Abstract
Purpose of Review
Progressive multifocal leukoencephalopathy (PML) is a severe opportunistic infection that remains an important cause of morbidity and mortality in people living with HIV (PLWH). Immune checkpoint molecules are negative regulators of the immune response that have been targeted as a strategy to bolster anti-viral immunity in PML, with varied outcomes reported. While initiation and optimization of antiretroviral therapy remains the standard of care in HIV-related PML, the specific opportunities and risks for checkpoint blockade in these cases should be explored.
Recent Findings
As of April 15, 2022, only 5 of the 53 total published cases of PML treated with checkpoint blockade had underlying HIV infection; four of these had a favorable outcome. The risk of promoting immune reconstitution inflammatory syndrome is a major concern and underscores the importance of patient selection and monitoring.
Summary
Checkpoint blockade warrants further exploration as a potentially promising option for treatment escalation in HIV-related PML.
Similar content being viewed by others
Introduction
Progressive multifocal leukoencephalopathy (PML) is a rare and often fatal central nervous system (CNS) disease caused by the JC virus (JCV) [1]. The transmissible form of JCV, or archetype variant, commonly infects the general population in childhood, establishing asymptomatic latent or persistent infection. In individuals with compromised cellular immunity, however, JCV can reactivate and undergo serial genomic mutations; this in turn can result in the acquired ability of the virus to infect CNS glial cells with development of PML [2]. Even in those with immune deficiencies, the rarity of these sequential genomic rearrangements may in part explain the low estimated incidence of PML of 1:200,000 [3, 4].
The major underlying conditions contributing to PML cases include human immunodeficiency virus (HIV) infection, hematological malignancy, and exposure to immunomodulatory therapies [5]. In contrast with most opportunistic infections, no antiviral treatment currently exists for PML. As such, the only viable treatment strategies are aimed at rapidly achieving immune reconstitution. These include standard-of-care introduction of antiretroviral therapy (ART) in HIV-related PML, and discontinuation of any immunosuppressive treatments. However, in many patients with PML, reversing the underlying immunosuppression may not be accomplished, which has led to experimental treatment strategies aimed at augmenting antiviral immune responses [1].
Advancing these therapies is challenging due to the rarity and severity of PML, presenting ethical and methodological concerns regarding clinical trial design. Moreover, no animal model exists for PML and the virus is difficult to propagate in culture [1]. The collective treatment experience for PML is thus largely limited to case reports or small case series, making it difficult for the treating physician to weigh treatment options beyond standard-of-care.
PML in HIV
Despite significant improvements in the management of HIV infection and associated complications, PML remains an important cause of morbidity and mortality among people living with HIV (PLWH). Prior to the AIDS pandemic, PML was first described as a rare occurrence in lymphoproliferative disease [6]. The AIDS pandemic resulted in a dramatic 12-fold increase in the frequency of PML between the early 1980s and early 1990s due to the significant immunosuppression of HIV infection [7]. The introduction of ART for PLWH has since lessened the impact of PML on this population. The incidence of PML decreased from a peak estimate of 14.8 cases per 1000 patient-years at risk (PYR) pre-ART to 2.6 cases per 1000 PYR during early years of ART in 2005, with a further decline to 0.8 cases per 1000 PYR by 2011 [8].
While widespread availability of ART and a greater recognition of both PML and PML-IRIS have improved survival from a dismal 10% in the pre-ART era [7], survival remains poor at approximately 50% [9, 10, 11••], underscoring the continued devastating consequences of JCV infection. Indeed, even among survivors, the majority are left with significant disability, explained by the extensive tissue injury associated with PML with marked volume loss observed on brain MRIs [9, 12••].
Although the past decade has seen a decline in the proportion of patients with HIV-related PML, cohort studies show HIV remains the single most frequent underlying condition, accounting for approximately 50% of total PML cases [12••, 13, 14••]. The distinct predisposition to PML in the setting of HIV infection remains incompletely unexplained. Synergistic effects of PML and HIV coinfection have been proposed. One hypothesis suggests HIV infection affects the CNS microenvironment with an up-regulation of adhesion molecules on endothelial cells which may facilitate the trafficking of JCV-infected B cells into the brain [13]. Moreover, the HIV trans-regulatory protein Tat has been reported to increase the activity of the JCV late promoter [15] and to enhance replication of JCV [1, 16]. Exploring this association further may provide insight when assessing what types of therapies may prove most beneficial for HIV-related PML. The continued impact of PML in PLWH, with persistently high morbidity and mortality rates, warrants the exploration of adjunctive therapies for HIV-associated PML.
Immune Checkpoint Inhibitors
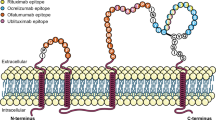
Immune checkpoint inhibitors (ICIs) have revolutionized the therapeutic approach to cancer treatment, opening the door for their application to re-establish immune competence in other diseases. ICIs block the inhibitory regulatory immune checkpoint pathways underlying immune exhaustion, thereby rescuing effective immune responses [17] (Fig. 1). The Food and Drug Administration (FDA) approved ipilimumab as the first ICI for the treatment of unresectable or metastatic melanoma in 2011 [18]. ICIs have since been widely applied to the treatment of a variety of cancers once thought less susceptible to treatment [17, 19]. Despite this success, the majority of patients with cancer do not respond to checkpoint inhibitors and so the identification of predictive biomarkers is an active area of investigation [20, 21].
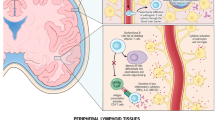
In the setting of impaired viral clearance and chronic exposure to high antigen burden, immune exhaustion may progressively dampen anti-viral immune responses leading to viral persistence (A). Treatment with immune checkpoint inhibitors (ICI), such as PD-1 inhibitors, may restore anti-viral immune responses and lead to effective viral clearance (B). The tipping point between effective immune reconstitution and immune activation inflammatory syndrome (IRIS) (C) is marked by development of new or worsening clinical symptoms attributable to the immune response itself rather than progression of the infection. In the setting of ART, adjunctive treatment with ICI could promote this overshoot
The mechanism of action of ICIs suggests their potential for restoring immune function in other diseases. T cell exhaustion is a feature of numerous cancers and chronic infections, such as PML [4, 22, 23]. T cell exhaustion is an anthropomorphic term used to indicate the programmed response of immune cells that occurs in the setting of persistent antigen exposure and T cell receptor (TCR) stimulation [24] and leads to progressive curtailing of T cell polyfunctionality. While per se not necessarily a dysfunctional state—indeed it appears evolutionarily conserved to protect tissues from excessive immune-mediated injury—T cell exhaustion reduces the ability to recognize and eradicate infected or neoplastic cells. Exhausted T cells are identified functionally and by persistently high expression of multiple inhibitory receptors, including cytotoxic T lymphocyte antigen 5 (CTLA-4), programmed cell death protein 1 (PD-1), T cell immunoglobulin and mucin domain 3 (TIM3), lymphocyte activation gene 3 (LAG3), and T cell immunoreceptor with Ig and ITIM domains (TIGIT) [25]. CTLA-4, PD-1 and its ligand PDL-1 have been developed as druggable targets, whereby their blockade re-establishes T cell activity. Importantly, it appears clear from animal models and human disease that the pool of exhausted T cells is phenotypically and functionally heterogeneous [26], with only a fraction of cells, most notably represented by TCF1 + memory-like T cells, able to respond to checkpoint blockade [27, 28, 29••, 30]. The proportion of these T cell subpopulations, versus terminally differentiated exhausted cells incapable of rescue, may greatly influence overall treatment response [31]. The tumor microenvironment itself has been identified as an important factor in treatment response, with PDL1 expression in biopsy samples already an FDA-approved predictive biomarker and the number and spatial arrangement of tumor infiltrating-cells appearing promising [20, 21].
The shared prominence of T cell exhaustion between cancer and HIV [32], as well as the success of ICIs for some cancers [17], have provided fuel for the exploration of ICIs for treating not only cancers but also opportunistic infections in PLWH.
Use of Checkpoint Inhibitors in HIV Populations
The advancement of antiretroviral therapy (ART) for PLWH has significantly reduced the risk of dying from AIDS-related illnesses. Despite this, PLWH still today have an increased risk of developing and dying from both AIDS-defining and non-AIDS-defining cancers [33, 34]. However, PLWH have been largely excluded from clinical trials investigating ICI for cancer treatment [35], due to concerns regarding the risks of activating an immune response against HIV-infected host cells, as well as the concern of developing immune-related adverse events (irAEs) across organ systems [32]. Recognizing the increased risk of incidence and mortality from cancers in populations with HIV [33, 34], as well as the benefit of ICIs for some cancers [17], more recent studies have sought to include PLWH in ICI clinical trials.
The application of ICIs for cancer treatment in a PLWH was first reported in a 2011 case report, with the use of ipilimumab for treating metastatic melanoma [36]. The therapy was tolerated well, showing reduced size of all four liver lesions and no adverse effect on HIV viral load or CD4 + cell counts. ICIs have since been applied in other PLWH. Kim and Cook [37] published a systematic review in 2019, detailing 73 PLWH who received ICI therapy for advanced-stage cancer. Of 28 patients with undetectable HIV load and paired pre-treatment and post-treatment data, 26 patients (93%) continued to have HIV levels below the limit of detection and essentially stable CD4 cell counts (mean [SD] increase, 12.3 [28.5]/μL). Six patients (8.6%) reported grade 3 or higher irAEs, further suggesting ICI therapy was generally well tolerated. Notably, there was no significant difference in efficacy of ICI therapy between patients with and without HIV, with response rates of 63% for Kaposi sarcoma, 30% for non-small cell lung cancer, and 27% for melanoma [37]. ICIs continue to be explored in PLWH, with recent studies supporting the safety of pembrolizumab for people taking ART [38••], as well as in combination with chemotherapy [39]. These findings suggest that ICI may be a safe and efficacious therapy for PLWH.
Recognizing the accumulating evidence supporting the safety and efficacy of ICI therapy in patients with HIV, the FDA published a guidance statement advocating the inclusion of people with HIV in ICI trials [40]. Despite this, a recent study by Vora et al. in 2021 concluded that the population with HIV continues to be excluded from most ICI trials [35]. Including PLWH in ICI clinical trials is necessary to provide access to groundbreaking treatments for this vulnerable population.
Effect of Checkpoint Inhibitors on Markers of HIV Infection
The HIV reservoir, comprised of latently infected host cells carrying the integrated viral genome, presents challenge for achieving permanent viral suppression in PLWH [41]. CD4 + cells with increased expression of immune checkpoints, such as PD-1, are major sources of replication-competent virus [42], and their levels correlate with harbored HIV DNA levels [43, 44]. This suggests that ICIs may impact HIV replication by potentially targeting latently infected cells.
In vitro, an ICI blocking PD-1 engagement increased the survival and proliferation of HIV-specific CD8 + T cells, as well as enhanced cytokine production to antigen exposure [45]. In vitro studies targeting immune checkpoints suggest an effect on reversing HIV latency [46, 47]. The impact of ICI on the HIV reservoir was described for the first time in a patient with HIV infection with metastatic melanoma in 2015 [48]. In addition to a decrease in plasma HIV RNA, there was a significant difference in number and phenotype of CD4 + T cells, predominantly an increase in total memory and effector memory CD4 + T cells [48].
In 2018, a case report by Guihot et al. [49••] described a profound decrease in the HIV reservoir following nivolumab treatment in an HIV-infected patient via a “shock and kill” mechanism—a brief reactivation of HIV replication within CD4 + T cells and a reduction in exhausted CD4 + and CD8 + T cells, followed by a dramatic increase in HIV-specific CD8 + T cells [49••]. However, a subsequent 2018 study detailed 3 HIV-positive patients receiving either nivolumab or pembrolizumab for cancer and with no consistent effect on HIV-specific T cell responses [50]. These inconsistent findings prompted a thorough systematic review in 2020 of the impact of ICI on the HIV reservoir [51]. Among 176 participants included, HIV plasma loads remained unchanged in 91.9%, and CD4 + cell count remained stable in 60.7%, increased in 24.6%, and decreased in 14.7%. More recently, a prospective study in 32 people with HIV on ART and cancer found that treatment with pembrolizumab led to modest transient increase in median viral transcription [52], while a separate study in 32 PLWH and cancer found a decrease in cell-associated HIV-DNA in 6 patients while 4 had transient increase [53]. Taken together, these findings suggest the potential of ICI for reversing viral latency is likely limited and variable, but worthy of further investigation. While PD-1 is effectively blocked on T cells in the cerebrospinal fluid (CSF) compartment following systemic administration [54••], to what extent drug reaches HIV reservoir-harboring cells in the brain parenchyma is unknown. An ongoing study investigating CNS effects of pembrolizumab for patients with HIV on ART (NCT03239899) addresses this open question.
These studies do however clearly demonstrate that ICI therapy is generally well tolerated among HIV populations. This, combined with ICI’s success in treating cancers and other chronic infections in PLWH, has opened the door for their investigation in HIV-associated PML.
Safety and Efficacy of Checkpoint Inhibition for PML in HIV
The involvement of the PD-1 pathway in PML contributes to the rationale for exploring ICIs to restore the immune response and improve viral clearance. Patients with PML display higher percentages of PD-1+ CD4+ and CD8+ T cells in the CSF and blood [54••, 55] and increased expression of PD-1 on JCV-specific CD8+ cytotoxic T-lymphocytes [55]. Moreover, blocking PD-1 in vitro resulted in an increase in IFN-γ expression in JCV-stimulated CD8+ T cells in an HIV-positive patient with active PML [55] and increased PD-1 and PD-L1 expression has been described in post-mortem PML lesion tissue [54••]. These findings suggest that targeting the PD-1 pathway through checkpoint inhibition may be an effective strategy to improve the cellular immune response to JCV.
ICIs have been explored in limited cases of patients with PML for whom immune reconstitution was not readily achieved and thus had low expectation for survival. As of April 15, 2022, only 5 of the 53 published cases of treated PML [5, 54••, 56,57,58,59,60,61,62,63,64,65,66,67, 68••, 69,70,71,72,73,74, 75••, 76,77,78,79,80, 81••, 82], although the number of unpublished cases of treated PML, both related to HIV and otherwise, exceeds this (personal communication). Nevertheless, evaluating the overall available experience and outcome of checkpoint inhibition in PML will likely provide helpful insights when translating this therapy’s potential to the HIV-PML population (Supplemental Table). Of the published patients, 41 (77%) received pembrolizumab, 11 (21%) received nivolumab, and a single case was treated with atezolizumab. In some cases, checkpoint inhibition demonstrated favorable outcome, with enhanced JCV-specific CD4 + and CD8 + T cell immune response [54••, 78, 82], a decline in JCV CSF copy number [54••, 56, 57, 62, 72, 73, 75••, 79, 83], and stable or improved clinical course [54••, 62, 67, 68••, 72,73,74, 75••, 78,79,80, 81••, 82].
Published cases of HIV-related PML treated with ICI are summarized in Table 1. Of the five patients with HIV-related PML, four were treated with pembrolizumab [54••, 75••, 81••]. Of these, one was newly stable on ART and two had a long history of ART, both with plasma HIV viral load less than the limit of detection at the time of pembrolizumab treatment; the fourth had a history of ART non-adherence and restarted ART contemporaneously to ICI treatment due to rapid worsening of PML. Of note, this last patient had evident contrast enhancement on baseline MRI [81••]. Three had pre- and post-treatment JCV CSF viral load measurements and exhibited a decrease between baseline and final visit upon treatment with ICI [54••, 75••]. Three showed significant improvement of neurological deficits and decrease in lesion size by MRI, with no recurrence of PML at last follow-up [54••, 75••, 81••]. One patient experienced clinical and radiological stabilization associated with increase in JCV-specific T cell response within 1 month of the first dose of pembrolizumab. This patient received an additional dose of pembrolizumab a year later due to renewed worsening of clinical symptoms and imaging, again followed by decline in CSF JCV load [54••]. None of the four developed notable adverse events nor PML-IRIS [54••, 75••, 81••].
Roos-Weil et al. [68••] documented a published case using nivolumab for HIV-associated PML in an individual with concomitant T cell lymphoma. This patient presented with severe disability (modified Rankin score of 5) and a JCV CSF viral load of over 200,000 copies/mL at the time of treatment. Unlike the previous report, this patient rapidly worsened following the first infusion, with no evidence of PML-IRIS by MRI, and death was attributed to progression of PML shortly thereafter [68••].
An optimal response to checkpoint inhibitors can be conceptualized as a balance between antigen burden and appropriate degree of immune reinvigoration. Suboptimal outcomes could therefore result on one end from inadequate reinvigoration, or on the other, from excessive inflammatory response leading to irAEs or, in the case of PML, to PML-IRIS (Fig. 1). At least two of the reported cases of HIV-related PML treated with ICI, the modest CSF viral load and disability (modified Rankin score of 3 and 2) at presentation [54••], suggesting relatively less advanced disease and possibly less severe immune exhaustion, may have favored their positive outcome compared to the case with poor outcome [68••].
Evaluating the collective published experience of ICI for the treatment of non-HIV related PML may provide further insight into what other factors are important to consider when weighing the likelihood of response to checkpoint inhibition. High CSF JCV load and severe disability suggesting advanced or aggressive PML, as well as severe lymphopenia, intrinsic T cell dysfunction, or ongoing treatment with immune suppressive therapies, each conceivably associated with decreased ability to mount any immune response, appear to be common features among cases with poor outcome [54••, 59, 61, 76, 79, 80, 84]. The presence of detectable JCV-specific T cells at baseline may on the other hand be a positive indicator [54••, 61, 78, 82], with similar observations seen in predicting ICI response in cancer [85]. This is further supported by in vitro evidence that PD-1 blockade resulted in an increase in JCV-specific IFN-γ immune response in healthy volunteers, but only in those with a detectable number of JCV-specific CD8 + T cells at baseline [39].
Challenges of Checkpoint Inhibitor Therapy for PML in HIV
In our center, HIV-related PML is most commonly observed in untreated HIV infection but can also occur in the setting of ART non-adherence or resistance. Standard-of-care for ART-naive patients with PML is first and foremost represented by immediate initiation of ART, which alone is associated with survival of more than half of cases. For patients with suboptimal HIV suppression related to non-adherence or resistance, first-line treatment is to optimize the ART regimen; with current potent ART regimens, there is no clear evidence suggesting improved outcome of PML using therapies with higher CNS penetration effectiveness score (CPE) [86,87,88]. The appropriate timing to consider adjunctive strategies to augment JCV anti-viral responses beyond standard-of-care is not established. Adjunctive strategies likely would be reserved for cases with low likelihood of achieving timely immune reconstitution in the face of progressing disease. While checkpoint inhibition may represent one possible consideration, several risks must be weighed.
The main concern is the risk of developing immune reconstitution inflammatory syndrome (IRIS). IRIS is a dysregulated, hyper-inflammatory response that can result in immune-mediated damage. In HIV-associated PML, IRIS is characterized by a worsening of neurological symptoms associated with an increase in CD4 + T cell count and a reduction in HIV RNA plasma levels [1]. The inflammatory features of PML-IRIS can be appreciated by MRI, with contrast enhancement, edema, and/or mass effect [89]. IRIS can be distinguished into “paradoxical IRIS,” occurring when symptoms of a known PML diagnosis worsen and “unmasking IRIS” when the inflammatory response to a previously inapparent infection results in new clinical symptoms [1]. While re-invigorating the immune system is an effective strategy to improve viral clearance, introducing checkpoint inhibition in combination with ART could enhance the risk of developing IRIS. Indeed, studies suggest as many as 25–30% of HIV patients on ART develop IRIS [90], and, specifically in the setting of PML, an estimated 28% of PML-IRIS cases are fatal [91]. Adjunctive therapy with checkpoint inhibition in patients newly initiating or escalating ART could compound the risk of developing IRIS. Importantly, the window for developing symptomatic IRIS is broad and unpredictable, occurring anytime up to 26 weeks after ART initiation [92]. This highlights a potentially prolonged continued risk for triggering IRIS in scenarios where ART does not quickly achieve adequate immune reconstitution to control infection and ICIs are considered.
The specific risk for developing PML-IRIS following ICI is not known. The majority of reported cases that used ICI as an experimental strategy for PML included patients with profound immune suppression that could not otherwise be reversed, and so the relatively low occurrence of IRIS in this setting is likely not generalizable to the HIV population. Independently of ICIs, known risk factors for developing IRIS include a history of multiple opportunistic infections, low starting CD4 count, and rapid decrease in HIV plasma viral load following initiation of ART [92, 93]. Close monitoring for development of PML-IRIS and timely management thus become particularly critical in the context of checkpoint inhibition. Steroids are the mainstay of treatment for PML-IRIS, although there is no standardized dosing schema. It is important to consider that corticosteroids dampens the JCV-specific cellular immune response, and while they have established benefit for the treatment of clinically manifest PML-IRIS, their use for prevention of IRIS is not recommended [94, 95].
Another challenge of checkpoint inhibition to note is the concern of triggering irAEs. irAEs have been reported across organ systems in response to ICIs, and can lead to colitis, pneumonitis, rash, or other organ dysfunctions [32]. Most irAEs in response to ICI are mild, but some can be serious and even life-threatening [96].
In two reported cases, PML has been diagnosed following checkpoint inhibition for other infections. One reports the use of nivolumab for a PML patient with stage IV Hodgkin lymphoma in 2018. To note, the patient’s lymphoma and steroid use rendered the patient highly immunocompromised. Therefore, it is difficult to conclude if the development of PML was due to the nivolumab, or rather an unfortunate complication of multiple factors [97]. In the second case report in 2019, a patient with stage IV Hodgkin lymphoma given nivolumab then displayed clinical signs of PML and IRIS diagnosed by brain biopsy, consistent with unmasking PML [58]. This patient survived 3 years, which significantly exceeds the 2-month median survival for PML patients with hematological malignancy [98]. Checkpoint inhibition thus appears to have promoted effective immune reconstitution to control the unmasked PML. As checkpoint inhibitors become more broadly applied to immunocompromised patients, the incidence of unmasking PML may increase [4].
Conclusions
PML is a devastating disease that continues to pose a significant burden due the lack of JC virus-specific treatment options. Still today, HIV accounts for a significant proportion of PML cases [11••].
ICIs have garnered much attention due to their success in treating numerous cancers. Their mode of action, represented by re-invigoration of the immune system, could be beneficial for promoting antiviral responses in PML, and more specifically in HIV-related PML when ART fails to control infection. Although PLWH have largely been excluded from ICI clinical trials [24], accumulating evidence demonstrates that ICIs are generally well tolerated in populations with HIV infection [26].
Studies exploring ICIs for patients with PML thus far have shown heterogenous results, not dissimilar to the experience in oncology. Nevertheless, ICIs hold promise as a strategy to promote immune reconstitution in selected cases of PML, and possibly HIV-associated PML. The specific risk of ICI in promoting PML-IRIS or IRIS in general is not completely understood, nor how this risk might affect appropriate timing of treatment or patient selection.
Robust biomarkers predictive of ICI treatment response are lacking. The experience available to date suggests profound lymphopenia and absence of any detectable anti-JCV activity are associated with poor likelihood of treatment response. Furthermore, study of ICI-responsive T cell subpopulations may provide insight into the inconsistencies observed, as well as into strategies to improve efficacy across indications. Formal, prospective investigations of ICI are necessary to determine whether ICI should be included in treatment escalation regimens of HIV-related PML.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Cortese I, Reich DS, Nath A. Progressive multifocal leukoencephalopathy and the spectrum of JC virus-related disease. Nat Rev Neurol. 2021;17(1):37–51.
Gosert R, Kardas P, Major EO, Hirsch HH. Rearranged JC virus noncoding control regions found in progressive multifocal leukoencephalopathy patient samples increase virus early gene expression and replication rate. J Virol. 2010;84(20):10448–56.
Tan CS, Koralnik IJ. Progressive multifocal leukoencephalopathy and other disorders caused by JC virus: clinical features and pathogenesis. Lancet Neurol. 2010;9(4):425–37.
Beck ES, Cortese I. Checkpoint inhibitors for the treatment of JC virus-related progressive multifocal leukoencephalopathy. Curr Opin Virol. 2020;40:19–27.
Lambert N, El Moussaoui M, Maquet P. Immune checkpoint inhibitors for progressive multifocal leukoencephalopathy: identifying relevant outcome factors. Eur J Neurol. 2021;28(11):3814–9.
Astrom KE, Mancall EL, Richardson EP Jr. Progressive multifocal leuko-encephalopathy; a hitherto unrecognized complication of chronic lymphatic leukaemia and Hodgkin’s disease. Brain. 1958;81(1):93–111.
Berger JR, Pall L, Lanska D, Whiteman M. Progressive multifocal leukoencephalopathy in patients with HIV infection. J Neurovirol. 1998;4(1):59–68.
Casado JL, Corral I, García J, Martinez-San Millán J, Navas E, Moreno A, et al. Continued declining incidence and improved survival of progressive multifocal leukoencephalopathy in HIV/AIDS patients in the current era. Eur J Clin Microbiol Infect Dis. 2014;33(2):179–87.
Engsig FN, Hansen AB, Omland LH, Kronborg G, Gerstoft J, Laursen AL, et al. Incidence, clinical presentation, and outcome of progressive multifocal leukoencephalopathy in HIV-infected patients during the highly active antiretroviral therapy era: a nationwide cohort study. J Infect Dis. 2009;199(1):77–83.
Antinori A, Cingolani A, Lorenzini P, Giancola ML, Uccella I, Bossolasco S, et al. Clinical epidemiology and survival of progressive multifocal leukoencephalopathy in the era of highly active antiretroviral therapy: data from the Italian Registry Investigative Neuro AIDS (IRINA). J Neurovirol. 2003;9(Suppl 1):47–53.
•• Summers NA, Kelley CF, Armstrong W, Marconi VC, Nguyen ML. Not a disease of the past: a case series of progressive multifocal leukoencephalopathy in the established antiretroviral era. AIDS Res Hum Retroviruses. 2019;35(6):544–52. (This is a case series that documents the continued impact of PML and PML-IRIS in people living with HIV.)
•• Anand P, Hotan GC, Vogel A, Venna N, Mateen FJ. Progressive multifocal leukoencephalopathy: a 25-year retrospective cohort study. Neurol Neuroimmunol Neuroinflamm. 2019;6(6). This is a hospital-based cohort study of PML that highlights shifts in the epidemiology of this disease.
Berger JR. Progressive multifocal leukoencephalopathy in acquired immunodeficiency syndrome: explaining the high incidence and disproportionate frequency of the illness relative to other immunosuppressive conditions. J Neurovirol. 2003;9(Suppl 1):38–41.
•• Graf LM, Rosenkranz SC, Hölzemer A, Hagel C, Goebell E, Jordan S, et al. Clinical presentation and disease course of 37 consecutive cases of progressive multifocal leukoencephalopathy (PML) at a German tertiary-care hospital: a retrospective observational study. Front Neurol. 2021;12: 632535. This is a hospital-based cohort study which describes the underlying risk factors, clinical course, and treatment strategies among 37 cases of PML.
Tada H, Rappaport J, Lashgari M, Amini S, Wong-Staal F, Khalili K. Trans-activation of the JC virus late promoter by the tat protein of type 1 human immunodeficiency virus in glial cells. Proc Natl Acad Sci U S A. 1990;87(9):3479–83.
Daniel DC, Kinoshita Y, Khan MA, Del Valle L, Khalili K, Rappaport J, et al. Internalization of exogenous human immunodeficiency virus-1 protein, Tat, by KG-1 oligodendroglioma cells followed by stimulation of DNA replication initiated at the JC virus origin. DNA Cell Biol. 2004;23(12):858–67.
Bagchi S, Yuan R, Engleman EG. Immune checkpoint inhibitors for the treatment of cancer: clinical impact and mechanisms of response and resistance. Annu Rev Pathol. 2021;16:223–49.
Lipson EJ, Drake CG. Ipilimumab: an anti-CTLA-4 antibody for metastatic melanoma. Clin Cancer Res. 2011;17(22):6958–62.
Garon EB, Hellmann MD, Rizvi NA, Carcereny E, Leighl NB, Ahn MJ, et al. Five-year overall survival for patients with advanced nonsmall-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 study. J Clin Oncol. 2019;37(28):2518–27.
Arora S, Velichinskii R, Lesh RW, Ali U, Kubiak M, Bansal P, et al. Existing and emerging biomarkers for immune checkpoint immunotherapy in solid tumors. Adv Ther. 2019;36(10):2638–78.
Havel JJ, Chowell D, Chan TA. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat Rev Cancer. 2019;19(3):133–50.
Blank C, Mackensen A. Contribution of the PD-L1/PD-1 pathway to T-cell exhaustion: an update on implications for chronic infections and tumor evasion. Cancer Immunol Immunother. 2007;56(5):739–45.
McLane LM, Abdel-Hakeem MS, Wherry EJ. CD8 T cell exhaustion during chronic viral infection and cancer. Annu Rev Immunol. 2019;37:457–95.
Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015;15(8):486–99.
Salmaninejad A, Valilou SF, Shabgah AG, Aslani S, Alimardani M, Pasdar A, et al. PD-1/PD-L1 pathway: basic biology and role in cancer immunotherapy. J Cell Physiol. 2019;234(10):16824–37.
Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature. 2006;439(7077):682–7.
Im SJ, Hashimoto M, Gerner MY, Lee J, Kissick HT, Burger MC, et al. Defining CD8+ T cells that provide the proliferative burst after PD-1 therapy. Nature. 2016;537(7620):417–21.
Utzschneider DT, Charmoy M, Chennupati V, Pousse L, Ferreira DP, Calderon-Copete S, et al. T Cell Factor 1-Expressing Memory-like CD8(+) T cells sustain the immune response to chronic viral infections. Immunity. 2016;45(2):415–27.
•• Blank CU, Haining WN, Held W, Hogan PG, Kallies A, Lugli E, et al. Defining ‘T cell exhaustion.’ Nat Rev Immunol. 2019;19(11):665–74. This review collects the viewpoints of experts in the field, providing a comprehensive discussion of definitions and biomakers of T cell exhaustion and the implications for treatments targeting the underlying pathways.
Sade-Feldman M, Yizhak K, Bjorgaard SL, Ray JP, de Boer CG, Jenkins RW, et al. Defining T cell states associated with response to checkpoint immunotherapy in melanoma. Cell. 2018;175(4):998-1013.e20.
Miller BC, Sen DR, Al Abosy R, Bi K, Virkud YV, LaFleur MW, et al. Subsets of exhausted CD8. Nat Immunol. 2019;20(3):326–36.
Castelli V, Lombardi A, Palomba E, Bozzi G, Ungaro R, Alagna L, et al. Immune checkpoint inhibitors in people living with HIV/AIDS: facts and controversies. Cells. 2021;10(9).
Coghill AE, Pfeiffer RM, Shiels MS, Engels EA. Excess Mortality among HIV-Infected Individuals with Cancer in the United States. Cancer Epidemiol Biomarkers Prev. 2017;26(7):1027–33.
Wang CC, Silverberg MJ, Abrams DI. Non-AIDS-defining malignancies in the HIV-infected population. Curr Infect Dis Rep. 2014;16(6):406.
Vora KB, Ricciuti B, Awad MM. Exclusion of patients living with HIV from cancer immune checkpoint inhibitor trials. Sci Rep. 2021;11(1):6637.
Burke MM, Kluger HM, Golden M, Heller KN, Hoos A, Sznol M. Case Report: response to ipilimumab in a patient with HIV with metastatic melanoma. J Clin Oncol. 2011;29(32):e792–4.
Cook MR, Kim C. Safety and efficacy of immune checkpoint inhibitor therapy in patients with HIV infection and advanced-stage cancer: a systematic review. JAMA Oncol. 2019;5(7):1049–54.
•• Uldrick TS, Goncalves PH, Abdul-Hay M, Claeys AJ, Emu B, Ernstoff MS, et al. Assessment of the safety of pembrolizumab in patients with HIV and advanced cancer-a phase 1 Study. JAMA Oncol. 2019;5(9):1332–9. This is an open-lable, phase 1 multicenter study assessing safety and tolerability of checkpoint blockade for the treatment of cancer in 30 participants with underlying HIV infection, demonstrating acceptable safety profile and clinical benefit.
Wang L, Yang Y, Yu J, Zhang S, Li X, Wu X, et al. Efficacy and safety of anti-PD-1/PD-L1 in combination with chemotherapy or not as first-line treatment for advanced non-small cell lung cancer: a systematic review and network meta-analysis. Thorac Cancer. 2021.
Administration” FaD. Cancer clinical trial eligibility criteria: patients with HIV, hepatitis B virus, or hepatitis C virus infections. Federal Register 2020.
Chun TW, Carruth L, Finzi D, Shen X, DiGiuseppe JA, Taylor H, et al. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature. 1997;387(6629):183–8.
Banga R, Procopio FA, Noto A, Pollakis G, Cavassini M, Ohmiti K, et al. PD-1(+) and follicular helper T cells are responsible for persistent HIV-1 transcription in treated aviremic individuals. Nat Med. 2016;22(7):754–61.
Fromentin R, Bakeman W, Lawani MB, Khoury G, Hartogensis W, DaFonseca S, et al. CD4+ T cells expressing PD-1, TIGIT and LAG-3 contribute to HIV persistence during ART. PLoS Pathog. 2016;12(7): e1005761.
Sun D, Guo SY, Yang L, Wang YR, Wei XH, Song S, et al. Silicone elastomer gel impregnated with 20(S)-protopanaxadiol-loaded nanostructured lipid carriers for ordered diabetic ulcer recovery. Acta Pharmacol Sin. 2020;41(1):119–28.
Trautmann L, Janbazian L, Chomont N, Said EA, Gimmig S, Bessette B, et al. Upregulation of PD-1 expression on HIV-specific CD8+ T cells leads to reversible immune dysfunction. Nat Med. 2006;12(10):1198–202.
Van der Sluis RM, Kumar NA, Pascoe RD, Zerbato JM, Evans VA, Dantanarayana AI, et al. Combination immune checkpoint blockade to reverse HIV latency. J Immunol. 2020;204(5):1242–54.
Fromentin R, DaFonseca S, Costiniuk CT, El-Far M, Procopio FA, Hecht FM, et al. PD-1 blockade potentiates HIV latency reversal ex vivo in CD4. Nat Commun. 2019;10(1):814.
Wightman F, Solomon A, Kumar SS, Urriola N, Gallagher K, Hiener B, et al. Effect of ipilimumab on the HIV reservoir in an HIV-infected individual with metastatic melanoma. AIDS. 2015;29(4):504–6.
•• Guihot A, Marcelin AG, Massiani MA, Samri A, Soulie C, Autran B, et al. Drastic decrease of the HIV reservoir in a patient treated with nivolumab for lung cancer. Ann Oncol. 2018;29(2):517–8. This case report is the first documentation of sustained depletion of the HIV reservoir following treatment with checkpoint blockade.
Scully EP, Rutishauser RL, Simoneau CR, Delagreverie H, Euler Z, Thanh C, et al. Inconsistent HIV reservoir dynamics and immune responses following anti-PD-1 therapy in cancer patients with HIV infection. Ann Oncol. 2018;29(10):2141–2.
Abbar B, Baron M, Katlama C, Marcelin AG, Veyri M, Autran B, et al. Immune checkpoint inhibitors in people living with HIV: what about anti-HIV effects? AIDS. 2020;34(2):167–75.
Uldrick TS, Adams SV, Fromentin R, Roche M, Fling SP, Gonçalves PH, et al. Pembrolizumab induces HIV latency reversal in people living with HIV and cancer on antiretroviral therapy. Sci Transl Med. 2022;14(629):eabl3836.
Baron M, Soulié C, Lavolé A, Assoumou L, Abbar B, Fouquet B, et al. Impact of anti PD-1 immunotherapy on HIV reservoir and anti-viral immune responses in people living with HIV and cancer. Cells. 2022;11(6).
•• Cortese I, Muranski P, Enose-Akahata Y, Ha SK, Smith B, Monaco M, et al. Pembrolizumab treatment for progressive multifocal leukoencephalopathy. N Engl J Med. 2019;380(17):1597–605. This is a case series of patients with PML with various underlying diseases (including two with HIV) treated with pembrolizumab.
Tan CS, Bord E, Broge TA Jr, Glotzbecker B, Mills H, Gheuens S, et al. Increased program cell death-1 expression on T lymphocytes of patients with progressive multifocal leukoencephalopathy. J Acquir Immune Defic Syndr. 2012;60(3):244–8.
Walter O, Treiner E, Bonneville F, Mengelle C, Vergez F, Lerebours F, et al. Treatment of Progressive Multifocal Leukoencephalopathy with Nivolumab. N Engl J Med. 2019;380(17):1674–6.
Rauer S, Marks R, Urbach H, Warnatz K, Nath A, Holland S, et al. Treatment of Progressive Multifocal Leukoencephalopathy with Pembrolizumab. N Engl J Med. 2019;380(17):1676–7.
Hoang E, Bartlett NL, Goyal MS, Schmidt RE, Clifford DB. Progressive multifocal leukoencephalopathy treated with nivolumab. J Neurovirol. 2019;25(2):284–7.
Medrano C, Vergez F, Mengelle C, Faguer S, Kamar N, Del Bello A. Effectiveness of immune checkpoint inhibitors in transplant recipients with progressive multifocal leukoencephalopathy. Emerg Infect Dis. 2019;25(11):2145–7.
Kupper C, Heinrich J, Kamm K, Bucklein V, Rothenfusser S, Straube A. Pembrolizumab for progressive multifocal leukoencephalopathy due to primary immunodeficiency. Neurol Neuroimmunol Neuroinflamm. 2019;6(6): e628.
Pawlitzki M, Schneider-Hohendorf T, Rolfes L, Meuth SG, Wiendl H, Schwab N, et al. Ineffective treatment of PML with pembrolizumab: exhausted memory T-cell subsets as a clue? Neurol Neuroimmunol Neuroinflamm. 2019;6(6): e627.
Audemard-Verger A, Gasnault J, Faisant M, Besse MC, Martin-Silva N, Berra M, et al. Sustained response and rationale of programmed cell death-1-targeting for progressive multifocal leukoencephalopathy. Open Forum Infect Dis. 2019;6(10):ofz374.
Holmes A, Wellings T, Walsh O, Rowlings P. Progressive multifocal leukoencephalopathy associated with a lymphoproliferative disorder treated with pembrolizumab. J Neurovirol. 2020;26(6):961–3.
Mahler C, Andrews M, Henson SM, Gnanapavan S. Sequential interleukin 2 and pembrolizumab use in progressive multifocal leukoencephalopathy. Neurol Neuroimmunol Neuroinflamm. 2020;7(4).
Goereci Y, Schweitzer F, Wellstein A, Silling S, Borchmann S, von Tresckow B, et al. Clearance of JC polyomavirus from cerebrospinal fluid following treatment with interleukin-2 and pembrolizumab in an individual with progressive multifocal leukoencephalopathy and no underlying immune deficiency syndrome. Eur J Neurol. 2020;27(11):2375–7.
Grassl N, Bunse L, Beutel T, Klockziem M, Gass A, Platten M, et al. Nivolumab for treatment of progressive multifocal leukoencephalopathy in Sezary syndrome. Eur J Neurol. 2020;27(11):2373–4.
Uzunov M, Demeret S, Nguyen-Quoc S, Morel V, Bellanger A, Chavez H, et al. Postallogeneic transplantation progressive multifocal leukoencephalopathy successfully treated by nivolumab. Br J Haematol. 2020;188(6):e82–4.
•• Roos-Weil D, Weiss N, Guihot A, Uzunov M, Bellanger A, Eymard B, et al. Immune checkpoint inhibitors for progressive multifocal leukoencephalopathy: a new gold standard? J Neurol. 2021;268(7):2458–65. This is a case report of patient with HIV-related PML treated with nivolumab.
Beudel M, Rovekamp F, van de Beek D, Brouwer M. Single-Dose pembrolizumab treatment for progressive multifocal leukoencephalopathy. Neurol Neuroimmunol Neuroinflamm. 2021;8(4).
Mohn N, Wattjes MP, Adams O, Nay S, Tkachenko D, Salge F, et al. PD-1-inhibitor pembrolizumab for treatment of progressive multifocal leukoencephalopathy. Ther Adv Neurol Disord. 2021;14:1756286421993684.
Fischbach L, Bauer T, Ruber T, Grobe-Einsler M, Sitter A, Radbruch A, et al. Treatment of progressive multifocal Leukoencephalopathy associated with idiopathic lymphocytopenia with Nivolumab. J Neurol Sci. 2021;427: 117503.
Lan TY, Chen YS, Cheng CF, Huang ST, Shen CY, Hsu PN. Progressive multifocal leukoencephalopathy in systemic lupus erythematosus managed with pembrolizumab: a case report with literature review. Lupus. 2021;30(11):1849–55.
Volk T, Warnatz K, Marks R, Urbach H, Schluh G, Strohmeier V, et al. Pembrolizumab for treatment of progressive multifocal leukoencephalopathy in primary immunodeficiency and/or hematologic malignancy: a case series of five patients. J Neurol. 2021.
Kapadia RK, Ney D. Stabilization of progressive multifocal leukoencephalopathy after pembrolizumab treatment. Neurohospitalist. 2020;10(3):238–9.
•• Mozo Ruiz M, Rosado Barrasa N, Tena Gómez D, Torralba González de Suso M. Pembrolizumab treatment for progressive multifocal leukoencephalopathy in a patient with Human Immunodeficiency Virus infection. Enferm Infecc Microbiol Clin (Engl Ed). 2020;38(8):396–7. This is a case report of a patient with HIV-related PML treated with pembrolizumab.
Patel A, Sul J, Gordon ML, Steinklein J, Sanguinetti S, Pramanik B, et al. Progressive multifocal leukoencephalopathy in a patient with progressive multiple sclerosis treated with ocrelizumab monotherapy. JAMA Neurol. 2021;78(6):736–40.
Haroche J, Cohen Aubart F, Mokhtari K, Seilhean D, Benameur N, Peyre M, et al. Immune reconstitution syndrome after nivolumab for progressive multifocal leukoencephalopathy. Neurol Clin Pract. 2021;11(3):e376–8.
Wicklein R, Heidegger S, Verbeek M, Eiz-Vesper B, Maecker-Kolhoff B, Kirschke JS, et al. Combined treatment with pembrolizumab and allogenic BK virus-specific T cells in progressive multifocal leukoencephalopathy: a case report. Neurol Neuroimmunol Neuroinflamm. 2021;8(5).
Nitsch L, Kaps V, Zschernack V, Gancarczyk N, van Essen F, Schmeel C, et al. [Immune checkpoint inhibitors in the treatment of progressive multifocal leukoencephalopathy]. Nervenarzt. 2021.
Darcy S, Alexander M, McCarthy A, O'Dowd S. Pembrolizumab treatment of inflammatory progressive multifocal leukoencephalopathy: a report of two cases. J Neurovirol. 2021.
•• Sim BZ, Smith LK, Yarwood T, Smith S. Successful treatment of HIV-associated progressive multifocal leukoencephalopathy with pembrolizumab. AIDS. 2022;36(3):483–5. This is a case report of HIV-related PML treated with pembrolizumab.
Boesl F, Allers K, Herm J, Scheider T, Franke C. Sequential treatment of progressive multifocal leukoencephalopathy with intravenous immunoglobulins and pembrolizumab. J Neurovirol. 2022.
Lambert N, Dauby S, Dive D, Sadzot B, Maquet P. Atezolizumab Treatment for progressive multifocal leukoencephalopathy. Emerg Infect Dis. 2022;28(1).
Mateen FJ, Muralidharan R, Carone M, van de Beek D, Harrison DM, Aksamit AJ, et al. Progressive multifocal leukoencephalopathy in transplant recipients. Ann Neurol. 2011;70(2):305–22.
Cardinale A, De Luca CD, Locatelli F, Velardi E. Thymic function and T-cell receptor repertoire diversity: implications for patient response to checkpoint blockade immunotherapy. Front Immunol. 2021;12: 752042.
Fanjul F, Riveiro-Barciela M, Gonzalez J, Delgado E, Murillas J, PayerasCifré A, et al. Evaluation of progressive multifocal leukoencephalopathy treatments in a Spanish cohort of HIV-infected patients: do protease inhibitors improve survival regardless of central nervous system penetration-effectiveness (CPE) score? HIV Med. 2013;14(5):321–5.
Garvey L, Winston A, Walsh J, Post F, Porter K, Gazzard B, et al. Antiretroviral therapy CNS penetration and HIV-1-associated CNS disease. Neurology. 2011;76(8):693–700.
Lanoy E, Guiguet M, Bentata M, Rouveix E, Dhiver C, Poizot-Martin I, et al. Survival after neuroAIDS: association with antiretroviral CNS Penetration-Effectiveness score. Neurology. 2011;76(7):644–51.
Fournier A, Martin-Blondel G, Lechapt-Zalcman E, Dina J, Kazemi A, Verdon R, et al. Immune reconstitution inflammatory syndrome unmasking or worsening AIDS-related progressive multifocal leukoencephalopathy: a literature review. Front Immunol. 2017;8:577.
Thapa S, Shrestha U. Immune Reconstitution inflammatory syndrome. Treasure Island: StatPearls; 2022.
Kartau M, Sipila JO, Auvinen E, Palomaki M, Verkkoniemi-Ahola A. Progressive multifocal leukoencephalopathy: current insights. Degener Neurol Neuromuscul Dis. 2019;9:109–21.
Tan K, Roda R, Ostrow L, McArthur J, Nath A. PML-IRIS in patients with HIV infection: clinical manifestations and treatment with steroids. Neurology. 2009;72(17):1458–64.
Shelburne SA, Visnegarwala F, Darcourt J, Graviss EA, Giordano TP, White AC Jr, et al. Incidence and risk factors for immune reconstitution inflammatory syndrome during highly active antiretroviral therapy. AIDS. 2005;19(4):399–406.
Antoniol C, Jilek S, Schluep M, Mercier N, Canales M, Le Goff G, et al. Impairment of JCV-specific T-cell response by corticotherapy: effect on PML-IRIS management? Neurology. 2012;79(23):2258–64.
Panel on Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV. Guidelines for the Prevention and Treatment of Opportunistic Infections in HIV-infected Adults and Adolescents: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America [Available from: https://clinicalinfo.hiv.gov/en/guidelines/adult-and-adolescent-opportunistic-infection.
Nagai H, Muto M. Optimal management of immune-related adverse events resulting from treatment with immune checkpoint inhibitors: a review and update. Int J Clin Oncol. 2018;23(3):410–20.
Martinot M, Ahle G, Petrosyan I, Martinez C, Gorun DM, Mohseni-Zadeh M, et al. Progressive multifocal leukoencephalopathy after treatment with nivolumab. Emerg Infect Dis. 2018;24(8):1594–6.
Neil EC, DeAngelis LM. Progressive multifocal leukoencephalopathy and hematologic malignancies: a single cancer center retrospective review. Blood Adv. 2017;1(23):2041–5.
Funding
This work is supported by the NINDS Intramural Research Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Sydney Corey and Bryan Smith declare that they have no conflict of interest.
Irene Cortese is a shareholder of Nouscom AG, Keires AG and PDC line*pharma, outside the submitted work.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Central Nervous System and Cognition
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Corey, S., Smith, B.R. & Cortese, I.C.M. Promise and Challenges of Checkpoint Inhibitor Therapy for Progressive Multifocal Leukoencephalopathy in HIV. Curr HIV/AIDS Rep 19, 580–591 (2022). https://doi.org/10.1007/s11904-022-00626-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11904-022-00626-w