Abstract
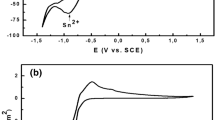
The oxidation of pure tin and Sn-0.7Cu, Sn-3.5Ag, Sn-1Zn, and Sn-9Zn alloys at 150°C was investigated. Both the chemical nature and the amount of oxides were characterized using electrochemical reduction analysis by measuring the electrolytic reduction potential and total transferred electrical charges. X-ray photoelectron spectroscopy was also conducted to support the results of reduction analysis. The effect of copper, silver, and zinc addition on surface oxidation of tin alloys is reported. For tin, Sn-0.7Cu, and Sn-3.5Ag, SnO grew first and then the mixture of SnO and SnO2 was found. SnO2 grew predominantly during long-time aging. For zinc-containing tin alloys, both ZnO and SnO2 were formed. Zinc promotes the formation of SnO2.
Similar content being viewed by others
References
A.T. Fromhold, Jr., Theory of Metal Oxidation, Vol. 1 Fundamentals (Burlington, MA: Elsevier [North-Holland Publishing Company], 1976).
Marcel Pourbaix, Atlas of Electrochemical Equilibria in Aqueous Solutions (Burlington, MA: Elsevier [Pergamon Press], 1966).
Frederick G. Yost, F. Michael Hosking, and Darrel R. Frear, The Mechanics of Solder Alloy, Wetting & Spreading (New York: Van Nostrand Reinhold, 1993), Chapter 6 (pp. 155–189).
D. Morgan Tench, D.P. Anderson, and P. Kim, “Solderability Assessment via Sequential Electrochemical Reduction Analysis,” J. Appl. Electrochem., 24 (1994), p. 18.
Rao R. Tummala et al., Fundamentals of Microsystems Packaging (New York: McGraw-Hill, 2001).
D.A. Sluzewski, Y.A. Chang, and V.C. Marcotte, “Oxidation of Pb-Sn and Pb-Sn-In Alloys: Bulk vs. Grain Boundary Regions,” Mat. Res. Soc. Symp. Proc., 167 (1990), p. 353.
R.J. Bird, “Corrosion-Resistant Lead-Indium and Lead-Tin Alloys: Surface Studies by Photoelectron Spectroscopy,” J. Met. Sci., 7 (1973), p. 109.
T. Farrell, “Surface Oxides on Al, Cu, Sn, Pb, and Some of Their Alloys: An ESCA Study,” Met. Sci., 10 (March 1976), p. 87.
R.A. Konetzki and Y.A. Chang, “Oxidation of Pb-2.9at.% Sn Alloys,” J. Mater. Res., 3 (3) (1988), p. 466.
A.J. Bevolo, J.D. Verhoeven, and M. Noack, “A LEELS and Auger Study of the Oxidation of Liquid and Solid Tin,” Surf. Sci., 134 (1983), p. 499.
S.N. Shah and D. Eurof Davies, “The Anodic Behaviour of Tin in Alkaline Solutions-1. 0.1M Sodium Borate Solution,” Electrochim. Acta, 8 (1963), p. 663.
R.L. Opila, “The Role of Grain Boundaries in the Surface Segregation of Tin in Tin-Lead Alloys,” J. Vac. Sci. Technol. A, 4 (2) (Mar./Apr. 1986), p. 173.
Sungil Cho et al., “Oxidation Study of Pure Tin and its Alloys via Electrochemical Reduction Analysis,” J. Electron. Mater., 34 (5) (2005).
E.E. de Kluizenaar, “Surface Oxidation of Molten Soft Solder: An Auger Study,” J. Vac. Sci. Technol. A, 1 (3) (1983), p. 1480.
M.L. Varsányi et al., “Electrochemical and In Situ Mössbauer Studies of Tin Passivation,” Electrochim. Acta, 30 (4) (1985), p. 529.
David R. Lide, Handbook of Chemistry and Physics, 73rd edition (Boca Raton, FL: CRC Press, 1993), pp. 75–78.
Frederick G. Yost et al., The Mechanics of Solder Alloy “Wetting & Spreading” (New York: Van Nostrand Reinhold, 1993), Chap. 6 (pp. 155–189).
D.M. Tench and D.P. Anderson, “Method of Assessing Solderability,” U.S. patent 5,262,022 (19 November 1993).
S.E.S. El Wakkad et al., “The Anodic Oxidation of Metals at Very Low Current Density,” J. Chem. Soc. Lond., (1954), p. 3103.
J.F. Moulder et al., Handbook of X-ray Photoelectron Spectroscopy (Wellesley, MA: Perkin-Elmer, 1992).
K.L. Lin and T.P. Liu, “High-Temperature Oxidation of Sn-Zn-Al Solder,” Oxidation of Metals, 50 (1998), p. 255.
Author information
Authors and Affiliations
Additional information
For more information, contact Sungil Cho, Korea Advanced Institute of Science and Technology, Department of Materials Science and Engineering, 373-1 Kusung-dong, Yusung-gu, Daejon 305-701, Republic of Korea; +82-17-394-1026; e-mail sicho@kaist.ac.kr.
Rights and permissions
About this article
Cite this article
Cho, S., Yu, J., Kang, S.K. et al. The oxidation of lead-free Sn alloys by electrochemical reduction analysis. JOM 57, 50–52 (2005). https://doi.org/10.1007/s11837-005-0136-8
Issue Date:
DOI: https://doi.org/10.1007/s11837-005-0136-8