Abstract
Purpose of Review
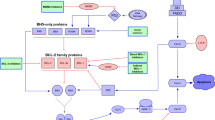
While the treatment of acute lymphoblastic leukemia (ALL) has improved significantly over the last 30 years, the majority of adult patients will have their disease relapse. The BCL-2 gene was initially discovered from follicular lymphoma research; however, the BH3 family of proteins has is emerging to be crucial in patients with ALL due to their reliance on the balance of these pro-apoptotic and anti-apoptotic proteins in the BH3 family. We discuss apoptosis in ALL, the reliance mechanisms, drug development in this space, and areas for future research.
Recent Findings
The first drugs that were developed to inhibit the BCL-2 pathway include both venetoclax (BCL-2 specific inhibitor) and navitoclax (BCL-2, BCL-XL, and BCL-W). These drugs show promise and have obtained complete remissions, minimal residual disease negative status, and have been used as a bridge to allogeneic hematopoietic stem cell transplantation in acute myeloid leukemia and chronic lymphocytic leukemia. There are multiple ongoing clinical trials looking to assess the use of BCL-2 inhibition with chemotherapy, targeted therapies, and bi-specific T-cell engager therapies not only in both frontline and relapsed refractory ALL but also in consolidation and maintenance phases.
Summary
There is still a large need for improvement of ALL outcomes in adult patients. Research has shown that ALL depends on the BCL-2 family of proteins for cell survival and proliferation. Targeting this pathway with BCL-2 inhibition has led to encouraging results, and future research is aimed at incorporating this targeted therapy into current treatment paradigms.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Terwilliger T, Abdul-Hay M. Acute lymphoblastic leukemia: a comprehensive review and 2017 update. Blood Cancer J. 2017;7(6):e577. https://doi.org/10.1038/bcj.2017.53 (Published 2017 Jun 30).
Kantarjian H, Stein A, Gökbuget N, et al. Blinatumomab versus chemotherapy for advanced acute lymphoblastic leukemia. N Engl J Med. 2017;376(9):836–47. https://doi.org/10.1056/NEJMoa1609783.
Gökbuget N, Dombret H, Bonifacio M, et al. Blinatumomab for minimal residual disease in adults with B-cell precursor acute lymphoblastic leukemia [published correction appears in blood. 2019 Jun 13;133(24):2625]. Blood. 2018;131(14):1522–31. https://doi.org/10.1182/blood-2017-08-798322.
Kantarjian HM, DeAngelo DJ, Stelljes M, et al. Inotuzumab ozogamicin versus standard therapy for acute lymphoblastic leukemia. N Engl J Med. 2016;375(8):740–53. https://doi.org/10.1056/NEJMoa1509277.
Shah BD, Ghobadi A, Oluwole OO, et al. KTE-X19 for relapsed or refractory adult B-cell acute lymphoblastic leukaemia: phase 2 results of the single-arm, open-label, multicentre ZUMA-3 study. Lancet. 2021;398(10299):491–502. https://doi.org/10.1016/S0140-6736(21)01222-8.
Martinelli G, Boissel N, Chevallier P, et al. Complete hematologic and molecular response in adult patients with relapsed/refractory Philadelphia chromosome-positive B-precursor acute lymphoblastic leukemia following treatment with blinatumomab: results from a phase II, single-arm, multicenter study [published correction appears in J Clin Oncol. 2017 Aug 10;35(23):2722] [published correction appears in J Clin Oncol. 2017 Aug 20;35(24):2856]. J Clin Oncol. 2017;35(16):1795–802. https://doi.org/10.1200/JCO.2016.69.3531.
Maude SL, Laetsch TW, Buechner J, et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med. 2018;378(5):439–48. https://doi.org/10.1056/NEJMoa1709866.
DiNardo CD, Jonas BA, Pullarkat V, et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383(7):617–29. https://doi.org/10.1056/NEJMoa2012971.
Fischer K, Al-Sawaf O, Bahlo J, et al. Venetoclax and obinutuzumab in patients with CLL and coexisting conditions. N Engl J Med. 2019;380(23):2225–36. https://doi.org/10.1056/NEJMoa1815281.
Seymour JF, Kipps TJ, Eichhorst B, et al. Venetoclax-rituximab in relapsed or refractory chronic lymphocytic leukemia. N Engl J Med. 2018;378(12):1107–20. https://doi.org/10.1056/NEJMoa1713976.
Wei AH, Strickland SA Jr, Hou JZ, et al. Venetoclax combined with low-dose cytarabine for previously untreated patients with acute myeloid leukemia: results from a phase Ib/II study. J Clin Oncol. 2019;37(15):1277–84. https://doi.org/10.1200/JCO.18.01600.
Geng H, Brennan S, Milne TA, et al. Integrative epigenomic analysis identifies biomarkers and therapeutic targets in adult B-acute lymphoblastic leukemia. Cancer Discov. 2012;2(11):1004–23. https://doi.org/10.1158/2159-8290.CD-12-0208.
Alford SE, Kothari A, Loeff FC, et al. BH3 inhibitor sensitivity and Bcl-2 dependence in primary acute lymphoblastic leukemia cells. Cancer Res. 2015;75(7):1366–75. https://doi.org/10.1158/0008-5472.CAN-14-1849.
Frismantas V, Dobay MP, Rinaldi A, et al. Ex vivo drug response profiling detects recurrent sensitivity patterns in drug-resistant acute lymphoblastic leukemia. Blood. 2017;129(11):e26–37. https://doi.org/10.1182/blood-2016-09-738070.
• Pullarkat VA, Lacayo NJ, Jabbour E, et al. Venetoclax and navitoclax in combination with chemotherapy in patients with relapsed or refractory acute lymphoblastic leukemia and lymphoblastic lymphoma. Cancer Discov. 2021;11(6):1440–53. https://doi.org/10.1158/2159-8290.CD-20-1465. (Phase 1 trial with combination of venetoclax and navitoclax that shows strong response in patients with ALL.)
Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9(1):47–59. https://doi.org/10.1038/nrm2308.
Tsujimoto Y, Cossman J, Jaffe E, Croce CM. Involvement of the bcl-2 gene in human follicular lymphoma. Science. 1985;228(4706):1440–3. https://doi.org/10.1126/science.3874430.
Lutz RJ. Role of the BH3 (Bcl-2 homology 3) domain in the regulation of apoptosis and Bcl-2-related proteins. Biochem Soc Trans. 2000;28(2):51–6. https://doi.org/10.1042/bst0280051.
Seyfried F, Stirnweiß FU, Niedermayer A, et al. Synergistic activity of combined inhibition of anti-apoptotic molecules in B-cell precursor ALL [published online ahead of print, 2022 Jan 14]. Leukemia. 2022. doi:https://doi.org/10.1038/s41375-021-01502-z
Czabotar PE, Lessene G, Strasser A, Adams JM. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol. 2014;15(1):49–63. https://doi.org/10.1038/nrm3722.
Kotschy A, Szlavik Z, Murray J, et al. The MCL1 inhibitor S63845 is tolerable and effective in diverse cancer models. Nature. 2016;538(7626):477–82. https://doi.org/10.1038/nature19830.
Bojarczuk K, Sasi BK, Gobessi S, et al. BCR signaling inhibitors differ in their ability to overcome Mcl-1-mediated resistance of CLL B cells to ABT-199. Blood. 2016;127(25):3192–201. https://doi.org/10.1182/blood-2015-10-675009.
Choudhary GS, Al-Harbi S, Mazumder S, et al. MCL-1 and BCL-xL-dependent resistance to the BCL-2 inhibitor ABT-199 can be overcome by preventing PI3K/AKT/mTOR activation in lymphoid malignancies. Cell Death Dis. 2015;6(1):e1593. https://doi.org/10.1038/cddis.2014.525 (Published 2015 Jan 15).
Li Z, He S, Look AT. The MCL1-specific inhibitor S63845 acts synergistically with venetoclax/ABT-199 to induce apoptosis in T-cell acute lymphoblastic leukemia cells. Leukemia. 2019;33(1):262–6. https://doi.org/10.1038/s41375-018-0201-2.
Jain N, Lamb AV, O’Brien S, et al. Early T-cell precursor acute lymphoblastic leukemia/lymphoma (ETP-ALL/LBL) in adolescents and adults: a high-risk subtype. Blood. 2016;127(15):1863–9. https://doi.org/10.1182/blood-2015-08-661702.
Coustan-Smith E, Mullighan CG, Onciu M, et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol. 2009;10(2):147–56. https://doi.org/10.1016/S1470-2045(08)70314-0.
Morita K, Jain N, Kantarjian H, et al. Outcome of T-cell acute lymphoblastic leukemia/lymphoma: focus on near-ETP phenotype and differential impact of nelarabine. Am J Hematol. 2021;96(5):589–98. https://doi.org/10.1002/ajh.26144.
Del Gaizo MV, Schlis KD, Sallan SE, Armstrong SA, Letai A. BCL-2 dependence and ABT-737 sensitivity in acute lymphoblastic leukemia. Blood. 2008;111(4):2300–9. https://doi.org/10.1182/blood-2007-06-098012.
Certo M, Del Gaizo MV, Nishino M, et al. Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell. 2006;9(5):351–65. https://doi.org/10.1016/j.ccr.2006.03.027.
Vo TT, Ryan J, Carrasco R, et al. Relative mitochondrial priming of myeloblasts and normal HSCs determines chemotherapeutic success in AML. Cell. 2012;151(2):344–55. https://doi.org/10.1016/j.cell.2012.08.038.
Ni Chonghaile T, Sarosiek KA, Vo TT, et al. Pretreatment mitochondrial priming correlates with clinical response to cytotoxic chemotherapy. Science. 2011;334(6059):1129–33. https://doi.org/10.1126/science.1206727.
Chonghaile TN, Roderick JE, Glenfield C, et al. Maturation stage of T-cell acute lymphoblastic leukemia determines BCL-2 versus BCL-XL dependence and sensitivity to ABT-199. Cancer Discov. 2014;4(9):1074–87. https://doi.org/10.1158/2159-8290.CD-14-0353.
Lin KH, Winter PS, Xie A, et al. Targeting MCL-1/BCL-XL forestalls the acquisition of resistance to ABT-199 in acute myeloid leukemia. Sci Rep. 2016;6:27696. https://doi.org/10.1038/srep27696 (Published 2016 Jun 10).
Hormi M, Birsen R, Belhadj M, et al. Pairing MCL-1 inhibition with venetoclax improves therapeutic efficiency of BH3-mimetics in AML. Eur J Haematol. 2020;105(5):588–96. https://doi.org/10.1111/ejh.13492.
Wang H, Guo M, Wei H, Chen Y. Targeting MCL-1 in cancer: current status and perspectives. J Hematol Oncol. 2021;14(1):67. https://doi.org/10.1186/s13045-021-01079-1 (Published 2021 Apr 21).
Tse C, Shoemaker AR, Adickes J, et al. ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res. 2008;68(9):3421–8. https://doi.org/10.1158/0008-5472.CAN-07-5836.
Shoemaker AR, Mitten MJ, Adickes J, et al. Activity of the Bcl-2 family inhibitor ABT-263 in a panel of small cell lung cancer xenograft models. Clin Cancer Res. 2008;14(11):3268–77. https://doi.org/10.1158/1078-0432.CCR-07-4622.
Wilson WH, O’Connor OA, Czuczman MS, et al. Navitoclax, a targeted high-affinity inhibitor of BCL-2, in lymphoid malignancies: a phase 1 dose-escalation study of safety, pharmacokinetics, pharmacodynamics, and antitumour activity. Lancet Oncol. 2010;11(12):1149–59. https://doi.org/10.1016/S1470-2045(10)70261-8.
Souers AJ, Leverson JD, Boghaert ER, et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med. 2013;19(2):202–8. https://doi.org/10.1038/nm.3048.
Roberts AW, Davids MS, Pagel JM, et al. Targeting BCL2 with venetoclax in relapsed chronic lymphocytic leukemia. N Engl J Med. 2016;374(4):311–22. https://doi.org/10.1056/NEJMoa1513257.
Jain N, et al. A multicenter phase I study combining venetoclax with mini-hyper-cvd in older adults with untreated and relapsed/refractory acute lymphoblastic leukemia. Blood. 2019;134:3867.
Kantarjian H, Ravandi F, Short NJ, et al. Inotuzumab ozogamicin in combination with low-intensity chemotherapy for older patients with Philadelphia chromosome-negative acute lymphoblastic leukaemia: a single-arm, phase 2 study. Lancet Oncol. 2018;19(2):240–8. https://doi.org/10.1016/S1470-2045(18)30011-1.
• Venugopal S, et al. A phase II study of mini-hyper-CVD plus venetoclax in patients with Philadelphia chromosome-negative acute lymphoblastic leukemia. Blood. 2021;138:1239. (Phase 2 study adding venetoclax to lower intensity chemotherapy backbone shows safety and efficacy in Ph-negative ALL.)
Zhang Y, Qian JJ, Shen YJ, Hang SJ, Jin J, Zhu HH. The first report of complete remission following treatment with venetoclax plus prednisone in elderly patients with Philadelphia chromosome-negative acute lymphoblastic leukemia [published online ahead of print, 2021 Oct 18]. Ann Hematol. 2021. doi:https://doi.org/10.1007/s00277-021-04699-2
Luskin MR, DeAngelo DJ. Mini-hyper-CVD combinations for older adults: results of recent trials and a glimpse into the future. Clin Lymphoma Myeloma Leuk. 2020;20(Suppl 1):S44–7. https://doi.org/10.1016/S2152-2650(20)30458-4.
• Short NJ, Konopleva M, Kadia T, et al. An effective chemotherapy-free regimen of ponatinib plus venetoclax for relapsed/refractory Philadelphia chromosome-positive acute lymphoblastic leukemia. Am J Hematol. 2021;96(7):E229–32. https://doi.org/10.1002/ajh.26175. (Chemotherapy-free regimen of ponatinib and venetoclax demonstrates good effect in patients with Ph-positive ALL.)
Wang H, Yang C, Shi T, et al. Venetoclax-ponatinib for T315I/compound-mutated Ph+ acute lymphoblastic leukemia. Blood Cancer J. 2022;12(1):20. https://doi.org/10.1038/s41408-022-00621-9 (Published 2022 Jan 28).
Palmisiano N, et al. Maximal tolerated dose determined for venetoclax in combination with liposomal vincristine in patients with relapsed or refractory Ph-negative T-cell or B-cell acute lymphoblastic leukemia: results of phase 1 portion of ECOG-ACRIN EA9152. Blood. 2021;138:3407.
Rahmat LT, Nguyen A, Abdulhaq H, Prakash S, Logan AC, Mannis GN. Venetoclax in combination with decitabine for relapsed T-cell acute lymphoblastic leukemia after allogeneic hematopoietic cell transplant. Case Rep Hematol. 2018;2018:6092646. https://doi.org/10.1155/2018/6092646 (Published 2018 Aug 26).
Farhadfar N, Li Y, May WS, Adams CB. Venetoclax and decitabine for treatment of relapsed T-cell acute lymphoblastic leukemia: a case report and review of literature. Hematol Oncol Stem Cell Ther. 2021;14(3):246–51. https://doi.org/10.1016/j.hemonc.2019.10.002.
Wan CL, Zou JY, Qiao M, et al. Venetoclax combined with azacitidine as an effective and safe salvage regimen for relapsed or refractory T-cell acute lymphoblastic leukemia: a case series. Leuk Lymphoma. 2021;62(13):3300–3. https://doi.org/10.1080/10428194.2021.1957876.
El-Cheikh J, Moukalled NM, El Darsa H, et al. Feasibility of the combination of venetoclax and asparaginase-based chemotherapy for adult patients with relapsed/refractory acute lymphoblastic leukemia. Clin Lymphoma Myeloma Leuk. 2018;18(10):e441–4. https://doi.org/10.1016/j.clml.2018.07.289.
Richard-Carpentier G, Jabbour E, Short NJ, et al. Clinical experience with venetoclax combined with chemotherapy for relapsed or refractory T-cell acute lymphoblastic leukemia. Clin Lymphoma Myeloma Leuk. 2020;20(4):212–8. https://doi.org/10.1016/j.clml.2019.09.608.
Numan Y, Alfayez M, Maiti A, et al. First report of clinical response to venetoclax in early T-cell precursor acute lymphoblastic leukemia. JCO Precis Oncol. 2018;2:PO1800127. https://doi.org/10.1200/PO.18.00127.
Zhang X, Li J, Jin J, Yu W. Relapsed/refractory early T-cell precursor acute lymphoblastic leukemia was salvaged by venetoclax plus HAG regimen. Ann Hematol. 2020;99(2):395–7. https://doi.org/10.1007/s00277-019-03902-9.
Zappone E, Cencini E, Defina M, et al. Venetoclax in association with decitabine as effective bridge to transplant in a case of relapsed early T-cell lymphoblastic leukemia. Clin Case Rep. 2020;8(10):2000–2. https://doi.org/10.1002/ccr3.3041 (Published 2020 Jul 7).
Kong J, Chen N, Li M, et al. Venetoclax and decitabine in refractory TP53-mutated early T-cell precursor acute lymphoblastic leukemia. Ann Hematol. 2022;101(3):697–9. https://doi.org/10.1007/s00277-021-04530-y.
Arora S, et al. Venetoclax with chemotherapy in relapse/refractory early T-cell precursor acute lymphoblastic leukemia. Leuk Lymphoma. 2021;62(9):2292–4.
Campana D. Minimal residual disease in acute lymphoblastic leukemia. Semin Hematol. 2009;46(1):100–6. https://doi.org/10.1053/j.seminhematol.2008.09.001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Acute Lymphocytic Leukemias
Rights and permissions
About this article
Cite this article
Smith, W.M., Reed, D.R. Targeting Apoptosis in ALL. Curr Hematol Malig Rep 17, 53–60 (2022). https://doi.org/10.1007/s11899-022-00661-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11899-022-00661-9