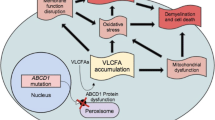
Abstract
Purpose of review
To present an updated appraisal of hematopoietic stem cell transplant (HSCT) and gene therapy for X-linked adrenoleukodystrophy (ALD) in the setting of a novel, presymptomatic approach to disease.
Recent findings
Outcomes in HSCT for ALD have been optimized over time due to early patient detection, improved myeloablative conditioning regimens, and adjunctive treatment for patients with advanced cerebral disease. Gene therapy has arrested disease progression in a cohort of boys with childhood cerebral ALD. New therapeutic strategies have provided the clinical basis for the implementation of Newborn Screening (NBS). With the help of advocacy groups, NBS has been implemented, allowing for MRI screening for the onset of cerebral ALD from birth.
Summary
Gene therapy and optimized hematopoietic stem cell transplant for childhood CALD have changed the natural history of this previously devastating neurological disease.


Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Moser HW, Loes DJ, Melhem ER, Raymond GV, Bezman L, Cox CS, et al. X-linked adrenoleukodystrophy: overview and prognosis as a function of age and brain magnetic resonance imaging abnormality. A study involving 372 patients. Neuropediatrics. 2000;31:227–39. https://doi.org/10.1055/s-2000-9236.
Moser AB, Kreiter N, Bezman L, et al. Plasma very long chain fatty acids in 3,000 peroxisome disease patients and 29,000 controls. Ann Neurol. 1999;45:100–10. https://doi.org/10.1002/1531-8249(199901)45:1<100::AID-ART16>3.0.CO;2-U.
Moser HW, Moser AB, Smith KD, Bergin A, Borel J, Shankroff J, et al. Adrenoleukodystrophy: phenotypic variability and implications for therapy. J Inherit Metab Dis. 1992;15:645–64. https://doi.org/10.1007/BF01799621.
Eichler FS, Ren JQ, Cossoy M, Rietsch AM, Nagpal S, Moser AB, et al. Is microglial apoptosis an early pathogenic change in cerebral X-linked adrenoleukodystrophy? Ann Neurol. 2008;63:729–42. https://doi.org/10.1002/ana.21391.
Lauer A, Da X, Hansen MB, et al. ABCD1 dysfunction alters white matter microvascular perfusion. BRAIN. 2017;140:3139–52. https://doi.org/10.1093/brain/awx262.
Korenke GC, Pouwels PJ, Frahm J, Hunneman DH, Stoeckler S, Krasemann E, et al. Arrested cerebral adrenoleukodystrophy: a clinical and proton magnetic resonance spectroscopy study in three patients. Pediatr Neurol. 1996;15:103–7.
Lin JE, Armour EA, Heshmati A, Umandap C, Couto JJ, Iglesias AD, et al. Pearls & Oy- sters: adolescent-onset adrenomyeloneuropathy and arrested cerebral adrenoleukodystrophy. Neurology. 2019;93:81–4. https://doi.org/10.1212/WNL.0000000000007755.
• Liberato AP, Mallack EJ, Aziz-Bose R, et al. MRI brain lesions in asymptomatic boys with X-linked adrenoleukodystrophy. Neurology. 2019;92:e1698–708. https://doi.org/10.1212/WNL.0000000000007294 Characterization of early cerebral lesions in ALD.
Raymond GV, Aubourg P, Paker A, et al. Survival and functional outcomes in boys with cerebral adrenoleukodystrophy with and without hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2018. https://doi.org/10.1016/j.bbmt.2018.09.036.
Van Geel BM, Bezman L, Loes DJ, et al. Evolution of phenotypes in adult male patients with X-linked adrenoleukodystrophy. Ann Neurol. 2001;49:186–94. https://doi.org/10.1002/1531-8249(20010201)49:2<186::AID-ANA38>3.0.CO;2-R.
Vogel BH, Bradley SE, Adams DJ, et al. Newborn screening for X-linked adrenoleukodystrophy in New York State: diagnostic protocol, surveillance protocol and treatment guidelines. Mol Genet Metab. 2015. https://doi.org/10.1016/j.ymgme.2015.02.002.
Wiens K, Berry SA, Choi H, et al. A report on state-wide implementation of newborn screening for X-linked adrenoleukodystrophy. Am J Med Genet Part A. 2019;179:ajmg.a.61171. https://doi.org/10.1002/ajmg.a.61171.
•• Eichler F, Duncan C, Musolino PL, Orchard PJ, De Oliveira S, Thrasher AJ, et al. Hematopoietic stem-cell gene therapy for cerebral adrenoleukodystrophy. N Engl J Med. 2017. https://doi.org/10.1056/NEJMoa1700554First successful phase 2/3 gene therapy trial in childhood cerebral ALD.
•• Gong Y, Berenson A, Laheji F, et al. Intrathecal adeno-associated viral vector-mediated gene delivery for adrenomyeloneuropathy. Hum Gene Ther. 2019;30:544–55. https://doi.org/10.1089/hum.2018.079 Preclinical experiments laying foundation for a possible gene therapy trial in adrenomyeloneuropathy.
Krivit W, Sung JH, Shapiro EG, Lockman LA. Microglia: the effector cell for reconstitution of the central nervous system following bone marrow transplantation for lysosomal and peroxisomal storage diseases. Cell Transplant. 1995;4:385–92. https://doi.org/10.1016/0963-6897(95)00021-o.
Cartier N, Aubourg P. Hematopoietic stem cell transplantation and hematopoietic stem cell gene therapy in X- linked adrenoleukodystrophy. Brain Pathol. 2009;20:857–62. https://doi.org/10.1111/j.1750-3639.2010.00394.x.
Benhamida S, Pflumio F, Dubart-Kupperschmitt A, et al. Transduced CD34+ cells from adrenoleukodystrophy patients with HIV-derived vector mediate long-term engraftment of NOD/SCID mice. Mol Ther. 2003;7:317–24. https://doi.org/10.1016/s1525-0016(03)00002-9.
Yamada T, Ohyagi Y, Shinnoh N, et al. Therapeutic effects of normal cells on ABCD1 deficient cells in vitro and hematopoietic cell transplantation in the X-ALD mouse model. J Neurol Sci. 2004;218:91–7. https://doi.org/10.1016/j.jns.2003.11.006.
Moser HW. Gene therapy in neurology. Eur Neurol. 1994;34:241–2. https://doi.org/10.1159/000117050.
Cartier N, Lopez J, Moullier P, Rocchiccioli F, Rolland MO, Jorge P, et al. Retroviral-mediated gene transfer corrects very-long-chain fatty acid metabolism in adrenoleukodystrophy fibroblasts. Proc Natl Acad Sci U S A. 1995;92:1674–8. https://doi.org/10.1073/pnas.92.5.1674.
Doerflinger N, Miclea JM, Lopez J, Chomienne C, Bougnères P, Aubourg P, et al. Retroviral transfer and long-term expression of the adrenoleukodystrophy gene in human CD34+ cells. Hum Gene Ther. 1998;9:1025–36. https://doi.org/10.1089/hum.1998.9.7-1025.
Mahmood A, Raymond GV, Dubey P, et al. Survival analysis of haematopoietic cell transplantation for childhood cerebral X-linked adrenoleukodystrophy: a comparison study. Lancet Neurol. 2007;6:687–92. https://doi.org/10.1016/S1474-4422(07)70177-1.
Shapiro E, Krivit W, Lockman L, et al. Long-term effect of bone-marrow transplantation for childhood-onset cerebral X-linked adrenoleukodystrophy. Lancet (Lond, Engl). 2000;356:713–8. https://doi.org/10.1016/S0140-6736(00)02629-5.
Peters C, Charnas LR, Tan Y, et al. Cerebral X-linked adrenoleukodystrophy : the international hematopoietic cell transplantation experience from 1982 to 1999 cerebral X-linked adrenoleukodystrophy : the international hematopoietic cell transplantation experience from 1982 to 1999. Blood. 2004;104:881–8. https://doi.org/10.1182/blood-2003-10-3402.
Loes DJ, Hite S, Moser H, Stillman AE, Shapiro E, Lockman L, et al. Adrenoleukodystrophy: a scoring method for brain MR observations. Am J Neuroradiol. 1994;15:1761–6. https://doi.org/10.1016/j.rcl.2013.11.008.
van den Broek BTA, Page K, Paviglianiti A, et al. Early and late outcomes after cord blood transplantation for pediatric patients with inherited leukodystrophies. Blood Adv. 2018. https://doi.org/10.1182/bloodadvances.2017010645.
Kuhl J-S, Kupper J, Baque H, et al. Potential risks to stable long-term outcome of allogeneic hematopoietic stem cell transplantation for children with cerebral X-linked adrenoleukodystrophy. JAMA Netw Open. 2018;1:e180769. https://doi.org/10.1001/jamanetworkopen.2018.0769.
Pierpont EI, Eisengart JB, Shanley R, Nascene D, Raymond GV, Shapiro EG, et al. Neurocognitive trajectory of boys who received a hematopoietic stem cell transplant at an early stage of childhood cerebral adrenoleukodystrophy. JAMA Neurol. 2017;74:710–7. https://doi.org/10.1001/jamaneurol.2017.0013.
Berger J, Forss-Petter S, Eichler FS. Pathophysiology of X- linked adrenoleukodystrophy. Biochimie. 2014;98:135–42. https://doi.org/10.1016/j.biochi.2013.11.023.
Powers JM, Pei Z, Heinzer AK, Deering R, Moser AB, Moser HW, et al. Adreno- leukodystrophy: oxidative stress of mice and men. J Neuropathol Exp Neurol. 2005;64:1067–79. https://doi.org/10.1097/01.jnen.0000190064.28559.a4.
Rockenbach FJ, Deon M, Marchese DP, Manfredini V, Mescka C, Ribas GS, et al. The effect of bone marrow transplantation on oxidative stress in X-linked adrenoleukodystrophy. Mol Genet Metab. 2012;106:231–6. https://doi.org/10.1016/j.ymgme.2012.03.019.
Mayer M, Noble M. N-acetyl-L-cysteine is a pluripotent protector against cell death and enhancer of trophic factor-mediated cell survival in vitro. Proc Natl Acad Sci. 1994;91:7496–500. https://doi.org/10.1073/pnas.91.16.7496.
Tolar J, Orchard PJ, Bjoraker KJ, et al. N-acetyl-L-cysteine improves outcome of advanced cerebral adrenoleukodystrophy. Bone Marrow Transplant. 2007. https://doi.org/10.1038/sj.bmt.1705571.
Magge RS, DeAngelis LM. The double-edged sword: neurotoxicity of chemotherapy. Blood Rev. 2015;29:93–100. https://doi.org/10.1016/j.blre.2014.09.012.
Bartelink IH, van Reij EML, Gerhardt CE, van Maarseveen E, de Wildt A, Versluys B, et al. Fludarabine and exposure-targeted busulfan compares favorably with busulfan/cyclophosphamide-based regimens in pediatric hematopoietic cell transplantation: maintaining efficacy with less toxicity. Biol Blood Marrow Transplant. 2014;20:345–53. https://doi.org/10.1016/j.bbmt.2013.11.027.
Beam D, Poe MD, Provenzale JM, et al. Outcomes of unrelated umbilical cord blood transplantation for X-linked adrenoleukodystrophy. Biol Blood Marrow Transplant. 2007;13:665–74. https://doi.org/10.1016/j.bbmt.2007.01.082.
Bartelink IH, Lalmohamed A, van Reij EML, et al. Association of busulfan exposure with survival and toxicity after haemopoietic cell transplantation in children and young adults: a multicentre, retrospective cohort analysis. Lancet Haematol. 2016;3:e526–36. https://doi.org/10.1016/S2352-3026(16)30114-4.
de Beer M, Engelen M, van Geel BM. Frequent occurrence of cerebral demyelination in adrenomyeloneuropathy. Neurology. 2014;83:2227 LP–2231. https://doi.org/10.1212/WNL.0000000000001074.
Kühl JS, Suarez F, Gillett GT, et al. Long-term outcomes of allogeneic haematopoietic stem cell transplantation for adult cerebral X-linked adrenoleukodystrophy. Brain. 2017. https://doi.org/10.1093/brain/awx016.
Waldhüter N, Köhler W, Hemmati PG, Jehn C, Peceny R, Vuong GL, et al. Allogeneic hematopoietic stem cell transplantation with myeloablative conditioning for adult cerebral X-linked adrenoleukodystrophy. J Inherit Metab Dis. 2019;42:313–24. https://doi.org/10.1002/jimd.12044.
• Cartier N, Hacein-bey-abina S, Bartholomae CC, et al. RESEARCH ARTICLE Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science. 2009;326:818–24 First in human gene therapy trial for childhood cerebral ALD.
van Geel BM, Assies J, Haverkort EB, Barth PG, Wanders RJ, Schutgens RB, et al. Delay in diagnosis of X-linked adrenoleukodystrophy. Clin Neurol Neurosurg. 1993;95:115–20.
Beckmann NB, Miller WP, Dietrich MS, Orchard PJ. Quality of life among boys with adrenoleukodystrophy following hematopoietic stem cell transplant. Child Neuropsychol. 2018;24:986–98. https://doi.org/10.1080/09297049.2017.1380176.
Kemper AR, Brosco J, Comeau AM, et al. Newborn screening for X-linked adrenoleukodystrophy: evidence summary and advisory committee recommendation. Genet Med. 2017;19. https://doi.org/10.1038/gim.2016.68.
Salzman A. Adrenoleukodystrophy patient perspective: turning despair into a gene therapy breakthrough. Hum Gene Ther. 2011;22:647–8. https://doi.org/10.1089/hum.2011.2503.
Salzman R. Venture philanthropy and gene therapy: lessons from adrenoleukodystrophy. Hum Gene Ther. 2016;27:14–8. https://doi.org/10.1089/hum.2015.29016.rsa.
van Geel BM, Poll-The BT, Verrips A, et al. Hematopoietic cell transplantation does not prevent myelopathy in X-linked adrenoleukodystrophy: a retrospective study. J Inherit Metab Dis. 2015;38:359–61. https://doi.org/10.1007/s10545-014-9797-1.
Schaumburg HH, Powers JM, Raine CS, et al. Adrenoleukodystrophy: a clinical and pathological study of 17 cases. Arch Neurol. 1975;32:577–91.
Kemp S, Berger J, Aubourg P. X-linked adrenoleukodystrophy: clinical, metabolic, genetic and pathophysiological aspects. Biochim Biophys Acta Mol basis Dis. 2012;1822:1465–74. https://doi.org/10.1016/j.bbadis.2012.03.012.
Gong Y, Mu D, Prabhakar S, Moser A, Musolino P, Ren J, et al. Adenoassociated virus serotype 9-mediated gene therapy for X-linked adrenoleukodystrophy. Mol Ther. 2015;23:824–34. https://doi.org/10.1038/mt.2015.6.
Finkel RS, Chiriboga CA, Vajsar J, Day JW, Montes J, de Vivo DC, et al. Treatment of infantile-onset spinal muscular atrophy with nusinersen: a phase 2, open-label, dose-escalation study. Lancet. 2016;388:3017–26. https://doi.org/10.1016/S0140-6736(16)31408-8.
Mendell JR, Al-Zaidy S, Shell R, et al. Single-dose gene- replacement therapy for spinal muscular atrophy. N Engl J Med. 2017. https://doi.org/10.1056/NEJMoa1706198.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Eric Mallack receives research support from the NINDS (5 K12 NS066274-08).
Bela Turk declares that he has no potential conflicts of interest.
Helena Yan declares that she has no potential conflicts of interest.
Florian S. Eichler receives research support from FDA Orphan Disease Group (R01 FD004127), NINDS (R01 NS072446, R01 NS082331), Retrophin, Minoryx, bluebird bio, and AGTC.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Pediatric Neurology
Rights and permissions
About this article
Cite this article
Mallack, E.J., Turk, B., Yan, H. et al. The Landscape of Hematopoietic Stem Cell Transplant and Gene Therapy for X-Linked Adrenoleukodystrophy. Curr Treat Options Neurol 21, 61 (2019). https://doi.org/10.1007/s11940-019-0605-y
Published:
DOI: https://doi.org/10.1007/s11940-019-0605-y