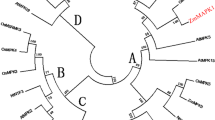
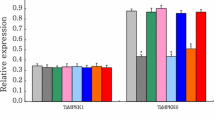
Abstract
Although plant MAPK cascades play decisive roles in stress responses, their contribution in wheat remains largely unknown. The bread wheat TMPK3 (wheat Mitogen Activated Protein Kinase) was previously recognized as a serine/threonine protein kinase carrying a Thr-Glu-Tyr (TEY) activation domain, but its functional role in environmental stress response is insufficiently studied. Here in, we showed that TMPK3 is auto-phosphorylated in vitro and can be phosphorylated by the constitutively active Arabidopsis AtMKK2. TMPK3 was also capable to phosphorylate its substrate, the MAP kinase phosphatase 1. Moreover, we investigated the involvement of TMPK3 in plant tolerance to salt/osmotic stresses. Our results show that TMPK3 complements the salt sensitivity of the Arabidopsis loss-of-function mpk3-1 mutant, where its overexpression promotes salt and osmotic stress tolerance to levels exceeding those observed in wild type plants. This tolerance is associated to a lower sensitivity to exogenous ABA, and increased stronger accumulation of proline contents, higher survival, and lower water loss rates as well as attenuated oxidative stress status. In summary, our data provide evidence that TMPK3 plays a positive role in salt and drought stress responses in plants.






Similar content being viewed by others
Data availability
Data are contained within the article or supplementary materials.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Asai T, Tena G, Plotnikova J, Willmann MR, Chiu WL, Gomez-Gomez L, Boller T, Ausubel FM, Sheen J (2002) MAP kinase signaling cascade in Arabidopsis innate immunity. Nature 415:977–983
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Beckers GJM, Jaskiewicz M, Liu Y, Underwood WR, He ShY, Zhang Sh, Conrath U (2009) Mitogen-activated protein kinases 3 and 6 are required for full priming of stress responses in Arabidopsis thaliana. Plant Cell 21(3):944–953
Bi G, Zhou Z, Wang W, Li RS, Wu S, Zhang H, Menke F, Chen LH, Zhou JM (2018) Receptor-like cytoplasmic kinases directly link diverse pattern recognition receptors to the activation of mitogen-activated protein kinase cascades in Arabidopsis. Plant Cell 18:458–462
Bigeard J, Hirt H (2018) Nuclear signaling of plant MAPKs. Front Plant Sci 9:469
Bigeard J, Colcombet J, Hirt H (2015) Signaling mechanisms in pattern-triggered immunity (PTI). Mol Plant 8:521–539
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein—dye binding. Anal Biochem 72:248–254
Cargnello M, Roux PP (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75:50–83
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Cui F, Sun W, Kong X (2018) RLCKs bridge plant immune receptors and MAPK cascades. Trends Plant Sci 23:1039–1041
Ding H, He J, Wu Y, Wu X, Ge C, Wang Y, Zhong S, Peiter E, Liang J, Xu W (2018) The tomato mitogen-activated protein kinase SlMPK1 is as a negative regulator of the high temperature stress response. Plant Physiol 177:633–651
Fujii H, Zhu JK (2012) Osmotic stress signaling via protein kinases. Cell Mol Life Sci 69:3165–3173
Gao M, Liu J, Bi D, Zhang Z, Cheng F, Chen S, Zhang Y (2008) MEKK1, MKK1/MKK2 and MPK4 function together in a mitogen-activated protein kinase cascade to regulate innate immunity in plants. Cell Res 18:1190–1198
Ghorbel M, Zaidi I, Ebel C, Hanin M (2019) Differential regulation of the durum wheat MAPK phosphatase 1 by calmodulin, bivalent cations and possibly mitogen activated protein kinase 3. Plant Physiol Biochem 135:242–252
Hanks SK (2003) Genomic analysis of the eukaryotic protein kinase superfamily: a perspective. Genome Biol 4(5):111
Ishihama N, Yamada R, Yoshioka M, Katou S, Yoshioka H (2011) Phosphorylation of the Nicotiana benthamiana WRKY8 transcription factor by MAPK functions in the defense response. Plant Cell 23:1153–1170
Kim HS, Park SC, Ji CY, Park S, Jeong JC, Lee HS, Kwak SS (2016) Molecular characterization of biotic and abiotic stress-responsive MAP kinase genes, IbMPK3 and IbMPK6, in sweet potato. Plant Physiol Biochem 108:37–48
Li Y, Cai H, Liu P, Wang C, Gao H, Wu C, Yan K, Zhang S, Huang J, Zheng C (2017) Arabidopsis MAPKKK18 positively regulates drought stress resistance via downstream MAPKK3. Biochem Biophys Res Commun 484:292–297
Liu XM, Nguyen XC, Kim KE, Han HJ, Yoo J, Lee K, Kim MC, Yun DJ, Chung WS (2013) Phosphorylation of the zinc finger transcriptional regulator ZAT6 by MPK6 regulates Arabidopsis seed germination under salt and osmotic stress. Biochem Biophys Res Commun 430:1054–1059
Livak KJ, Schmittgen ThD (2001) Analysis of relative gene expression data using real time quantitative PCR and the 22DDCT method. Methods 25:402–408
Ma Q, Xia Z, Cai Z, Li L, Cheng Y, Liu J, Nian H (2019) GmWRKY16 enhances drought and salt tolerance through an ABA-mediated pathway in Arabidopsis thaliana. Front Plant Sci 9:1979
Marín-Aguilar F, Pavillard LE, Giampieri F, Bullón P, Cordero MD (2017) Adenosine monophosphate (AMP)-activated protein kinase: a new target for nutraceutical compounds. Int J Mol Sci 18:288
McKinney EC, All N, Traut A, Feldmann KA, Belostotsky DA, McDowell JM, Meegher RB (1995) Sequence-based identification of T-DNA insertion mutations in Arabidopsis actin mutants act2-1 and act4-1. Plant J 8(4):613–622
Mohanta TK, Arora PK, Mohanta N, Parida P, Bae H (2015) Identification of new members of the MAPK gene family in plants shows diverse conserved domains and novel activation loop variants. BMC Genom 16:314–318
Nickel RS, Cunningham BA (1969) Improved peroxidase assay method usingleuco-2,3_,6-trichloroindophenoland application to comparative measurements of peroxidase catalysis. Anal Biochem 27:292–299
Persak H, Pitzschke A (2013) Tight interconnection and multi-level control of Arabidopsis MYB44 in MAPK cascade signaling. PLoS ONE 8:e57547
Peti W, Page R (2013) Molecular basis of MAP kinase regulation. Protein Sci 22:1698–1710
Pitzschke A, Djamei A, Bitton F, Hirt H (2009) A major role of the MEKK1–MKK1/2-MPK4 pathway in ROS signaling. Mol Plant 2:120–137
Pitzschke A, Datta S, Persak H (2014) Salt stress in Arabidopsis: lipid transfer protein AZI1 and its control by mitogen-activated protein kinase MPK3. Mol Plant 4:722–738
Ramegowda V, Gill US, Sivalingam PN, Gupta A, Gupta C, Govind G, Nataraja KN, Pereira A, Udayakumar M, Mysore KS, Senthil-Kumar M (2017) GBF3 transcription factor imparts drought tolerance in Arabidopsis thaliana. Sci Rep 7:9148
Ranty B, Aldon D, Galaud JP (2006) Plant calmodulins and calmodulin-related proteins. Multifaceted relays to decode calcium signals. Plant Signal Behav 1:36–39
Rodriguez MC, Petersen M, Mundy J (2010) Mitogen-activated protein kinase signaling in plants. Annu Rev Plant Biol 61:621–649
Šamajová O, Plíhal O, Al-Yousif M, Hirt H, Šamaj J (2013) Improvement of stress tolerance in plants by genetic manipulation of mitogen-activated protein kinases. Biotechnol Adv 31:118–128
Sayed MA, Schumann H, Pillen K, Naz AA, Leon J (2012) AB-QTL analysis reveals new alleles associated to proline accumulation and leaf wilting under drought stress conditions in barley (Hordeum vulgare L.). BMC Genet 13:61–65
Schweighofer A, Kazanaviciute V, Scheikl E, Teige M, Doczi R, Hirt H (2007) The PP2C-type phosphatase AP2C1, which negatively regulates MPK4 and MPK6, modulates innate immunity, jasmonic acid, and ethylene levels in Arabidopsis. Plant Cell 19:2213–2224
Shi J, Fu XZ, Peng T, Huang XS, Fan QJ, Liu JH (2010) Spermine pretreatment confers dehydration tolerance of citrus in vitro plants via modulation of antioxidative capacity and stomatal response. Tree Physiol 30:914–922
Takezawa D (1999) Elicitor- and A23187-induced expression of WCK-1, a gene encoding mitogen-activated protein kinase in wheat. Plant Mol Biol 40:921–933
Teige M, Scheikl E, Eulgem Th et al (2004) The MKK2 pathway mediates cold and salt stress signaling in Arabidopsis. Mol Cell 15(1):141–152
Verbruggen N, Herman Ch (2008) Proline accumulation in plants: a review. Amino Acids 35(4):753–9
Wei-wei L, Yi-miao T, Shi-qing G, Zhao Z, Xi Z, Chang-ping Z (2012) Phylogenetic analysis and expression patterns of the MAPK gene family in wheat (Triticum aestivum L.). J Integr Agric 11:1227–1235
Yan H, Zhao Y, Shi H, Li J, Wang Y, Tang D (2018) BRASSINOSTEROID-SIGNALING KINASE1 phosphorylates MAPKKK5 to regulate immunity in Arabidopsis. Plant Physiol 176:2991–3002
Yu L, Nie J, Cao C, Jin Y, Yan M, Wang F, Liu J, Xiao Y, Liang Y, Zhang W (2010) Phosphatidic acid mediates salt stress response by regulation of MPK6 in Arabidopsis thaliana. New Phytol 188:762–773
Yu L, Yan J, Yang Y, Zhu W (2015) Overexpression of tomato mitogen-activated protein kinase SlMPK3 in tobacco increases tolerance to low temperature stress. Plant Cell Tissue Organ Cult 121:21–34
Zaidi I, Ebel C, Touzri M, Herzog E, Evrard JL, Schmit AC, Masmoudi K, Hanin M (2010) TMKP1 is a novel wheat stress responsive MAP kinase phosphatase localized in the nucleus. Plant Mol Biol 73:325–338
Acknowledgements
This study was supported by a grant from Ministry of Higher Education, and Scientific Research of Tunisia.
Funding
This work was supported by the core funding provided by the Ministry of Higher Education and Scientific Research, Tunisia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statements
Seeds of Arabidopsis mpk3-1 mutant line (SALK_151594) were kindly provided by Prof. Roman Ulm from the University of Geneva, Switzerland.
Additional information
Communicated by E. Kuzniak-Gebarowska.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplemental Fig. 1. TMPK3 shares high homology with other plant MPKs.
Amino acid sequence alignments of Triticum aestivum TMPK3 (known also as WCK-1, accession no. AAC28850), Arabidopsis thaliana AtMPK1 (AT1G10210.2); AtMPK2 (AT1G59580); AtMPK3 (At1G43700); AtMPK14 (At4G36450); tobacco NtMPK14 (Genbank accession number X69971); and Zea mays ZmMPK3 (GenBank accession number: EU130900). The eleven conserved subdomains and the TEY domain are indicated by rectangles. The common docking domain (CD) is underlined, and the consensus indicated. (PPTX 89 KB)
Supplemental Fig. 2. Phosphorylation of the dead phosphatase TMKP1
C214G by GST-TMPK3. The phosphorylation assay was performed in the absence (A) and in the presence (B) of AtMKK2 as indicated. The dead phosphatase is not phosphorylated by GST-AtMKK2 as shown in (C). The positions of GST-MPK3, GST-AtMKK2 and His-TMKP1C214G on the autoradiogram are indicated. (PPTX 838 KB)
Supplemental Fig. 3. PCR analysis of TMPK3 transgenic lines.
(A) Physical map of the pCAMBIA 1302-TMPK3 construct used for Arabidopsis transformation. (B) TMPK3 transgene PCR amplifications were performed using primers indicated on panel A, and DNA from the four transgenic lines generated in WT (L11, L12) or in mpk3-1 mutant background (L3 and L8). Position of the PCR products with expected size (1100 bp) is indicated by an arrow on the right side of the gel. DNA from WT and from mpk3-1 mutant were also used. M: marker, (-) indicates the control for PCR, where H2O was used instead of DNA. (+) indicates the positive control using the plasmid pCAMBIA 1302-TMPK3. (C) QRT-PCR analysis of TMPK3 expression in WT, TMPK3-transgenic lines (L11 and L12), mpk3-1 mutants and mpk3-1 35S::TMPK3 transgenic lines (L3 and L8). (PPTX 254 KB)
Supplemental Fig. 4.
Immunoblot analysis of TMPK3 in the four T3 homozygous lines (L11, L12, L3 and L8). The anti-phospho-p44/42 MAPK antibody allows detection of activated MPKs (pMPK3 and pMPK6). Protein extracts from WT and mpk3-1 mutant were used as controls. Equal loading is indicated by a replicate blot probed with anti-actin antibody. Positions of molecular weight markers are shown on the left side. (B) Comparison of transgenic and wild-type plants exposed to salt and osmotic stress. Seeds were placed on MS medium containing 200 mM Mannitol and 150 mM NaCl. Plants were photographed 15 days after seeds germination. (PPTX 555 KB)
Supplemental Fig. 5.
Statistical analysis using one-way ANOVA test comparison; (p< 0.05) relative to WT control for (A) Survival rate and (B) Fresh weight in presence of 150 mM NaCl; (C) Proline and (D) MDA. (PPTX 39 KB)
Supplemental Fig. 6.
Statistical analysis using one-way ANOVA test comparison; (p< 0.05) relative to WT control for (A) SOD, (B) POD, and (C) CAT. (PPTX 37 KB)
Supplemental Fig. 7.
Statistical analysis using one-way ANOVA test comparison; (p< 0.05) relative to WT control for (A) plants treated by 3 µM ABA and (B) plants treated by 5 µM ABA. (PPTX 37 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghorbel, M., Zaidi, I., Ebel, C. et al. The wheat Mitogen Activated Protein Kinase TMPK3 plays a positive role in salt and osmotic stress response. Acta Physiol Plant 45, 71 (2023). https://doi.org/10.1007/s11738-023-03548-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-023-03548-1