Abstract
Objectives
To control intraoperative hyperglycemia in patients who underwent aortic surgery using STG-55® artificial endocrine pancreas and clarify the effectiveness of this device.
Methods
Blood glucose control using the STG-55® was performed in 18 patients (15 men and 3 women; age, 66 ± 10 years) who required hypothermic circulatory arrest (STG-55® group). Seventeen patients (10 men and 7 women; age, 71 ± 8 years) whose blood glucose was controlled using the conventional method were included in the control group. Glucose concentration was controlled with the aim of maintaining it at 150 mg/dl.
Results
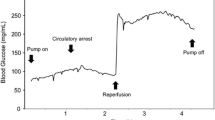
In both groups, the blood glucose concentrations did not significantly change during the interruption of systemic perfusion; however, a sharp increase was noted immediately after reperfusion. Although the hyperglycemic status persisted after reperfusion in the control group, it was effectively suppressed in the STG-55® group (STG® vs. control group at 50 min after reperfusion: 180 ± 35 vs. 212 ± 47 mg/dl, p = 0.026) and blood glucose concentration reached the target value of 150 mg/dl at 100 min after reperfusion (STG® vs. control group: 153 ± 29 vs. 215 ± 43 mg/dl, p = 0.0008). The total administered insulin dose was 175 ± 81 U and 5 ± 3 U in the STG® and control groups, respectively (p < 0.0001).
Conclusions
To treat the accelerated hyperglycemic status in aortic surgery requiring circulatory arrest, strict glycemic control using an artificial endocrine pancreas might be beneficial.


Similar content being viewed by others
References
van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, et al. Intensive insulin therapy in critically ill patients. N Engl J Med. 2001;345:1359–67.
Furnary AP, Gao G, Grunkemeier GL, Wu Y, Zerr KJ, Bookin SO, et al. Continuous insulin infusion reduces mortality in patients with diabetes undergoing coronary artery bypass grafting. J Thorac Cardiovasc Surg. 2003;125:1007–21.
Doenst T, Wijeysundera D, Karkouti K, Zechner C, Maganti M, Rao V, et al. Hyperglycemia during cardiopulmonary bypass is an independent risk factor for mortality in patients undergoing cardiac surgery. J Thorac Cardiovasc Surg. 2005;130:1144–50.
Desai SP, Henry LL, Holmes SD, Hunt SL, Martin CT, Hebsur S, et al. Strict versus liberal target range for perioperative glucose in patients undergoing coronary artery bypass grafting: a prospective randomized controlled trial. J Thorac Cardiovasc Surg. 2012;143:318–25.
Minakata K, Sakata R. Perioperative control of blood glucose level in cardiac surgery. Gen Thorac Cardiovasc Surg. 2013;61:61–6.
Navaratnarajah M, Rea R, Evans R, Gibson F, Antoniades C, Keiralla A, et al. Effect of glycaemic control on complications following cardiac surgery: literature review. J Cardiothorac Surg. 2018;13:10.
Kawahito K, Sato H, Kadosaki M, Egawa A, Misawa Y. Spike in glucose levels after reperfusion during aortic surgery: assessment by continuous blood glucose monitoring using artificial endocrine pancreas. Gen Thorac Cardiovasc Surg. 2018;66:150–4.
Tsukamoto Y, Okabayashi T, Hanazaki K. Progressive artificial endocrine pancreas: the era of novel perioperative blood glucose control for surgery. Surg Today. 2011;41:1344–51.
Namikawa T, Munekage M, Yatabe T, Kitagawa H, Hanazaki K. Current status and issues of the artificial pancreas: abridged English translation of a special issue in Japanese. J Artif Organs. 2018;21:132–7.
Kawahito S, Higuchi S, Mita N, Kitagawa T, Kitahata H. Novel blood sampling method of an artificial endocrine pancreas via the cardiopulmonary bypass circuit. J Artif Organs. 2013;16:508–9.
Yatabe T, Kitagawa H, Kawano T, Munekage M, Okabayashi T, Yamashita K, et al. Continuous monitoring of glucose levels in the hepatic vein and systemic circulation during the Pringle maneuver in beagles. J Artif Organs. 2011;14:232–7.
Ungerstedt J, Nowak G, Ungerstedt U, Ericzon BG. Microdialysis monitoring of porcine liver metabolism during warm ischemia with arterial and portal clamping. Liver Transpl. 2009;15:280–6.
The NICE-SUGAR Study Investigators. Hypoglycemia and risk of death in critically ill patients. N Engl J Med. 2012;367:1108–18.
Rangasamy V, Xu X, Susheela AT, Subramaniam B. Comparison of glycemic variability indices blood glucose risk index and coefficient of variation in predicting adverse outcomes for patients undergoing cardiac surgery. J Cardiothorac Vasc Anesth. 2020. https://doi.org/10.1053/j.jvca.2019.12.032.
Hanazaki K, Maeda H, Okabayashi T. Tight perioperative glycemic control using an artificial endocrine pancreas. Surg Today. 2010;40:1–7.
Uemura H, Sekiya N, Mitsuno M, Yamamura M, Tanaka H, Ryomoto M, et al. Usefulness of glycemic control using an artificial pancreas apparatus for cardiovascular surgery. ASAIO J. 2019;65:503–8.
Inzucchi SE, Siegel MD. Glucose control in the ICU–how tight is too tight? N Engl J Med. 2009;360:1346–9.
Srinivasan V, Spinella PC, Drott HR, Roth CL, Helfaer MA, Nadkarni V. Association of timing, duration, and intensity of hyperglycemia with intensive care unit mortality in critically ill children. Pediatr Crit Care Med. 2004;5:329–36.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aizawa, K., Muraoka, A., Kurumisawa, S. et al. Artificial endocrine pancreas with a closed-loop system effectively suppresses the accelerated hyperglycemic status after reperfusion during aortic surgery. Gen Thorac Cardiovasc Surg 69, 14–18 (2021). https://doi.org/10.1007/s11748-020-01415-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11748-020-01415-1