2142–2151
World Health Organization (2003) World Health Organization Prevention and Management of Osteoporosis, Technical Report Series, No. 921, WHO Marketing and Dissemination, Geneva
Krakauer JC, McKenna MJ, Buderer NF, Rao DS, Whitehouse FW, Parfitt AM (1995) Bone loss and bone turnover in diabetes. Diabetes 44:775–782
Kemink SA, Hermus AR, Swinkels LM, Lutterman JA, Smals AG (2000) Osteopenia in insulin-dependent diabetes mellitus; prevalence and aspects of pathophysiology. J Endocrinol Invest 23:295–303
Dominguez LJ, Muratore M, Quarta E, Zagone G, Barbagallo M (2004) Osteoporosis and diabetes. Reumatismo 56(4):235–241
Schwartz AV (2003) Diabetes Mellitus: does it Affect Bone? Calcif Tissue Int 73(6):515–519
Duarte VM, Ramos AM, Rezende LA, Macedo UB, Brandao-Neto J, Almeida MG, Rezende AA (2005) Osteopenia: a bone disorder associated with diabetes mellitus. J Bone Miner Metab 23(1):58–68
Herrero S, Calvo OM, Garcia-Moreno C, Martin E, San Roman JI, Martin M, Garcia-Talavera JR, Calvo JJ, Pino-Montes J (1998) Low bone density with normal bone turnover in ovariectomized and streptozotocin-induced diabetic rats. Calcif Tissue Int 62:260–265
Schwartz AV, Sellmeyer DE, Ensrud KE, Cauley JA, Tabor HK, Schreiner PJ, Jamal SA, Black DM, Cummings SR (2001) Older women with diabetes have an increased risk of fracture: a prospective study. J Clin Endocrinol Metab 86:32–38
Forsen L, Meyer HE, Midthjell K, Edna TH (1999) Diabetes mellitus and the incidence of hip fracture: results from the Nord-Trondelag Health Survey. Diabetologia 42:920–925
White CB, Turner NS, Lee GC, Haidukewych GJ (2003) Open ankle fractures in patients with diabetes mellitus. Clin Orthop 414:37–44
Folk JW, Starr AJ, Early JS (1999) Early wound complications of operative treatment of calcaneus fractures: analysis of 190 fractures. J Orthop Trauma 13:369–372
Thrailkill KM, Lumpkin CK Jr., Bunn RC, Kemp SF, Fowlkes JL (2005) Is insulin an anabolic agent in bone? Dissecting the diabetic bone for clues. Am J Physiol Endocrinol Metab 289:735–745
Bouillon R (1991) Diabetic bone disease [editorial]. Calcif Tissue Int 49:155–160
Hashizume M, Yamaguchi M (1993) Stimulatory effect of beta-alanyl-Lhistidinato zinc on cell proliferation is dependent on protein synthesis in osteoblastic MC3T3-E1 cells. Mol Cell Biochem 122:59–64
Pun KK, Lau P, Ho PW (1989) The characterization, regulation, and function of insulin receptors on osteoblast-like clonal osteosarcoma cell line. J Bone Miner Res 4:853–862
Mishima N, Sahara N, Shirakawa M, Ozawa H (2002) Effect of streptozotocin-induced diabetes mellitus on alveolar bone deposition in the rat. Arch Oral Biol 47:843–849
Glajchen N, Epstein S, Ismail F, Thomas S, Fallon M, Chakrabarti, S (1988) Bone mineral metabolism in experimental diabetes mellitus: Osteocalcin as a measure of bone remodeling. Endocrinology 123:290–295
Follak N, Kloting L, Wolf E, Merk H (2004) Delayed remodeling in the early period of fracture healing in spontaneously diabetic BB/OK rats depending on the diabetic metabolic state. Histol Histopathol 19:473–486
Follak N, Kloting I, Merk H (2005) Influence of diabetic metabolic state on fracture healing in spontaneously diabetic rats. Diabetes Metab Res Rev 21:288–296
Tein MS, Breen SA, Loveday BE, Devlin H, Balment RJ, Boyd RD, Sibley CP, Garland HO (1998) Bone mineral density and composition in rat pregnancy: effects of streptozotocin-induced diabetes mellitus and insulin replacement. Exp Physiol 83(2):165–174
Simeckova A et al (1990) The effect of streptozotocin-induced diabetes treated with insulin on the metabolism of calcium, magnesium and phosphorus. Vnitr Lek 36(6):526–530
Hasegawa T, et al (2003) Major-to-ultratrace element in bone-marrow fluid as determined by ICP-AES and ICP-MS. Analytical sciences vol. 19
Matsuura H et al (2001) Element in black tea leaves by ICP-AES and ICP-MS with the acid of size exclusion chromatography. Anal Sci 17:391
Jasminka Z, Ilich JZ, Kerstetter JE (2000) Nutrition in bone health revisited: a story beyond calcium. J Am Coll Nutr 19:715–737
Basabe Tuero B et al (2004) The influence of calcium and phosphorus intake on bone mineral density in young women. ArchLatinoam Nutr 54(2):203–208
Zeni S, Weisstaub A (2003) Bone mass changes in vivo during the entire reproductive cycle in rats feeding different dietary calcium and calcium/phosphorus ratio content. Calcif Tissue Int 73(6):594–600
Flynn A (2003) The role of dietary calcium in bone health. Proc Nutr Soc 62(4):851–858
Purroy J, Spurr NK (2002) Molecular genetics of calcium sensing in bone cells. Hum Mol Genet 11:2377–2384
Basabe Tuero B, Mena Valverde MC, Faci Vega M, Aparicio Vizuete A, Lopez Sobaler AM, Ortega Anta RM (2004) The influence of calcium and phosphorus intake on bone mineral density in young women. Arch Latinoam Nutr 54(2):203–208
Sasaki S (2006) Calcium, magnesium, and potassium as dietary nutrients. Clin Calcium 16(1):110–115
Pointillart A, Gueguen L (1993) Meal-feeding and phosphorus ingestion influence calcium bioavailability evaluated by calcium balance and bone breaking strength in pigs. Bone Miner 21(1):75–81
Carpenter TO, Barton CN, Park YK (2000) Usual dietary magnesium intake in NHANES III is associated with femoral bone mass. J Bone Min Res 15(1):S292
Kawaura A, Nishida Y, Takeda E (2005) Phosphorus intake and bone mineral density (BMD). Clin Calcium 15(9):1501–1506
Koshihara M, Katsumata S, Uehara M, Suzuki K (2005) Effects of dietary phosphorus intake on bone mineralization and calcium absorption in adult female rats. Biosci Biotechnol Biochem 69(5):1025–1028
Heaney RP (2004) Phosphorus nutrition and the treatment of osteoporosis. Mayo Clin Proc 79(1):91–97
Fawcett WJ, Haxby EJ (1999) Male DA Magnesium: physiology and pharmacology. BJA 83:302–320
Hartwig A (2001) Role of magnesium in genomic stability. Mut Res 475:113–121
Rude RK, Gruber HE, Norton HJ, Wei LY, Frausto A, Kilburn J (2006) Reduction of dietary magnesium by only 50% in the rat disrupts bone and mineral metabolism. Osteoporos Int 17(7):1022–1032
Matsuzaki H, Uehara M, Suzuki K (2005) Effect of dietary magnesium supplementation on bone loss in rats fed a high phosphorus diet. Magnes Res 18(2):91–96
Nishimuta M, Kodama N, Morikuni E, Yoshioka YH, Yamada H, Kitajima H, Takeyama H, Suzuki K (2004) Balance of magnesium positively correlates with that of calcium. J Am Coll Nutr 23(6):768S–770S
Lemann J, Pleuss JA, Gray RW (1993) Potassium causes calcium retention in healthy adults. J Nutr 123:1623–1626
Sebastian A, Harris ST, Ottaway JH, Todd KM, Morris RC Jr. (1994) Improved mineral balance and skeletal metabolism in postmenopausal women treated with potassium bicarbonate. N Engl J Med 330:1776–1781
Granda TG, Velasco A (2002) Sodium and potassium clearance rhythmicity in diabetic rats with insulin treatment. Life Sciences 71:2475–2487
Macdonald HM, New SA, Fraser WD, Campbell MK, Reid DM (2005) Low dietary potassium intakes and high dietary estimates of net endogenous acid production are associated with low bone mineral density in premenopausal women and increased markers of bone resorption in postmenopausal women. Am J Clin Nutr 81:923–933
New SA, Robins SP, Campbell MK, Martin JC, Garton MJ, Bolton-Smith C, Grubb DA, Lee SJ, Reid DM (2000) Dietary influences on bone mass and bone metabolism further evidence of a positive link between fruit and vegetable consumption and bone health? Am J Clin Nutr 71:142–151
Zhang Y, Cheng F, Li D, Wang Y, Zhang G, Liao W, Tang T, Huang Y, He W (2005) Investigation of elemental content distribution in femoral head slice with osteoporosis by SRXRF microprobe. Biol Trace Elem Res 103:177–185
Marie PJ (2005) Strontium as therapy for osteoporosis. Curr Opin Pharmacol 5:633–636
Marie PJ, Ammann P, Boivin G, Rey C (2001) Mechanisms of action and therapeutic potential of strontium in bone. Calcif Tissue Int 69(3):121–129
Coulombe J, Faure H, Robin B (2004) In vitro effects of strontium ranelate on the extracellular calcium-sensing receptor. Biochem Biophys Res 323(4):1184–1190
Reginster JY, Seeman E, De Vernejoul MC, Adami S, Compston J, Phenekos C (2005) Strontium ranelate reduces the risk of nonvertebral fractures in postmenopausal women with osteoporosis: Treatment of Peripheral Osteoporosis (TROPOS) study. J Clin Endocrinol Metab 90(5):2816–2822
Fei Y, Zhang M, Li M, Huang Y, He W, Ding W, Yang J (2007) Element analysis in femur of diabetic osteoporosis model by SRXRF microprobe. Micron 38(6):637–642
Acknowledgement
This work is supported by the Scientific Research Foundation of Graduate University of Chinese Academy of Sciences (No. 055101FM03) and China National Natural Sciences Foundation (No. 20571084).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, X., Fei, Y., Zhang, M. et al. Reversal of Osteoporotic Changes of Mineral Composition in Femurs of Diabetic Rats by Insulin. Biol Trace Elem Res 121, 233–242 (2008). https://doi.org/10.1007/s12011-007-8043-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-007-8043-1