Abstract
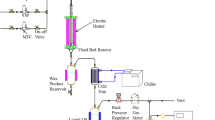
During catalytic pyrolysis of HFC-134a over γ-alumina, the formation of HF and coke causes catalyst deactivation. Catalyst deactivation and product selectivity depend on the contact time during catalytic pyrolysis of HFC-134a as reported in this paper. γ-Alumina calcined at 650 oC was used as the catalyst due to its higher quantity of acidic sites and larger surface area, which are crucial for catalytic pyrolysis. X-ray diffraction (XRD), scanning electron microscope-energy dispersive X-ray spectroscopy (SEM-EDS), and thermogravimetric analysis (TGA) of the catalysts were performed to determine the influence of contact time and flow rate of HFC-134a. 2 mL/min of HFC-134a balanced with nitrogen to 25, 50, 100, and 200 mL/min total flow rates was studied at 600 °C. 200 mL/min showed a 9.4 h catalyst lifetime with a small number of by-products. Shorter contact time between HFC-134a and HF with the catalyst was found to be the key to the longer lifetime of the catalyst. The catalyst lifetime was decreased with an increase in the HFC-134a input amount. Among 2, 4, and 6 mL/min input of HFC-134a, 2 mL/min showed the longest catalytic activity followed by 4 and 6 mL/min, respectively. Conversion of γ-alumina into AlF3 and deposition of coke were responsible for the deactivation.
Similar content being viewed by others
References
D. J. Wuebbles, D. R. Easterling, K. Hayhoe, T. Knutson, R. E. Kopp, J. P. Kossin, K. E. Kunkel, A. N. LeGran-de, C. Mears, W. V. Sweet, P. C. Taylor, R. S. Vose and M. F. Wehner, 2017: Our globally changing climate. In: Climate Science Special Report: Fourth National Climate Assessment, Volume I [Wuebbles, D. J., D. W. Fahey, K. A. Hibbard, D. J. Dokken, B. C. Stewart and T. K. Maycock (eds.)]. U.S. Global Change Research Program, Washington, DC, USA, pp. 35–72.
J. Srinivasan, Resonance, 13(12), 1146 (2008).
IPCC, 2014: Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Core Writing Team, R. K. Pachauri and L. A. Meyer (eds.)]. IPCC, Geneva, Switzerland, 151 pp.
https://ec.europa.eu/clima/policies/strategies/progress/kyoto_2_en (accessed Sep. 21, 2020).
G. J. M. Velders, A. R. Ravishankara, M. K. Miller, M. J. Molina, J. Alcamo, J. S. Daniel, D. W. Fahey, S. A. Montzka and S. Reimann, Science, 335(6071), 922 (2012).
M. J. Molina and F. S. Rowland, Nature, 249, 810 (1974).
https://ozone.unep.org/treaties/montreal-protocol (accessed Oct. 07, 2020).
G. Myhre, D. Shindell, F.-M. Bréon, W. Collins, J. Fuglestvedt, J. Huang, D. Koch, J.-F. Lamarque, D. Lee, B. Mendoza, T. Nakajima, A. Robock, G. Stephens, T. Takemura and H. Zhang, 2013: Anthropogenic and Natural Radiative Forcing. In: Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change [Stocker, T. F., D. Qin, G.-K. Plattner, M. Tignor, S. K. Allen, J. Boschung, A. Nauels, Y. Xia, V. Bex and P. M. Midgley (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA.
https://eia-international.org/wp-content/uploads/EIA-Kigali-Amendment-to-the-Montreal-Protocol-FINAL.pdf (accessed Oct. 28, 2020).
UNFCCC, CDM Methodology Booklet, 2019.
M. Shin, D. Jang, Y. Lee, Y. Kim and E. Kim, J. Mater. Cycles Waste Manag., 19(2), 754 (2017).
W. Han, Y. Li, H. Tang and H. Liu, J. Fluorine Chem., 140, 7 (2012).
M. Ohno, Y. Ozawa and T. Ono, Int. J. Plasma Environ. Sci. Technol., 1(2), 159 (2007).
M. S. Gandhi and Y. S. Mok, Int. J. Environ. Sci. Technol., 12(2), 499 (2013).
Y. F. Wang, W. J. Lee, C. Y. Chen and L. Te Hsieh, Ind. Eng. Chem. Res., 38(9), 3199 (1999).
Y. Takita, T. Tanabe, M. Ito, M. Ogura, T. Muraya, S. Yasuda, H. Nishiguchi and T. Ishihara, Ind. Eng. Chem. Res., 41(11), 2585 (2002).
Z. M. El-Bahy, R. Ohnishi and M. Ichikawa, Appl. Catal. B Environ., 40(2), 81 (2003).
A. Iizuka, H. Ishizaki, A. Mizukoshi, M. Noguchi, A. Yamasaki and Y. Yanagisawa, Ind. Eng. Chem. Res., 50(21), 11808 (2011).
T. U. Han, B. Yoo, Y. Kim, B. Hwang, G. L. Sudibiya, Y. Park and S. Kim, Korean J. Chem. Eng., 35(8), 1611 (2018).
X. F. Xu, J. Y. Jeon, M. H. Choi, H. Y. Kim, W. C. Choi and Y. K. Park, J. Mol. Catal. A Chem., 266(1–2), 131 (2007).
W. Han, Y. Chen, B. Jin, H. Liu and H. Yu, Greenh. Gases Sci. Technol., 4(1), 121 (2014).
J. Song, S. Chung, M. Kim, Y. Lee, K. Lee and J. Kim, J. Mol. Catal. A Chem., 370, 50 (2013).
W. Jia, M. Liu, X. Lang, C. Hu, J. Li and Z. Zhu, Catal. Sci. Technol., 5(6), 3103 (2015).
C. M. A. Swamidos, M. Sheraz, A. Anus, S. J. Jeong, Y. Park, Y. Kim and S. Kim, Catalysts, 9(3), 270 (2019).
W. Jia, Q. Wu, X. Lang, C. Hu, G. Zhao, J. Li and Z. Zhu, Catal. Lett., 145(2), 654 (2015).
J. Ryu, K. No, Y. Kim, E. Park and S. Hong, Sci. Rep., 6, 36176 (2016).
Y. Higashi, N. Sakoda, M. A. Islam, Y. Takata, S. Koyama and R. Akasaka, J. Chem. Eng. Data, 63(2), 417 (2018).
N. Yaghobi, J. King Saud Univ. — Eng. Sci., 25(1), 1 (2013).
K. Açikalin, F. Karaca and E. Bolat, Fuel, 95, 169 (2012).
H. Yang, R. Coolman, P. Karanjkar, H. Wang, P. Dornath, H. Chen, W. Fan, W. C. Conner, T. J. Mountziaris, and G. Huber, Green Chem., 19(1), 286 (2017).
H. Y. Li, Y. J. Yan and Z. W. Ren, J. Fuel Chem. Technol., 36(6), 666 (2008).
A. Aho, A. Tokarev, P. Backman, N. Kumar, K. Eränen, M. Hupa, B. Holmbom, T. Salmi and D. Y. Murzin, Top. Catal., 54, 941 (2011).
E. Pütün, Energy, 35(7), 2761 (2010).
S. J. Jeong, G. L. Sudibya, J. Jeon, Y. Kim, C. M. A. Swamidoss and S. Kim, Catalysts, 9(11), 901 (2019).
W. J. Tseng and P. S. Chao, Ceram. Int., 39, 3779 (2013).
R. A. Spurr and H. Myers, Anal. Chem., 29, 760 (1957).
J. Y. Jeon, X.-F. Xu, M. H. Choi, H. Y. Kim and Y.-K. Park, Chem. Commun., 3, 1244 (2003).
T. Krahl and E. Kemnitz, Catal. Sci. Technol., 7(4), 773 (2017).
Z. Xi, X. Liu, J. Li, J. Yuan, W. Jia, X. Liu, M. Liu and Z. Zhu, ChemistrySelect, 4(15), 4506 (2019).
M. Kim, Y. Kim, J. Youn, I. Choi, K. Hwang, S. G. Kim, Y. Park, S. Moon, K. B. Lee and S. Jeon, Catalysts, 10(7), 766 (2020).
Acknowledgement
This research was supported by a grant (20UGCP-B157945-01) from the Urban-based complex plant demonstration study utilizing underground space program funded by the Ministry of Land, Infrastructure, and Transport, Republic of Korea, and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number) (NRF-2019R1A6A3A01096378).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Supporting Information
11814_2021_776_MOESM1_ESM.pdf
The role of contact time and input amount of 1,1,1,2-tetrafluoroethane (HFC-134a) on the catalyst lifetime and product selectivity in catalytic pyrolysis
Rights and permissions
About this article
Cite this article
Anus, A., Sheraz, M., Jeong, S. et al. The role of contact time and input amount of 1,1,1,2-tetrafluoroethane (HFC-134a) on the catalyst lifetime and product selectivity in catalytic pyrolysis. Korean J. Chem. Eng. 38, 1240–1247 (2021). https://doi.org/10.1007/s11814-021-0776-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-021-0776-6