Abstract
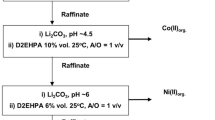
This research was carried out to recover some valuable metals from the electrodes of spent mobile phone batteries by leaching process in laboratory scale. Two types of spent mobile phone batteries were employed in this study: nickel-metal hydride (Ni-MH) and lithium-ion (Li-ion) batteries. Effects of parameters including types of acid (H2SO4, HNO3 and HCl), acid concentration (1-6 M), solid-liquid ratio (10-40 g/l), leaching time (5-120 min) and leaching temperature (303-363 K) on the leaching percentages of Co and Ni were investigated. The preliminary results indicated that HCl provided higher leaching percentages than that of H2SO4 and HNO3 for both metals at all leaching conditions. At optimum conditions, greater than 92% and 84% of Ni and Co were leached, respectively. Further investigation indicated that the leaching process of both metals was endothermic with the rate law of a second-order reaction and its kinetics was principally controlled by the physical process.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakultung, S., Pruksathorn, K. & Hunsom, M. Simultaneous recovery of valuable metals from spent mobile phone battery by an acid leaching process. Korean J. Chem. Eng. 24, 272–277 (2007). https://doi.org/10.1007/s11814-007-5040-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-007-5040-1