Abstract
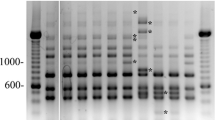
American foulbrood is an important bacterial disease affecting the larvae of honey bees (Apis mellifera L.) caused by Paenibacillus larvae. Due to easy transmission of disease and the ability of bacteria to create spores, it is a bacterium resistant to both physical and chemical conditions. The study aims to isolate, perform microbiological analyses, and determine biochemical properties and genotypes P. larvae strains from AFB samples collected from Turkey's Central and Eastern Black Sea regions. An isolation study was conducted on adult bees, larvae, honey, and primary honeycomb samples from suspected colonies in the regions under study. After the purification of bacterial isolates from samples, P. larvae strains were identified using biochemical and molecular methods. The genetic diversity and ERIC types of P. larvae isolates were determined by rep-PCR DNA genotyping using BOX A1R and MBO REP1 primers and multiplex-PCR. A phylogenetic tree of P. larvae strains was constructed in the study. All P. larvae isolates were determined as ERIC I type. According to the rep-PCR results of P. larvae strains, 15 of the 28 isolates were Ab genotype (54%), 7 (25%) Aβ genotype, 4 (14%) AB genotype, 1 (3.5%) αB genotype, and 1 (3.5%) ab genotype. From an epidemiological viewpoint, it was determined that Ab and Aβ genotypes were widely distributed, while other genotypes (AB, αB, and ab) showed less spread. The results of the study will guide researchers in taking relevant measures to prevent and control American foulbrood.




Similar content being viewed by others
Data Availability
Datasets used in this study will be made available by the corresponding author with no hesitation upon genuine request.
Abbreviations
- AFB:
-
American foulbrood
- PFGE:
-
Pulsed-field gel electrophoresis
- RFLP:
-
Restriction fragment length polymorphic DNA analysis
- RAPD:
-
Random amplified polymorphic DNA assay
- AP-PCR:
-
Arbitrary primed PCR
- AFLP:
-
Amplified fragment length polymorphism
- Rep-PCR:
-
Repetitive element based PCR
- ERIC:
-
Enterobacterial repetitive intergenic consensus sequence
- NCBI:
-
National Center for Biotechnology Information
- BLAST:
-
Basic Local Alignment Search Tool
References
Alippi AM, Aguilar OM (1998) Characterization of isolates of Paenibacillus larvae subsp. larvae from diverse geographical origin by the polymerase chain reaction and BOX primers. J Invertebr Pathol 72(1):21–27. https://doi.org/10.1006/jipa.1998.4748
Amšiejute P, Jurgelevičius V, Mačiulskis P, Butrimaite-Ambrozevičiene C, Pilevičiene S, Janeliunas Z, Kutyriova T, Jacevičiene I, Paulauskas A (2022) Molecular diversity of Paenibacillus larvae strains isolated from Lithuanian apiaries. Front Vet Sci 9:959636. https://doi.org/10.3389/fvets.2022.959636
Balkaya İ, Gülbaz H, Avcıoğlu H, Güven E (2016) Türkiye’de görülen bal arısı (Apis mellifera) hastalıkları. Atatürk Univ Vet Bil Derg 11(3): 339–347. https://doi.org/10.17094/ataunivbd.282993
Bassi S, Formato G, Milito M, Trevisiol K, Salogni C, Carra E (2015) Phenotypic characterization and ERIC-PCR based genotyping of Paenibacillus larvae isolates recovered from American foulbrood outbreaks in honey bees from Italy. Vet Q 35(1):27–32. https://doi.org/10.1080/01652176.2014.993095
Baş Y, Alpay Karaoglu Ş (2015) Characterization and antimicrobial susceptibility of spore forming bacilli isolated from honeycomb. Recep Tayyip Erdogan Univ Graduate School Natural Appl Sci Depart Biol, Master Thesis, pp123
Beims H, Bunk B, Erler S, Mohr KI, Spröer C, Pradella S, Günther G, Rohde M, der Ohe W, Steinert M (2020) Discovery of Paenibacillus larvae ERIC V: Phenotypic and genomic comparison to genotypes ERIC I-IV reveal different inventories of virulence factors which correlate with epidemiological prevalences of American Foulbrood. Int J Med Microbiol 310(2):151394. https://doi.org/10.1016/j.ijmm.2020.151394
Benson DA, Karsch-Mizrachi I, Clark K, Lipman, DJ, Ostell J, Sayers EW (2012) GenBank. Nucleic Acids Res 40(Database issue):D48–D53. https://doi.org/10.1093/nar/gkr1202
Bertolotti AC, Forsgren E, Schäfer MO, EuroPLarva Consortium, Sircoulomb, F, Gaïani N, Ribière-Chabert M, Paris L, Lucas P, de Boisséson C, Skarin J, Rivière MP (2021). Development and evaluation of a core genome multilocus sequence typing scheme for Paenibacillus larvae, the deadly American foulbrood pathogen of honeybees. Environ Microbiol 23(9):5042–5051. https://doi.org/10.1111/1462-2920.15442
Biová J, Bzdil J, Dostálková S, Petřivalský M, Brus J, Carra E, Danihlík J (2021) American Foulbrood in the Czech Republic: ERIC II genotype of Paenibacillus larvae is prevalent. Front Vet Sci 8:698976. https://doi.org/10.3389/fvets.2021.698976
Cherif A, Borin S, Rizzi A, Ouzari H, Boudabous A, Daffonchio D (2002) Characterization of a repetitive element polymorphism-polymerase chain reaction chromosomal marker that discriminates Bacillus anthracis from related species. J Appl Microbiol 93(3):456–462. https://doi.org/10.1046/j.1365-2672.2002.01712.x
Çakmak İ, Aydın L, Güleğen E, Korkut M (2003) Honeybee pests and diseases survey in Southern Marmara Region of Turkey. Uludag Bee J 3(2):33–35. https://dergipark.org.tr/en/pub/uluaricilik/issue/53709/162636
de Graaf DC, Alippi AM, Antúnez K, Aronstein KA, Budge G, Koker DDe, Smet LD, Dingman DW, Evans JD, Foster LJ, Fünfhaus A, Garcia-Gonzalez E, Gregore A, Human H, Murray KD, Nguyen BK, Poppinga L, Spivak M, Engelsdorp DV, Wilkins S, Genersch E (2013) Standard methods for American Foulbrood research. J Apicul Res 52(1):1–28. https://doi.org/10.3896/IBRA.1.52.1.11
Genersch E, Forsgren E, Pentikäinen J, Ashiralieva A, Rauch S, Kilwinski J, Fries I (2006) Reclassification of Paenibacillus larvae subsp. pulvifaciens and Paenibacillus larvae subsp. larvae as Paenibacillus larvae without subspecies differentiation. Int J Syst Evol Microbiol 56(Pt 3):501–511. https://doi.org/10.1099/ijs.0.63928-0
Genersch E, Otten C (2003) The use of repetitive element PCR fingerprinting (rep-PCR) for genetic subtyping of German field isolates of Paenibacillus larvae subsp. larvae. Apidologie 34(3):195–206. https://doi.org/10.1051/apido:2003025
Genersch E (2010) American Foulbrood in honeybees and its causative agent, Paenibacillus larvae. J Invertebr Pathol 103(Suppl 1):S10–S19. https://doi.org/10.1016/j.jip.2009.06.015
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp 41:95–98
Hamdi C, Essanaa J, Sansonno L, Crotti E, Abdi K, Barbouche N, Balloi A, Gonella E, Alma A, Daffonchio D, Boudabous A, Cherif A (2013) Genetic and biochemical diversity of Paenibacillus larvae isolated from Tunisian infected honey bee broods. BioMed Res Int 1-9. https://doi.org/10.1155/2013/479893
Hansen H, Brødsgaard CJ (1999) American foulbrood: a review of its biology, diagnosis and control. Bee World 80(1):5–23. https://doi.org/10.1080/0005772X.1999.11099415
Harvey SP, Minter JM (2005) Ribotyping of Burkholderia mallei isolates. FEMS Immunol Med Mic 44(1):91–97. https://doi.org/10.1016/j.femsim.2004.12.002
Herschleb J, Ananiev G, Schwartz DC (2007) Pulsed-field gel electrophoresis. Nat Prot 2(3):677–684. https://doi.org/10.1038/nprot.2007.94
Hirai Y, Suzuki T, Inaba N, Minoguchi N, Takamatsu D (2016) Existence of Paenibacillus larvae genotypes ERIC I-ST2, ERIC I-ST15 and ERIC II-ST10 in the western region of Aichi prefecture, Japan. J Vet Med Sci 78(7):1195–1199. https://doi.org/10.1292/jvms.16-0041
Hulaj B, Goga I, Cana A, Merovci X, Rossi F, Crudele S, Ricchiuti L, Mutinelli F (2021) Passive surveillance of American Foulbrood in the Republic of Kosovo: geographic distribution and genotype characterization. J Apicult Res 60:1–7. https://doi.org/10.1080/00218839.2021.1892400
Kim M, Oh HS, Park SC, Chun J (2014) Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes. Int J Syst Evol Microbiol 64(2):346–351. https://doi.org/10.1099/ijs.0.059774-0
Koeuth T, Versalovic J, Lupski JR (1995) Differential subsequence conservation of interspersed repetitive Streptococcus pneumoniae BOX elements in diverse bacteria. Genome Res 5(4):408–418. https://doi.org/10.1101/gr.5.4.408
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Li W, Raoult D, Fournier PE (2009) Bacterial strain typing in the genomic era. FEMS Microbiology Reviews 33(5):892–916. https://doi.org/10.1111/j.1574-6976.2009.00182.x
Loncaric I, Derakhshifar I, Oberlerchner JT, Köglberger H, Moosbeckhofer R (2009) Genetic diversity among isolates of Paenibacillus larvae from Austria. J Invertebr Pathol 100(1):44–46. https://doi.org/10.1016/j.jip.2008.09.003
Matiašovic J, Bzdil J, Papežíková I, Čejková D, Vasina E, Bizos J, Navrátil S, Šedivá M, Klaudiny J, Pikula J (2023) Genomic analysis of Paenibacillus larvae isolates from the Czech Republic and the neighbouring regions of Slovakia. Res Vet Sci 158:34–40. Advance online publication. https://doi.org/10.1016/j.rvsc.2023.03.007
Morrissey BJ, Helgason T, Poppinga L, Fünfhaus A, Genersch E, Budge GE (2015) Biogeography of Paenibacillus larvae, the causative agent of American foulbrood, using a new multilocus sequence typing scheme. Environ Microbiol 17(4):1414–1424. https://doi.org/10.1111/1462-2920.12625
Neuendorf S, Hedtke K, Tangen G, Genersch E (2004) Biochemical characterization of different genotypes of Paenibacillus larvae subsp. larvae, a honey bee bacterial pathogen. Microbiol 150(7):2381–2390. https://doi.org/10.1099/mic.0.27125-0
OIE (2008) In Manual of diagnostic tests and vaccines for terrestrial animals (mammals, birds and bees), Chapter. 2.2.2. American Foulbrood. vol. 1(Sixth Edition). pp. 395–404
OIE (2018) Manual of diagnostic tests and vaccines for terrestrial animals. Chapter 3.2.2. American foulbrood of honey bees (infection of honey bees with Paenibacillus larvae)(version adopted in May 2016). pp. 719–735
Okamoto M, Furuya H, Sugimoto I, Kusumoto M, Takamatsu D (2022) A novel multiplex PCR assay to detect and distinguish between different types of Paenibacillus larvae and Melissococcus plutonius, and a survey of foulbrood pathogen contamination in Japanese honey. J Vet Med Sci 84(3):390–399. https://doi.org/10.1292/jvms.21-0629
Peters M, Kilwinski J, Beringhoff A, Reckling D, Genersch E (2006) American foulbrood of the honey bee: occurrence and distribution of different genotypes of Paenibacillus larvae in the administrative district of Arnsberg (North Rhine-Westphalia). J Vet Med B Infect Dis Vet Public Health 53(2):100–104. https://doi.org/10.1111/j.1439-0450.2006.00920.x
Rusenova N, Parvanov P, Stanilova S (2013) Molecular typing of Paenibacillus larvae strains isolated from Bulgarian apiaries based on repetitive element polymerase chain reaction (rep-PCR). Curr Microbiol 66(6):573–577. https://doi.org/10.1007/s00284-013-0318-5
Rusenova N, Parvanov P (2014) Biochemical profile of Paenibacillus larvae repetitive element polymerase chain reaction (rep-PCR) genotypes in Bulgaria. Kafkas Univ Vet Fak Derg 20(2):313–316. https://doi.org/10.9775/kvfd.2013.9853
Rusenova N, Parvanov P (2016) Prevalence of American Foulbrood and Paenibacillus larvae Genotypes in Bulgaria. İstanbul Üniv Vet Fak Derg 42(1):98–102. https://doi.org/10.16988/iuvfd.2016.16660
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schiesser A (2014) Determination of Paenibacillus larvae genotypes in Turkey and antimicrobial effect of different propolis against these genotypes. Hacettepe University Master Thesis. pp 125
Sevim E, Baş Y, Çelik G, Pınarbaş M, Bozdeveci A, Özdemir T, Akpınar R, Yaylı N, Alpay Karaoğlu Ş (2017) Antibacterial activity of bryophyte species against Paenibacillus larvae isolates. Turk J Vet Anim Sci 41(4):521–531. https://doi.org/10.3906/vet-1611-70
Shimanuki H, Knox DA (2000) Diagnosis of honey bee diseases. Agriculture Handbook No. AH690. United States Department of Agriculture, Beltsville
Todd R, Donoff RB, Kim Y, Wong DT (2001) From the chromosome to DNA: restriction fragment length polymorphism analysis and its clinical application. J Oral Maxil Surg 59(6):660–667. https://doi.org/10.1053/joms.2001.22707
Vos P, Hogers R, Bleeker M, Reijans M, Van De LT, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23(21):4407–4414. https://doi.org/10.1093/nar/23.21.4407
Welsh J, McClelland M (1990) Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acids Res 18(24):7213–7218. https://doi.org/10.1093/nar/18.24.7213
Williams JG, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 18(22):6531–6535. https://doi.org/10.1093/nar/18.22.6531
Žugelj A, Papić B, Zdovc I, Zajc U, Golob M, Avberšek J, Kušar D (2021) ERIC and WGS typing of Paenibacillus larvae in Slovenia: Investigation of ERIC I outbreaks. Insects 12(4):362. https://doi.org/10.3390/insects12040362
Funding
This study was financially funded by the Scientific Research Funding of the Recep Tayyip Erdogan University (Project Number: RTEU-BAP 2015.53001.102.03.04).
Author information
Authors and Affiliations
Contributions
Şengül Alpay Karaoglu: designed the experiments, analyzed the data, completed the final version manuscript, and provided funding acquisition. Arif Bozdeveci: performed the experiments, analyzed the data, and revised the final version of the manuscript. Müberra Pınarbaş Çetin: analyzed data and performed the experiments. Elif Sevim: designed the experiments, and performed the experiments. Şeyma Suyabatmaz: analyzed data and wrote the draft version of the manuscript. Rahşan Akpınar: collected sample materials and participated in the experiments. All authors have reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alpay Karaoğlu, Ş., Bozdeveci, A., Pinarbaş Çetin, M. et al. Isolation, characterization, and genetic diversity of Paenibacillus larvae from AFB suspected specimens in the Central and Eastern Black Sea Regions. Biologia 78, 2919–2929 (2023). https://doi.org/10.1007/s11756-023-01448-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01448-w