Abstract
It is well known that the interactions of insect herbivores and their host plant can be mediated by microbes. Our central hypothesis is that herbivore-associated fungi might directly or indirectly affect plant–insect interactions. In this study, we identified five orally secreted fungi from field-collected black cutworm, Agrotis ipsilon, including Aspergillus parasiticus, Aspergillus niger, Geotrichum candidum, Fusarium subglutinans, and Mucor circinelloides f. lusitanicus. We found that caterpillars inoculated with F. subglutinans or M. circinelloides f. lusitanicus induced higher defense responses in plants, but with different patterns between different plants. These herbivore-induced defense responses reduced the growth of caterpillars. However, direct application of fungi to mechanically wounded tomato did not induce JA-related defense responses. The application of regurgitant from fungi-inoculated or non-inoculated caterpillars, suggested that regurgitant might be responsible for the fungi-mediated defense response in plants against caterpillar attack. Furthermore, both F. subglutinans and M. circinelloides f. lusitanicus benefited caterpillar growth when they fed on detached tomato leaves, but had no influence when caterpillars fed on artificial diet. Our finding suggests that insect-associated fungi could influence plant–insect interactions by indirectly mediating plant defense responses, and directly affecting caterpillar performance on host plants.





Similar content being viewed by others
References
Acevedo FE, Rivera-Vega LJ, Chung SH et al (2015) Cues from chewing insects-the intersection of DAMPs, HAMPs, MAMPs and effectors. Curr Opin Plant Biol 26:80–86. https://doi.org/10.1016/j.pbi.2015.05.029
Acevedo FE, Peiffer M, Tan CW et al (2017) Fall armyworm-associated gut bacteria modulate plant defense responses. Mol Plant–Microbe Interact 30:127–137. https://doi.org/10.1094/MPMI-11-16-0240-R
Acevedo FE, Smith P, Peiffer M et al (2019) Phytohormones in fall armyworm saliva modulate defense responses in plants. J Chem Ecol 45:598–609. https://doi.org/10.1007/s10886-019-01079-z
Alborn HT, Turlings TCJ, Jones TH et al (1997) An elicitor of plant volatiles from beet armyworm oral secretion. Science 276:945–949. https://doi.org/10.1126/science.276.5314.945
Anderson IC, Campbell CD, Prosser JI (2003) Potential bias of fungal 18S rDNA and internal transcribed spacer polymerase chain reaction primers for estimating fungal biodiversity in soil. Environ Microbiol 5:36–47. https://doi.org/10.1046/j.1462-2920.2003.00383.x
Bonaventure G, VanDoorn A, Baldwin IT (2011) Herbivore-associated elicitors: FAC signaling and metabolism. Trends Plant Sci 16:294–299. https://doi.org/10.1016/j.tplants.2011.01.006
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Brownlie JC, Cass BN, Riegler M et al (2009) Evidence for metabolic provisioning by a common invertebrate endosymbiont, wolbachia pipientis, during periods of nutritional stress. PLoS Pathog 5:e1000368. https://doi.org/10.1371/journal.ppat.1000368
Brune A, Dietrich C (2015) The gut microbiota of termites: digesting the diversity in the light of ecology and evolution. Annu Rev Microbiol 69:145–166. https://doi.org/10.1146/annurev-micro-092412-155715
Casteel CL, Hansen AK (2014) Evaluating insect-microbiomes at the plant-insect interface. J Chem Ecol 40:836–847. https://doi.org/10.1007/s10886-014-0475-4
Chaudhary R, Atamian HS, Shen Z et al (2014) GroEL from the endosymbiont Buchnera aphidicola betrays the aphid by triggering plant defense. Proc Natl Acad Sci 111:8919–8924. https://doi.org/10.1073/pnas.1407687111
Chen CY, Liu YQ, Song WM et al (2019) An effector from cotton bollworm oral secretion impairs host plant defense signaling. Proc Natl Acad Sci 116:14331–14338. https://doi.org/10.1073/pnas.1905471116
Chippendale GM (1970) Metamorphic changes in fat body proteins of the southwestern corn borer, Diatraea grandiosella. J Insect Physiol 16:1057–1068. https://doi.org/10.1016/0022-1910(70)90198-8
Chung SH, Rosa C, Scully ED et al (2013) Herbivore exploits orally secreted bacteria to suppress plant defenses. Proc Natl Acad Sci 110:15728–15733. https://doi.org/10.1073/pnas.1308867110
Desjardins AE, Hohn TM (1997) Mycotoxins in plant pathogenesis. Mol Plant–Microbe Interact 10:147–152. https://doi.org/10.1094/MPMI.1997.10.2.147
Douglas AE (2016) How multi-partner endosymbioses function. Nat Rev Microbiol 14:731–743. https://doi.org/10.1038/nrmicro.2016.151
Gange AC, Eschen R, Wearn JA et al (2012) Differential effects of foliar endophytic fungi on insect herbivores attacking a herbaceous plant. Oecologia 168:1023–1031. https://doi.org/10.1007/s00442-011-2151-5
Garantonakis N, Pappas ML, Varikou K et al (2018) Tomato inoculation with the endophytic strain Fusarium solani k results in reduced feeding damage by the zoophytophagous predator Nesidiocoris tenuis. Front Ecol Evol 6:126. https://doi.org/10.3389/fevo.2018.00126
Hannula SE, Zhu F, Heinen R, Bezemer TM (2019) Foliar-feeding insects acquire microbiomes from the soil rather than the host plant. Nat Commun 10:1254. https://doi.org/10.1038/s41467-019-09284-w
Hansen AK, Moran NA (2014) The impact of microbial symbionts on host plant utilization by herbivorous insects. Mol Ecol 23:1473–1496. https://doi.org/10.1111/mec.12421
Hong SB, Go SJ, Shin HD et al (2005) Polyphasic taxonomy of Aspergillus fumigatus and related species. Mycologia 97:1316–1329. https://doi.org/10.1080/15572536.2006.11832738
Hosokawa T, Koga R, Kikuchi Y et al (2010) Wolbachia as a bacteriocyte-associated nutritional mutualist. Proc Natl Acad Sci 107:769–774. https://doi.org/10.1073/pnas.0911476107
Jiggins FM, Hurst GDD, Jiggins CD et al (2000) The butterfly Danaus chrysippus is infected by a male-killing Spiroplasma bacterium. Parasitology 120:439–446. https://doi.org/10.1017/S0031182099005867
Lee SC, Billmyre RB, Li A et al (2014) Analysis of a food-borne fungal pathogen outbreak: virulence and genome of a Mucor circinelloides isolate from yogurt. MBio 5:e01390–e1414. https://doi.org/10.1128/mBio.01390-14
Leroy PD, Sabri A, Heuskin S et al (2011) Microorganisms from aphid honeydew attract and enhance the efficacy of natural enemies. Nat Commun 2:348. https://doi.org/10.1038/ncomms1347
Leslie JF (1995) Gibberella fujikuroi: available populations and variable traits. Can J Bot 73:282–291. https://doi.org/10.1139/b95-258
Logrieco A, Moretti A, Fornelli F et al (1996) Fusaproliferin production by Fusarium subglutinans and its toxicity to Artemia salina, SF-9 insect cells, and IARC/LCL 171 human B lymphocytes. Appl Environ Microbiol 62:3378–3384
Machouart M, Larche J, Burton K et al (2006) Genetic identification of the main opportunistic mucorales by PCR-restriction fragment length polymorphism. J Clin Microbiol 44:805–810. https://doi.org/10.1128/JCM.44.3.805-810.2006
Maciá-Vicente JG, Jansson H-B, Abdullah SK et al (2008) Fungal root endophytes from natural vegetation in Mediterranean environments with special reference to Fusarium spp. FEMS Microbiol Ecol 64:90–105. https://doi.org/10.1111/j.1574-6941.2007.00443.x
Malook S, Qi J, Hettenhausen C et al (2019) The oriental armyworm (Mythimna separata) feeding induces systemic defence responses within and between maize leaves. Philos Trans R Soc B Biol Sci 374:20180307. https://doi.org/10.1098/rstb.2018.0307
Mason CJ, Jones AG, Felton GW (2019a) Co-option of microbial associates by insects and their impact on plant-folivore interactions. Plant Cell Environ 42:1078–1086. https://doi.org/10.1111/pce.13430
Mason CJ, Ray S, Shikano I et al (2019b) Plant defenses interact with insect enteric bacteria by initiating a leaky gut syndrome. Proc Natl Acad Sci 116:15991–15996. https://doi.org/10.1073/pnas.1908748116
Moisan K, Cordovez V, van de Zande EM et al (2019) Volatiles of pathogenic and non-pathogenic soil-borne fungi affect plant development and resistance to insects. Oecologia 190:589–604. https://doi.org/10.1007/s00442-019-04433-w
Moriyama M, Nikoh N, Hosokawa T, Fukatsu T (2015) Riboflavin provisioning underlies Wolbachia’s fitness contribution to its insect host. MBio 6:1–8. https://doi.org/10.1128/mBio.01732-15
Musser RO, Cipollini DF, Hum-Musser SM et al (2005) Evidence that the caterpillar salivary enzyme glucose oxidase provides herbivore offense in solanaceous plants. Arch Insect Biochem Physiol 58:128–137. https://doi.org/10.1002/arch.20039
O’Donnell K, Nirenberg HI, Aoki T, Cigelnik E (2000) A Multigene phylogeny of the Gibberella fujikuroi species complex: Detection of additional phylogenetically distinct species. Mycoscience 41:61–78. https://doi.org/10.1007/BF02464387
Peiffer M, Felton GW (2014) Insights into the saliva of the brown marmorated stink bug Halyomorpha halys (Hemiptera: Pentatomidae). PLoS ONE 9:e88483. https://doi.org/10.1371/journal.pone.0088483
Peiffer M, Felton GW (2009) Do caterpillars secrete “oral secretions”? J Chem Ecol 35:326–335. https://doi.org/10.1007/s10886-009-9604-x
Pinto CF, Torrico-Bazoberry D, Penna M et al (2019) Chemical responses of Nicotiana tabacum (Solanaceae) induced by vibrational signals of a generalist herbivore. J Chem Ecol 45:708–714. https://doi.org/10.1007/s10886-019-01089-x
Ray S, Basu S, Rivera-Vega LJ et al (2016) Lessons from the far end: caterpillar frass-induced defenses in maize, rice, cabbage, and tomato. J Chem Ecol 42:1130–1141. https://doi.org/10.1007/s10886-016-0776-x
Reymond P (2013) Perception, signaling and molecular basis of oviposition-mediated plant responses. Planta 238:247–258. https://doi.org/10.1007/s00425-013-1908-y
Rings RW, Arnold FJ, Keaster AJ et al (1974) Worldwide annotated bibliography of the black cutworm Agrotis ipsilon, Hufnagel. Ohio Agric Res Dev Cent Res Circ 1:106
Sabree ZL, Kambhampati S, Moran NA (2009) Nitrogen recycling and nutritional provisioning by Blattabacterium, the cockroach endosymbiont. Proc Natl Acad Sci 106:19521–19526. https://doi.org/10.1073/pnas.0907504106
Schuster E, Dunn-Coleman N, Frisvad J, Van Dijck P (2002) On the safety of Aspergillus niger-a review. Appl Microbiol Biotechnol 59:426–435. https://doi.org/10.1007/s00253-002-1032-6
Stahl E, Hilfiker O, Reymond P (2018) Plant–arthropod interactions: who is the winner? Plant J 93:703–728. https://doi.org/10.1111/tpj.13773
Thaler JS, Humphrey PT, Whiteman NK (2012) Evolution of jasmonate and salicylate signal crosstalk. Trends Plant Sci 17:260–270. https://doi.org/10.1016/j.tplants.2012.02.010
Turner JG, Ellis C, Devoto A (2002) The jasmonate signal pathway. Plant Cell 14:153–164. https://doi.org/10.1139/b95-258
Wang J, Chung SH, Peiffer M et al (2016) Herbivore oral secreted bacteria trigger distinct defense responses in preferred and non-preferred host plants. J Chem Ecol 42:463–474. https://doi.org/10.1007/s10886-016-0712-0
Wang J, Peiffer M, Hoover K et al (2017) Helicoverpa zea gut-associated bacteria indirectly induce defenses in tomato by triggering a salivary elicitor(s). New Phytol 214:1294–1306. https://doi.org/10.1111/nph.14429
Wang J, Yang M, Song Y et al (2018) Gut-associated bacteria of Helicoverpa zea indirectly trigger plant defenses in maize. J Chem Ecol 44:690–699. https://doi.org/10.1007/s10886-018-0970-0
Xu HX, Qian LX, Wang XW et al (2019) A salivary effector enables whitefly to feed on host plants by eliciting salicylic acid-signaling pathway. Proc Natl Acad Sci 116:490–495. https://doi.org/10.1073/pnas.1714990116
Acknowledgements
This work was supported by the Agricultural and Food Research Initiative Program of United States Department of Agriculture (Grant No. 2017-67013-26596). Xuewei Chen was financially supported by China Scholarship Council (File No. 201706350199).
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Heikki Hokkanen.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11829_2020_9767_MOESM1_ESM.tif
Supplementary file1 (TIF 1705 kb) Supplemental Fig. 1 The detection of regurgitant using fluorescent dye Alexa 488. A) Fluorescent dye (green) was detected on the edges of the damaged leaves fed by caterpillars which were pretreated with fluorescent dye. B) No fluorescence was detected on the edges of the damaged leaves fed by caterpillars which were not pretreated with fluorescent dye.
11829_2020_9767_MOESM2_ESM.tif
Supplementary file2 (TIF 2171 kb) Supplemental Fig. 2 Fungi from regurgitant of fungi-inoculated or non-inoculated caterpillars and fungi from plant tissues fed by fungi-inoculated or non-inoculated caterpillars. Caterpillars were fed with Fusarium subglutinans (FS), or Mucor circinelloides f. lusitanicus (MC), or without fungi (CK).
11829_2020_9767_MOESM3_ESM.tif
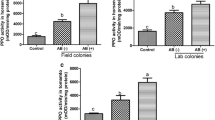
Supplementary file3 (TIF 711 kb) Supplemental Fig. 3 Polyphenol oxidase (PPO) and trypsin proteinase inhibitor (TPI) activities of plants fed on by Agrotis ipsilon. Plants were damaged by caterpillars which were pretreated with fungal solution of Aspergillus parasiticus (AP), Aspergillus niger (AN), or Geotrichum candidum (GC), or without fungi (CK), and a set of undamaged plants was included as negative control (Untreated). Values are mean ± SEM, different letters indicate significant differences obtained with ANOVA following by LSD test. PPO (F4,37 = 1.760, P = 0.158) and TPI (F4,31 = 10.140, P < 0.001) activities in tomato.
Rights and permissions
About this article
Cite this article
Chen, X., Peiffer, M., Tan, CW. et al. Fungi from the black cutworm Agrotis ipsilon oral secretions mediate plant–insect interactions. Arthropod-Plant Interactions 14, 423–432 (2020). https://doi.org/10.1007/s11829-020-09767-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-020-09767-4