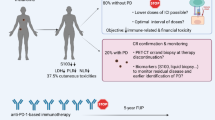
Abstract
Purpose of Review
Immune checkpoint inhibitors (ICIs) demonstrated robust antitumor activity and tolerable safety in advanced melanoma. Data on long-term outcome of patients who benefited from this therapy and who are still free of progression despite ICI discontinuation is now available. We review here the characteristics of long-term ICI responders and address the critical question of cure.
Recent Findings
Long-term outcome of patients with metastatic melanoma enrolled in large phase 2 and phase 3 clinical trials evaluating ICI in metastatic melanoma is now available. Durable responses, with more than 6 years of median follow-up, may persist after discontinuation. They occur more frequently in patients who achieved a complete response rather than in patients who had partial response or stable disease. Although long-term clinical benefit is more frequent in patients with high PDL-1 expression and smaller tumor burden, durable response may also be observed regardless of baseline characteristics. In patients with asymptomatic brain metastasis, combined immunotherapy (ipilimumab plus nivolumab) may also lead to long-term remission.
Summary
Clinical trials confirm the durable antitumor activity of ICI. Although the hope for cure seems reasonable for many patients in this situation, late relapses may occur and no relapse-predictive biomarkers have been identified yet. Long-term responders who relapse can respond to a rechallenge of ICI although data are limited concerning the rate and the duration of this new response.
Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Garbe C, Eigentler TK, Keilholz U, et al. Systematic review of medical treatment in melanoma: current status and future prospects. Oncologist. 2011;16:5–24.
Wolchock JD, Neyns B, Linette G, et al. Ipilimumab monotherapy in patients with pretreated advanced melanoma: a randomised, double-blind, multicentre, phase 2, dose-ranging study. Lancet Oncol. 2010;11:155–64.
Hodi FS, O’Day SJ, McDermotte DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711–23.
Ribas A, Puzanov I, Dummer R, et al. Pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractory melanoma (KEYNOTE-002): a randomised, controlled, phase 2 trial. Lancet Oncol. 2015;16(8):908–18.
Robert C, Schachter J, Long GV, et al. KEYNOTE-006 Investigators. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372(26):2521–32.
Robert C, Schachter J, Long GV, et al. Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med. 2015;372:2521–32.
Robert C, Long GV, Brady B, et al. Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med. 2015;372:320–30.
Weber JS, D’Angelo SP, Minor D, et al. Nivolumab versus chemotherapy in patients with advanced melanoma who progressed after anti–CTLA-4 treatment (CheckMate 037): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2015;16(4):375–84.
Robert C, Ribas A, Hamid O, et al. Durable complete response after discontinuation of pembrolizumab in patients with metastatic melanoma. J Clin Oncol. 2018;36:1668–74.
Ribas A, Hamid O, et al. Association of pembrolizumab with tumor response and survival among patients with advanced melanoma. JAMA. 2016;315(15):1600–9.
Robert C, Ribas A, et al. Three-year overall survival for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. JCO. 2016;34(Suppl 15):9503.
Schachter J, Ribas A, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet. 2017;390(10105):1853–62.
Hamid O, Puzanov I, et al. Final analysis of a randomized trial comparing pembrolizumab versus investigator-choice chemotherapy for ipilimumab-refractor advanced melanoma. Eur J Cancer. 2017;86:37–45.
Hamid O, Robert C, et al. Five-year survival outcomes for patients with advanced melanoma treated with pembrolizumab in KEYNOTE-001. Ann Oncol. 2019;30(4):582–8.
Hodi FS, Chiarion-Sileni V, et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018;19(11):1480–92.
Larkin J, Chiarion-Sileni V, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2019;381(16):1535–46.
•• Wolchok JD, Chiarion-Sileni V, et al. Long-term outcomes with nivolumab plus ipilimumab or nivolumab alone versus ipilimumab in patients with advanced melanoma. J Clin Oncol. 2022;40(2):127–37. These 6.5-year CheckMate 067 results showed durable, improved clinical outcomes with nivolumab plus ipilimumab or nivolumab versus ipilimumab in patients with advanced melanoma and with the combination over nivolumab monotherapy.
•• Robert C, Marabelle A, et al. Immunotherapy discontinuation — how, and when? Data from melanoma as a paradigm. Nat Rev Clin Oncol. 2022;17:707–15. For the first time in the management of patients with metastatic cancer, considering stopping therapy has become possible for patients who had considerable benefit from treatment. These observations first became apparent in patients with melanoma and are now beginning to be described for several other cancer types. Today, the best marker for long-term survival and a minimal risk of disease relapse after stopping therapy is the achievement of a CR, which should, therefore, become a new crucial end point for clinical trials.
Robert C, Ribas A, et al. Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol. 2019;20:1239–51.
Tawbi HA, Forsyth PA, et al. Long-term outcomes of patients with active melanoma brain metastases treated with combination nivolumab plus ipilimumab (CheckMate 204): final results of an open-label, multicentre, phase 2 study. Lancet Oncol. 2021;22(12):1692–704.
Di Giacomo AM, Chiarion-Sileni V, et al. Primary analysis and 4-year follow-up of the phase III NIBIT-M2 trial in melanoma patients with brain metastases. Clin Cancer Res. 2021;27:4737–45.
Long GV, Atkinson V, et al. Five-year overall survival from Anti-PD1 Brain Collaboration (ABC study): randomised phase 2 study of nivolumab or nivolumab+ipilimumab in patients with melanoma brain metastases. American Society of Clinical Oncology (ASCO) Congress; 2021. (abstr 9508)
•• Robert C, Carlino M, et al. Seven-year follow-up of the phase 3 KEYNOTE-006 study: pembrolizumab versus ipilimumab in advanced melanoma. JCO. 2023; In press. The results of this long-term follow-up showed that the survival benefits previously observed with pembrolizumab in advanced melanoma remain stable over time, with a 7-year OS of 37.8% for all patients and 41.2% for those who received first-line pembrolizumab, and a 7-year PFS of 23.8%. An OS benefit was seen regardless of BRAF-mutation status and receipt of prior BRAF/MEK-inhibitor therapy and poor prognostic characteristics.
Schadendorf D, Wolchok J, et al. Efficacy and safety outcomes in patients with advanced melanoma who discontinued treatment with nivolumab and ipilimumab because of adverse events: a pooled analysis of randomized phase II and III trials. J Clin Oncol. 2017;35(34):3807–14.
. Robert C, Hwu WJ, et al. Long-term safety of pembrolizumab monotherapy and relationship with clinical outcome: a landmark analysis in patients with advanced melanoma. Eur J Cancer. 2021;144:182–91. In landmark analyses, pembrolizumab efficacy was similar regardless of imAEs or systemic corticosteroid use.
Weber JS, Hodi FS, et al. Safety profile of nivolumab monotherapy: a pooled analysis of patients with advanced melanoma. J Clin Oncol. 2017;35(7):785–92.
Freeman-Keller M, Kim Y, et al. Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Canc Res. 2016;22(4):886–94.
Jansen YJL, Rozeman EA, et al. Discontinuation of anti-PD-1 antibody therapy in the absence of disease progression or treatment limiting toxicity: clinical outcomes in advanced melanoma. Ann Oncol. 2019;30(7):1154–61.
Dimitriou F, Zaremba A, et al. Sustainable responses in metastatic melanoma patients with and without brain metastases after elective discontinuation of anti-PD1-based immunotherapy due to complete response. Eur J Cancer. 2021;149:37–48.
Reck M, Rodriguez-Abreu D, et al. Five-year outcomes with pembrolizumab versus chemotherapy for metastatic non-small-cell lung cancer with PD-L1 tumor proportion score ≥ 50. J Clin Oncol. 2021;39(21):2339–49.
Sholl M, L. Biomarkers of response to checkpoint inhibitors beyond PD-L1 in lung cancer. Mod Pathol. 2022;35(Suppl 1):66–74.
Dimitriou F, Frauchiger AL, Urosevic-Maiwald M, et al. Sarcoid-like reactions in patients receiving modern melanoma treatment. Melanoma Res. 2018;28:1–236.
Tan AC, Emmett L, et al. FDG-PET response and outcome from anti-PD-1 therapy in metastatic melanoma. Ann Oncol. 2018;29(10):2115–20.
Vilain RE, Menzies AM, et al. Dynamic changes in PD-L1 expression and immune infiltrates early during treatment predict response to PD-1 blockade in melanoma. Clin Cancer Res. 2017;23(17):5024–33.
Dall'Olio FG, Marabelle A, et al. Tumour burden and efficacy of immune-checkpoint inhibitors. Nat Rev Clin Oncol. 2022;19(2):75–90.
Lee JH, Menzies AM, et al. Longitudinal monitoring of ctDNA in patients with melanoma and brain metastases treated with immune checkpoint inhibitors. Clin Cancer Res. 2020;26(15):4064–71.
Marsavela G, Lee J, et al. Circulating tumor DNA predicts outcome from first-, but not second-line treatment and identifies melanoma patients who may benefit from combination immunotherapy. Clin Cancer Res. 2020;26(22):5926–33.
Jamshidi A, Liu MC, et al. Evaluation of cell-free DNA approaches for multi-cancer early detection. Cancer Cell. 2022;40(12):1537–49.
Girault I, Adam J, et al. A PD-1/PD-L1 proximity assay as a theranostic marker for PD-1 blockade in patients with metastatic melanoma. Clin Cancer Res. 2022;28(3):518–25.
Helmink BA, Reddy SM, et al. B cells and tertiary lymphoid structures promote immunotherapy response. Nature. 2020;577(7791):549–55.
Asrir A, Tardiveau C, et al. Tumor-associated high endothelial venules mediate lymphocyte entry into tumors and predict response to PD-1 plus CTLA-4 combination immunotherapy. Cancer Cell. 2022;40(3):318–34.
Shen S, Vagner S, et al. Persistent cancer cells: the deadly survivors. Cell. 2020;183(4):860–74.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Djaouida Belkadi-Sadou, Antoine Marchand, and Severine Roy declare no conflict of interest. Celine Boutros has received travel fees from Pierre Fabre, Amgen, and MSD, and has received compensation from MSD for participation in conferences, and from BMS for participation to advisory boards. Emilie Routier has received compensation from BMS, Novartis, and Pierre Fabre, and conference fees from BMS, Novartis, and Pierre Fabre. Caroline Robert reports consultancy for BMS, Roche, Novartis, Pierre Fabre, MSD, Sanofi, AstraZeneca, and Merck, and is cofounder of Ribonexus.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boutros, C., Belkadi-Sadou, D., Marchand, A. et al. Cured or Not? Long-term Outcomes of Immunotherapy Responders. Focus on Melanoma. Curr Oncol Rep 25, 989–996 (2023). https://doi.org/10.1007/s11912-023-01429-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11912-023-01429-x