Abstract
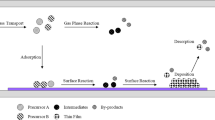
Non-thermal plasma technologies have shown their promising potential specially for the low concentration of volatile organic compound control in indoor air in recent years. But it is also high energy consuming. So, to improve the energy efficiency, adding catalysts which enhance the plasma chemical reactions to plasma reactors may be a good selection. Therefore, in this study the manganese dioxide assisted silent discharge plasma was developed for benzene conversion at a relatively high energy efficiency. The results show that MnO2 could promote complete oxidation of benzene with O2 and O3 produced in the plasma discharge zone. The energy efficiency of benzene conversion with MnO2 was two folds as much as that without catalysts. It was also found that the site of MnO2 in the reactor and the energy density had effects on benzene conversion. While the energy density was lower than 48 J/L, benzene conversion decreased with the increase in the distance between MnO2 bed and the plasma discharge zone. Whereas when the energy density was higher than 104 J/L, benzene conversion had an optimal value that was governed by the distance between MnO2 bed and the plasma discharge zone. The mechanism of benzene oxidation in plasma discharges and over MnO2 is discussed in detail.
Similar content being viewed by others
References
Kim K, Kang C, You Y, Chung M, Woo M, Jeong W, Park N, Ahn H. Adsorption-desorption characteristics of VOCs over impregnated activated carbons. Catalysis Today, 2006, 111: 223–228
Oda T. Non-thermal plasma processing for environmental protection: Decomposition of dilute VOCs in air. J Electrostat, 2003, 57: 293–311
Demidiouk V, Moon S I, Chae J O. Toluene and butyl acetate removal from air by plasma-catalytic system. Catal Commun, 2003, 4: 51–56
Hippler H, Rahn R, Troe J. Temperature and pressure dependence of ozone formation rates in the range 1–1000 bar and 90–370 K. Journal of Chemical Physics, 1990, 93–9: 6,560–6,569
Peyrous R, Pignolet P, Held B. Kinetic simulation of gaseous species created by an electrical discharge in dry or humid oxygen. Journal of Physics D: Applied Physics, 1989, 22: 1,658–1,667
Herron J T, Huie R E. Rate constants for the reactions of atomic oxygen (O3P) with organic compounds in the gas phase. Journal of Physical and Chemical Reference Data, 1973, 2: 467–518
Naydenov A, Mehandjiev D. Complete oxidation of benzene on manganese dioxide by ozone. Appl Catal A: Gen, 1993, 97: 17–22
Eliasson B, Kogelschatz U. Modeling and applications of silent discharge plasmas. IEEE Transactions on Plasma Science, 1991, 19(2): 309–323
Nunez C M, Ramsey G H, Ponder W H, Abbott J H, Hamel L E, Kariher P H. Corona destruction: an innovative control technology for VOCs and air toxics. Air Wastes, 1993, 43: 242–247
Radhakrishnan R, Oyama S T, Chen J G, Asakura K. Electron transfer effects in ozone decomposition on supported manganese oxide. J Phys Chem B, 2001, 105(19): 4,245–4,253
Bielański A, Najbar M. V2O5-MoO3 catalysts for benzene oxidation. Appl Catal A: Gen, 1997, 157: 223–261
Author information
Authors and Affiliations
Corresponding author
Additional information
__________
Translated from China Environmental Science, 2006, 26(6): 703–707 [译自: 中国环境科学]
Rights and permissions
About this article
Cite this article
Lu, B., Ji, M., Yu, X. et al. Benzene conversion by manganese dioxide assisted silent discharge plasma. Front. Environ. Sci. Eng. China 1, 477–481 (2007). https://doi.org/10.1007/s11783-007-0076-3
Issue Date:
DOI: https://doi.org/10.1007/s11783-007-0076-3