Abstract
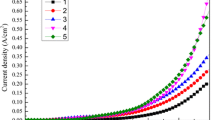
Heavy particulate matter (PM) pollution and high energy consumption are the bottlenecks of hydrometallurgy, especially in the electrolysis process. Therefore, an urgent need is to explore PM reduction methods with production performance co-benefits. This study presents three PM reduction methods based on controlling operating parameters, i.e., lowering electrolyte temperature, H2SO4 concentration, and current density of the cathode. The optimized conditions were also investigated using the response surface methodology to balance the PM reduction effect and Zn production. The results showed that lowering electrolyte temperature is the most efficient, with an 89.0% reduction in the PM generation flux (GFPM). Reducing H2SO4 concentration led to the minimum side effects on the current efficiency of Zn deposition (CEZn) or power consumption (PC). With the premise of non-deteriorating CEZn and PC, GFPM can be reduced by 86.3% at the optimal condition (electrolyte temperature = 295 K, H2SO4 = 110 g/L, current density = 373 A/m2). In addition, the reduction mechanism was elucidated by comprehensively analyzing bubble characteristics, electrochemical reactions, and surface tension. Results showed that lower electrolyte temperature inhibited the oxygen evolution reaction (OER) and compressed gas volume. Lower H2SO4 concentration inhibited the hydrogen evolution reaction (HER) and reduced electrolyte surface tension. Lower current density inhibited both OER and HER by decreasing the reaction current. The inhibited gas evolutions reduced the microbubbles’ number and size, thereby reducing GFPM. These results may provide energy-efficient PM reduction methods and theoretical hints of exploring cleaner PM reduction approaches for industrial electrolysis.

Article PDF
Similar content being viewed by others
References
Al Shakarji R, He Y, Gregory S (2011a). The sizing of oxygen bubbles in copper electrowinning. Hydrometallurgy, 109(1): 168–174
Al Shakarji R, He Y, Gregory S (2011b). Statistical analysis of the effect of operating parameters on acid mist generation in copper electrowinning. Hydrometallurgy, 106(1–2): 113–118
Al Shakarji R, He Y, Gregory S (2013). Performance evaluation of acid mist reduction techniques in copper electrowinning. Hydrometallurgy, 131–132: 76–80
Berny A, Popinet S, Seon T, Deike L (2021). Statistics of jet drop production. Geophysical Research Letters, 48(10): e2021GL092919
Bouakaz A, Versluis M, De Jong N (2005). High-speed optical observations of contrast agent destruction. Ultrasound in Medicine & Biology, 31(3): 391–399
Cai Z, Wang J, Lu Z, Zhan R, Ou Y, Wang L, Dahbi M, Alami J, Lu J, Amine K, et al. (2022). Ultrafast metal electrodeposition revealed by in situ optical imaging and theoretical modeling towards fast-charging Zn battery chemistry. Angewandte Chemie International Edition, 61(14): e202116560
Deike L, Reichl B G, Paulot F (2022). A mechanistic sea spray generation function based on the sea state and the physics of bubble bursting. AGU Advances, 3(6): 14942
Dhak D, Mahon M, Asselin E, Alfantazi A (2011). The effects of mixtures of acid mist suppression reagents on zinc electrowinning from spent electrolyte solutions. Hydrometallurgy, 108(1–2): 1–10
Dusen J V, Smith J W (1989). Comparison of the effectiveness of bubble coalescence and foamed surfactant in controlling the acid mist formed by electrowinning cells. American Industrial Hygiene Association Journal, 50(5): 252–256
Dwivedi K A, Huang S J, Wang C T (2022). Integration of various technology-based approaches for enhancing the performance of microbial fuel cell technology: a review. Chemosphere, 287: 132248
Ghabache E, Seon T (2016). Size of the top jet drop produced by bubble bursting. Physical Review Fluids, 1(5): 051901
He H, Yu Q, Lai C, Zhang C, Liu M, Huang B, Pu H, Pan X (2021). The treatment of black-odorous water using tower bipolar electro-flocculation including the removal of phosphorus, turbidity, sulfion, and oxygen enrichment. Frontiers of Environmental Science & Engineering, 15(2): 18
He Y, Cui Y, Shang W, Zhao Z, Tan P (2022). Insight into the bubble-induced over potential towards high-rate charging of Zn-air batteries. Chemical Engineering Journal, 448: 137782
Ji S L, Weon B M, Su J P, Je J H, Fezzaa K, Lee W K (2011). Size limits the formation of liquid jets during bubble bursting. Nature Communications, 2(1): 367
Kargl-Simard C, Huang J H, Alfantazi A M (2003). Electrical conductivity and density of CoSO4/H2SO4 solutions in the range of modern cobalt electrowinning electrolytes. Minerals Engineering, 16(6): 529–535
Ke W R, Kuo Y M, Lin C W, Huang S H, Chen C C (2017). Characterization of aerosol emissions from single bubble bursting. Journal of Aerosol Science, 109: 1–12
Lasia A (2019). Mechanism and kinetics of the hydrogen evolution reaction. International Journal of Hydrogen Energy, 44(36): 19484–19518
Luo J, Jiao P, Duan N, Xu F, Jiang L (2018). Flow field characterization in the vicinity of vertical plane electrodes in a bench-scale zinc electrowinning cell. Hydrometallurgy, 181: 103–112
Ma Z, Duan L, Jiang J, Deng J, Xu F, Jiang L, Li J, Wang G, Huang X, Ye W, Wen Y, Zhang G, Duan N (2020a). Characteristics and threats of particulate matter from zinc electrolysis manufacturing facilities. Journal of Cleaner Production, 259: 120874
Ma Z, Jiang J, Duan L, Li Z, Deng J, Li J, Zhang R, Zhou C, Xu F, Jiang L, Duan N (2020b). Ultrasonication to reduce particulate matter generated from bursting bubbles: a case study on zinc electrolysis. Journal of Cleaner Production, 272: 122697
Maslova M V, Ivanenko V I, Yanicheva N Y, Mudruk N V (2020). Comparison of the sorption kinetics of lead(II) and zinc(II) on titanium phosphate ion-exchanger. International Journal of Molecular Sciences, 21(2): 447
Mason T J, Lorimer J P, Saleem S, Paniwnyk L (2001). Controlling emissions from electroplating by the application of ultrasound. Environmental Science & Technology, 35(16): 3375–3377
Mcginnity J J, Nicol M J (2014). Sulfuric acid mist: generation, suppression, health aspects, and analysis. Mineral Processing and Extractive Metallurgy Review, 35(3): 149–192
Mokarram M, Pourghasemi H R, Zhang H (2020). Predicting non-carcinogenic hazard quotients of heavy metals in pepper (Capsicum annum L.) utilizing electromagnetic waves. Frontiers of Environmental Science & Engineering, 14(6): 114
Papachgristodoulou A, Foulkes F R, Smith J W (1985). Bubble characteristics and aerosol formation in electrowinning cells. Journal of Applied Electrochemistry, 15(4): 581–590
Qu C, Li K, Xie Z, Yang S, Zhao J, Sun D, Sun Y (2022). Stacking polymer microspheres matrix: a facile, practical, and energy-saving strategy for suppression of acid mist. Environmental Technology, 44: 18, 2693–2701
Shaw L, Shaw D, Hardisty M, Britz-Mckibbin P, Verma D K (2020). Relationships between inhalable and total hexavalent chromium exposures in steel passivation, welding and electroplating operations of Ontario. International Journal of Hygiene and Environmental Health, 230: 113601
Sorour N, Zhang W, Ghali E, Houlachi G (2017). A review of organic additives in zinc electrodeposition process (performance and evaluation). Hydrometallurgy, 171: 320–332
Theerthagiri J, Madhavan J, Lee S J, Choi M Y, Ashokkumar M, Pollet B G (2020). Sonoelectrochemistry for energy and environmental applications. Ultrasonics Sonochemistry, 63: 104960
Wang X, Deane G B, Moore K A, Ryder O S, Stokes M D, Beall C M, Collins D B, Santander M V, Burrows S M, Sultana C M, Prather K A (2017). The role of jet and film drops in controlling the mixing state of submicron sea spray aerosol particles. Proceedings of the National Academy of Sciences of the United States of America, 114(27): 6978–6983
Wu X, Liu Z, Liu X (2014). The effects of additives on the electrowinning of zinc from sulphate solutions with high fluoride concentration. Hydrometallurgy, 141: 31–35
Xu K, Lu J, Tegladza I D, Xu Q, Yang Z, Lv G (2020a). Combined metal/air fuel cell and electrocoagulation process: energy generation, flocs production and pollutant removal. Chemosphere, 255:126925
Xu X, Li D, Chen L, Liu M, Jia J J H (2020b). Improve the energy efficiency: effects of additives on longtime zinc electrowinning. Hydrometallurgy, 193: 105326
Xue T, Xu L, Wang Q (2019). Measurement of seawater surface tension coefficient based on bubble rising behavior. Measurement, 138: 332–340
Zeng M, Li Y (2015). Recent advances in heterogeneous electrocatalysts for the hydrogen evolution reaction. Journal of Materials Chemistry. A, Materials for Energy and Sustainability, 3(29): 14942–14962
Zhang C, Duan N, Jiang L, Xu F (2018). The impact mechanism of Mn2+ ions on oxygen evolution reaction in zinc sulfate electrolyte. Journal of Electroanalytical Chemistry (Lausanne, Switzerland), 811: 53–61
Acknowledgements
This research was financially supported by the National Natural Science Foundation of China (No. 22106081), the Natural Science of Foundation of Shandong Province, China (No. ZR202103040646), the special fund of State Key Joint Laboratory of Environment Simulation and Pollution Control (China) (No. 20K09ESPCT), the Major Basic Research Projects of Natural Science Foundation of Shandong Province (China) (No. ZR2020KE025), the Fundamental Research Funds for the Central Universities (China) (No. 22120220166).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors Fuyuan Xu and Ning Duan are Editorial Board Members of Frontiers of Environmental Science & Engineering. The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Highlights
• Electrolytic PM can be reduced by controlling operation parameters.
• The optimization conditions exist, reducing PM without deteriorating PC and CEZn.
• Abatement essence is to inhibit gas evolution reactions.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ma, Z., Jiang, J., Duan, L. et al. Synergistic promotion of particulate matter reduction and production performance via adjusting electrochemical reactions in the zinc electrolysis industry. Front. Environ. Sci. Eng. 18, 2 (2024). https://doi.org/10.1007/s11783-024-1762-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11783-024-1762-0