Abstract
Background
Subarachnoid haemorrhage (SAH) clinical decision rules can provide successful results in the differential diagnosis of non-traumatic headache.
Aim
The aim of this study is to investigate whether a laboratory parameter that can be added to clinical decision-making rules can better predict subarachnoid haemorrhages in patients presenting to the emergency department with headache.
Methods
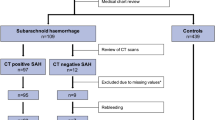
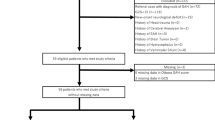
We conducted a retrospective cohort study between March 2017 and March 2019. Patients over the age of 18 who admitted to the emergency department with non-traumatic, acute headache last 14 days before admission and evaluated by imaging and laboratory studies were included in the study.
Results
A total of 867 patients were included and 141 of them had SAH. In detecting the SAH, Ottawa SAH rule sensitivity (85.1%), specificity (47.1%), positive predictive value (PPV) (23.8%) and negative predictive value (NPV) (94.2%), and for EMERALD SAH, rule sensitivity (96.4%), specificity (43.2%), PPV (24.8%) and NPV (98.4%). In the regression analysis, statistically significant result was obtained to exclude the diagnosis of SAH with the decrease in the eosinophil value (cutoff < 0.085 10^3/µL, OR: 0.011, 95% CI: 0.001–0.213, p = 0.003). When eosinophil value was added to EMERALD SAH rule, it provided a 100% of sensitivity, a 38.4% of specificity, a 24% of PPV and a 100% of NPV in detecting the SAH.
Conclusions
EMERALD SAH rule plus eosinophil, which offers 100% sensitivity and NPV for predicting SAH in adult non-traumatic headaches, may be recommended as a successful and practical decision rule for clinical use according to the Ottawa and EMERALD SAH rule.


Similar content being viewed by others
References
Centers for Disease Control and Prevention (US) (2016) National hospital ambulatory medical care survey: 2016 emergency department summary tables. Available at https://www.cdc.gov/nchs/data/nhamcs/web_tables/2016_ed_web_tables.pdf
Koyfman A, Long B (2020) Headache. In: Tintinalli JE, Ma OJ, Yealy DM, Meckler GD, Stapczynski JS, Cline DM, Thomas SH (eds) Tintinalli’s emergency medicine: a comprehensive study guide, 9th edn. McGraw-Hill, New York, pp 1107–1114
Russi CS, Headache WL (2018) In: Walls RM, Hockberger RS, Gausche-Hill M et al (eds) Rosen’s emergency medicine: concepts and clinical practıce, 9th edn. Elsevier, Philadelphia, pp 153–159
Singer RJ, Ogilvy CS, Rordorf G (2021) Aneurysmal subarachnoid hemorrhage: clinical manifestations and diagnosis. In: Goddeau RP, Biller J, Rabinstein AA, Edlow JA (eds) UpToDate. Available at https://www.uptodate.com/contents/aneurysmal-subarachnoid-hemorrhage-clinical-manifestations-and-diagnosis
Singer RJ, Ogilvy CS, Rordorf G (2022) Aneurysmal subarachnoid hemorrhage: epidemiology, risk factors, and pathogenesis. In: Goddeau RP, Biller J (ed) UpToDate. Available at https://www.uptodate.com/contents/aneurysmal-subarachnoid-hemorrhage-epidemiology-risk-factors-and-pathogenesis
Connolly ES Jr, Rabinstein AA, Carhuapoma JR et al (2012) Guidelines for the management of aneurysmal subarachnoid hemorrhage. A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 43:1711–1737. https://doi.org/10.1161/STR.0b013e3182587839
Kimura A, Kobayashi K, Yamaguchi H et al (2016) New clinical decision rule to exclude subarachnoid haemorrhage for acute headache: a prospective multicentre observational study. BMJ Open 6:e010999. https://doi.org/10.1136/bmjopen-2015-010999
Stiell IG, Wells GA (1999) Methodologic standards for the development of clinical decision rules in emergency medicine. Ann Emerg Med 33:437–447
Chu KH, Keijzers G, Furyk JS et al (2018) Applying the Ottawa subarachnoid haemorrhage rule on a cohort of emergency department patients with headache. Eur J Emerg Med 25(6):e29–e32. https://doi.org/10.1097/MEJ.0000000000000523
Etminan N, Chang HS, Hackenberg K et al (2019) Worldwide incidence of aneurysmal subarachnoid hemorrhage according to region, time period, blood pressure, and smoking prevalence in the population: a systematic review and meta-analysis. JAMA Neurol 76(5):588–597. https://doi.org/10.1001/jamaneurol.2019.0006
Sengul S, Akpolat T, Erdem Y et al (2016) Changes in hypertension prevalence, awareness, treatment, and control rates in Turkey from 2003 to 2012. J Hypertens 34:1208–1217. https://doi.org/10.1097/HJH.0000000000000901
Williams B, Mancia G, Spiering W et al (2018) 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur Heart J 39(33):3021–3104. https://doi.org/10.1093/eurheartj/ehy339
Ginsberg BH (2009) Factors affecting blood glucose monitoring: sources of errors in measurement. J Diabetes Sci Technol 3(4):903–913. https://doi.org/10.1177/193229680900300438
Asirvatham JR, Moses V, Bjornson L (2013) Errors in potassium measurement: a laboratory perspective for the clinician. N Am J Med Sci 5(4):255–259. https://doi.org/10.4103/1947-2714.110426
Lee TS, Kim J, Uh Y et al (2017) Correction equation for estimation of actual potassium concentration in hemolyzed specimen. Clin Lab 63(2):271–275. https://doi.org/10.7754/Clin.Lab.2016.160733
Gumz ML, Rabinowitz L, Wingo CS (2015) An integrated view of potassium homeostasis. N Engl J Med 373(1):60–72. https://doi.org/10.1056/NEJMra1313341
Ybanez N, Agrawal V, Tranmer BI et al (2014) Severe hypokalemia in a patient with subarachnoid hemorrhage. Am J Kidney Dis 63(3):530–535. https://doi.org/10.1053/j.ajkd.2013.07.005
Chen I, Mitchell P (2016) Serum potassium and sodium levels after subarachnoid haemorrhage. Br J Neurosurg 30(5):554–559. https://doi.org/10.1080/02688697.2016.1181151
van Donkelaar CE, Dijkland SA, van den Bergh WM et al (2016) Early circulating lactate and glucose levels after aneurysmal subarachnoid hemorrhage correlate with poor outcome and delayed cerebral ıschemia: a two-center cohort study. Copyright © 2016 by the Society of Critical Care Medicine and Wolters Kluwer Health, Inc. All Rights Reserved. https://doi.org/10.1097/CCM.0000000000001569
Kruyt ND, Biessels GJ, DeVries JH et al (2010) Hyperglycemia in aneurysmal subarachnoid hemorrhage: a potentially modifiable risk factor for poor outcome. J Cereb Blood Flow Metab 30:1577–1587. https://doi.org/10.1038/jcbfm.2010.102
Hemphill JC 3rd, Greenberg SM, Anderson CS et al (2015) Guidelines for the management of spontaneous intracerebral hemorrhage. A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 46:2032–2060. https://doi.org/10.1161/STR.0000000000000069
Cho WS, Kim JE, Park SQ et al (2018) Korean clinical practice guidelines for aneurysmal subarachnoid hemorrhage. J Korean Neurosurg Soc 61(2):127–166. https://doi.org/10.3340/jkns.2017.0404.005
Hansen KB, Vilsbøll T, Bagger JI et al (2011) Increased postprandial GIP and glucagon responses, but unaltered GLP-1 response after intervention with steroid hormone, relative physical inactivity, and high-calorie diet in healthy subjects. J Clin Endocrinol Metab 96(2):447–453. https://doi.org/10.1210/jc.2010-1605
Bolayir A, Cigdem B, Gokce SF et al (2017) The effect of eosinopenia on mortality in patients with intracerebral hemorrhage. J Stroke Cerebrovasc Dis 26(10):2248–2255. https://doi.org/10.1016/j.jstrokecerebrovasdis.2017.05.007
Hori YS, Kodera S, Sato Y et al (2016) Eosinopenia as a predictive factor of the short-term risk of mortality and ınfection after acute cerebral ınfarction. J Stroke Cerebrovasc Dis 25(6):1307–1312. https://doi.org/10.1016/j.jstrokecerebrovasdis.2015.12.007
Zhao HM, Qin WQ, Wang PJ et al (2019) Eosinopenia is a predictive factor for the severity of acute ischemic stroke. Neural Regen Res 14(10):1772–1779. https://doi.org/10.4103/1673-5374.258411
Acknowledgements
All authors thank Nazif Calis, for his assistance with statistical analyses.
Author information
Authors and Affiliations
Contributions
AK was the coordination researcher of the study and was concerned with study design, implementation, study monitoring, data collection and interpretation. SS was involved in study design, study monitoring and supervision and interpretation of data. MG, SA and KAA were researchers and were involved in data collection and interpretation. HD, MG and SA was involved in study design, statistical analysis and data interpretation. All authors reviewed the drafts and approved the final version of the article for submission.
Corresponding author
Ethics declarations
Ethics approval
Before starting the study, an ethical approval was obtained from the Ethics Committee of the Faculty of Medicine, dated 08.03.2019 and numbered 86.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kaya, A., Satar, S., Gulen, M. et al. Adding eosinophil count to EMERALD rules predicts subarachnoid haemorrhage better in emergency department. Ir J Med Sci 192, 1453–1462 (2023). https://doi.org/10.1007/s11845-022-03106-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-022-03106-7