Abstract
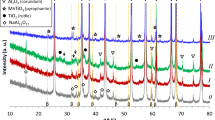
The precursors with NiCO3 · 2Ni(OH)2 · 2H2O, Fe2O3 · nH2O coated alumina microspheres were prepared by the aqueous heterogeneous precipitation using metal salts, ammonium bicarbonate and α-Al2O3 micropowders as the starting materials. Magnetic metal Ni, α-Fe coated alumina, core-shell structural microspheres were successfully obtained by thermal reduction of the precursors at 700 °C for 2 h, respectively. Powders of the precursors and the resultant metal (Ni, α-Fe) coated alumina micropowders were characterized by scanning electron microscopy, energy dispersive spectroscopy and X-ray diffraction. The results show that optimized precipitation parameters are concentration of alumina micropowders of 15 g/L, rate of adding reactants of 5 mL/min and pH value of 7.5. And under the optimized conditions, the spherical precursors without aggregations or agglomerations are obtained, then transferred into Ni, α-Fe coated alumina microspheres by thermal reduction. It is possible to adjust metal coating thicknesses and fabricate a multilayer structured metal/ceramics, core-shell microspherical powder materials.
Similar content being viewed by others
References
LI Guo-jun, ZHAO Shi-ke, HUANG Xiao-xian. Preparation of nano-Ni-coated-Al2O3 composite powder[J]. Journal of Inorganic Materials, 2002, 17(2): 235–240. (in Chinese)
Xue D S, Huang Y L, Ma Y, et al. Magnetic properties of pure Fe-Al2O3 nanocomposites[J]. Journal of Materials Science Letters, 2003, 22(24): 1817–1820.
Oh S T, Namkung S, Lee J S. Synthesis and magnetic properties of a Fe-Ni alloy dispersed Al2O3 nanocomposite powder prepared by a chemical method[J]. Journal of Materials Science Letters, 2002, 21(4): 275–277.
Kim S S, Kim S T, Ahn J M, et al. Magnetic and microwave absorbing properties of Co-Fe thin films plated on hollow ceramic microspheres of low density[J]. Journal of Magnetism and Magnetic Materials, 2004, 271(1): 39–45.
Vendange V, Tronc E, Clomban P H. Towards microwave absorbing metal-oxide nanocomposites: the problem of metal/matrix reaction[J]. Journal of Sol-Gel Science and Technology, 1998, 11(3): 299–307.
Kishimoto S, Shinya N. New fabrication method for metallic closed cellular materials containing polymers [J]. Materials and Design, 2001, 22(7): 535–539.
FENG Ze-kun, ZHANG Xiu-cheng, HE Hua-hui. Preparation of magnetic nano-granular films and investigation of its microwave electromagnetic property [J]. Ordnance Materials Science and Engineering, 2002, 25(3): 19–21. (in Chinese)
LI Ya-dong, LI Cheng-wei, DUAN Xiang-feng, et al. Preparation of nanocrystalline NiO in mixed solvent [J]. Journal of China University of Science and Technology, 1997, 27(3): 346–349. (in Chinese)
LIU Xiao-hong, LU Fang-yi, ZHENG Dian-mo, et al. Experimental study on producing K2SO4 and oxidation iron red from FeSO4[J]. Chemical Reaction Engineering and Technology, 2000, 16(3): 295–230. (in Chinese)
PENG Mei-xun, WANG Ling-seng, SHEN Xiang-qian, et al. Microstructures and formation mechanism of spherical β-Ni(OH)2[J]. The Chinese Journal of Nonferrous Metals, 2003, 13(5): 1130–1135. (in Chinese)
AN Sheng-li, SONG Xi-wen, ZHAO Wen-guang. Chemical principles on preparation of ultrafine oxide powders[J]. Journal of Baotou University of Iron and Steel Technology, 2000, 19(2): 107–110. (in Chinese)
CAO Mao-sheng, QIN Ru-ru, QIU Cheng-jun, et al. Matching design and mismatching analysis towards radar absorbing coatings based on conducting plate [J]. Materials and Design, 2003, 24(5): 391–396.
JIAO Huan, LUO Fa, ZHOU Wan-cheng. Development and prospect of nano radar absorber[J]. Aerospace Materials and Technology, 2001, 31(5): 9–11. (in Chinese)
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Project(50134020) supported by the National Natural Science Foundation of China; project(04CX01) supported by the Nature Science Innovative Foundation of Jiangsu University
Rights and permissions
About this article
Cite this article
Shen, Xq., Li, Dh., Jing, Mx. et al. Coating of Fe, Ni on α-alumina microspheres by heterogeneous precipitation. J Cent. South Univ. Technol. 13, 22–26 (2006). https://doi.org/10.1007/s11771-006-0100-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-006-0100-y