Abstract
Purpose
Basic science research has shown that obesity is associated with microvascular endothelial dysfunction. However, whether bariatric surgery impacts the microvascular networks has yet to be explored. This study sought to evaluate the impact of gastric bypass in the retinal microvasculature.
Methods
Patients with obesity (BMI ≥ 35 kg/m2) scheduled to gastric bypass were consecutively recruited and included in the study. Patients were evaluated before surgery and 6–12 months after the intervention. Macular microvascular properties were evaluated using optical coherence tomography (OCT) angiography. Foveal avascular zone area, perimeter, circularity, and foveal and perifoveal vascular density (in both superficial and deep vascular plexus) were computed.
Results
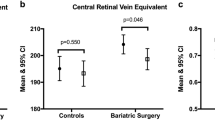
In total, 40 eyes from 20 patients were included (30% male, mean BMI 43.4 ± 4.5 kg/m2 (range 35.7–51.4). From these, 45% were diabetic before bariatric surgery. After surgery, there was a significant increase in foveal avascular zone circularity (from 0.85 ± 0.09 to 0.92 ± 0.07, p = 0.001) and vascular density in perifoveal deep vascular plexus (from 0.69 ± 0.12 to 0.73 ± 0.12; p = 0.04), whereas foveal avascular zone perimeter decreased (from 2.34 ± 0.37 to 2.20 ± 0.35 mm, p = 0.007). Preoperative diabetic status was not a predictor of microvascular retinal changes after bariatric surgery. However, after multivariate adjustments, the increased drop in HbA1c after the surgery remained associated with the increase in perifoveal vascular density in the deep vascular plexus (B = 0.05; 95% CI 0.05–0.10; p = 0.03).
Conclusions
Gastric bypass improves retinal microvascular perfusion as demonstrated by the increased parafoveal vascular density in the deep vascular plexus, increased foveal avascular zone circularity, and decreased foveal avascular zone perimeter.


Similar content being viewed by others
Abbreviations
- BMI:
-
Body mass index
- CKD-EPI:
-
Chronic Kidney Disease Epidemiology Collaboration
- Cr:
-
Creatinine
- GFR:
-
Glomerular filtration rate
- HbA1c:
-
Glycosylated hemoglobin
- OCT:
-
Optical coherence tomography
- OCTA:
-
Optical coherence tomography angiography
- OSA:
-
Obstructive sleep apnea
References
WHO. Obesity and overweight - key facts. 2020. Available from: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight.
Csige I, Ujvárosy D, Szabó Z, et al. The impact of obesity on the cardiovascular system. J Diabetes Res. 2018;2018:1–12.
Wolfe BM, Kvach E, Eckel RH. Treatment of obesity: weight loss and bariatric surgery. Circ Res. 2016;118:1844–55. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27230645%0A. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=PMC4888907
Caceres BA, Moskowitz D, O’Connell T. A review of the safety and efficacy of bariatric surgery in adults over the age of 60: 2002-2013. J Am Assoc Nurse Pract. 2015;27:403–10.
Park CH, Nam SJ, Choi HS, et al. Comparative efficacy of bariatric surgery in the treatment of morbid obesity and diabetes mellitus: a systematic review and network meta-analysis. Obes Surg. 2019;29:2180–90.
Arterburn DE, Courcoulas AP. Bariatric surgery for obesity and metabolic conditions in adults. BMJ. 2014;349:1–11.
Phillips BT, Shikora SA. The history of metabolic and bariatric surgery: development of standards for patient safety and efficacy. Metabolism. Elsevier Inc.; 2018;79:97–107. Available from: https://doi.org/10.1016/j.metabol.2017.12.010
Fried M, Ribaric G, Buchwald JN, et al. Metabolic surgery for the treatment of type 2 diabetes in patients with BMI. Obes Surg. Diabetologia. 2010;20:776–90. https://doi.org/10.1007/s11695-010-0113-3.
Karaca Ü, Schram MT, Houben AJHM, et al. Microvascular dysfunction as a link between obesity, insulin resistance and hypertension. Diabetes Res Clin Pract. Elsevier Ireland Ltd. 2014;103:382–7. https://doi.org/10.1016/j.diabres.2013.12.012.
Campia U, Tesauro M, Cardillo C. Human obesity and endothelium-dependent responsiveness. Br J Pharmacol. 2012;165:561–73.
Virdis A, Masi S, Colucci R, et al. Microvascular endothelial dysfunction in patients with obesity. Curr Hypertens Rep. 2019;21:1–7.
Rodríguez F, Staurenghi G, … RG-GA for, 2018 undefined. The role of OCT-A in retinal disease management. Springer. Graefe’s Archive for Clinical and Experimental Ophthalmology; 2018. Available from: https://doi.org/10.1007/s00417-018-4109-3
Gildea D. The diagnostic value of optical coherence tomography angiography in diabetic retinopathy: a systematic review. Int Ophthalmol. Springer Netherlands. 2019;39:2413–33. https://doi.org/10.1007/s10792-018-1034-8.
Tan ACS, Tan GS, Denniston AK, et al. An overview of the clinical applications of optical coherence tomography angiography. Eye. 2018 [cited 2018 Jun 24];32:262–86. Available from: http://www.ncbi.nlm.nih.gov/pubmed/28885606.
Tey KY, Teo K, Tan ACS, et al. Optical coherence tomography angiography in diabetic retinopathy: a review of current applications. Eye Vis. 2019;6:1–10.
Kashani AH, Chen CL, Gahm JK, et al. Optical coherence tomography angiography: a comprehensive review of current methods and clinical applications. Prog Retin Eye Res. Elsevier Ltd. 2017;60:66–100. https://doi.org/10.1016/j.preteyeres.2017.07.002.
Raman M, Middleton RJ, Kalra PA, et al. Estimating renal function in old people: an in-depth review. Int Urol Nephrol. Springer Netherlands. 2017;49:1979–88.
Rasband WS. ImageJ, U. S. National Institutes of Health, Bethesda, Maryland, USA. 1997–2018. Available from: https://imagej.nih.gov/ij/.
Yao X, Alam MN, Le D, et al. Quantitative optical coherence tomography angiography: a review. Exp Biol Med. 2020;245:301–12.
Kim K, Kim ES, Yu SY. Optical coherence tomography angiography analysis of foveal microvascular changes and inner retinal layer thinning in patients with diabetes. Br J Ophthalmol. 2018;102:1226–31.
Vujosevic S, Toma C, Villani E, et al. Diabetic macular edema with neuroretinal detachment: OCT and OCT-angiography biomarkers of treatment response to anti-VEGF and steroids. Acta Diabetol. 2019 [cited 2019 Oct 29];57:287–96. https://doi.org/10.1007/s00592-019-01424-4.
Stehouwer CDA. Microvascular dysfunction and hyperglycemia: a vicious cycle with widespread consequences. Diabetes. 2018;67:1729–41.
De Jongh RT, Serné EH, Ijzerman RG, et al. Impaired microvascular function in obesity: implications for obesity-associated microangiopathy, hypertension, and insulin resistance. Circulation. 2004;109:2529–35.
Jackson S, le Roux CW, Docherty NG. Bariatric surgery and microvascular complications of type 2 diabetes mellitus. Curr Atheroscler Rep. 2014;16:1–9.
Gryglewska B, Głuszewska A, Zarzycki B, et al. Post-occlusive reactive hyperemic response of skin microcirculation among extremely obese patients in the short and long term after bariatric surgery. Microcirculation. 2019:0–1.
Ministrini S, Fattori C, Ricci MA, et al. Microcirculatory improvement induced by laparoscopic sleeve gastrectomy is related to insulin sensitivity retrieval. Obes Surg. 2018;28:3151–8.
Rossi M, Nannipieri M, Anselmino M, et al. Skin vasodilator function and vasomotion in patients with morbid obesity: effects of gastric bypass surgery. Obes Surg. 2011;21:87–94.
Martín-Rodríguez JF, Cervera-Barajas A, Madrazo-Atutxa A, et al. Effect of bariatric surgery on microvascular dysfunction associated to metabolic syndrome: a 12-month prospective study. Int J Obes. 2014;38:1410–5.
Aghamohammadzadeh R, Greenstein AS, Yadav R, et al. Effects of bariatric surgery on human small artery function: evidence for reduction in perivascular adipocyte inflammation, and the restoration of normal anticontractile activity despite persistent obesity. J Am Coll Cardiol. 2013;62:128–35.
De Ciuceis C, Rossini C, Porteri E, et al. Circulating endothelial progenitor cells, microvascular density and fibrosis in obesity before and after bariatric surgery. Blood Press. 2013;22:165–72.
De Ciuceis C, Porteri E, Rizzoni D, et al. Effects of weight loss on structural and functional alterations of subcutaneous small arteries in obese patients. Hypertension. 2011;58:29–36.
Dogan B, Dogan U, Erol MK, et al. Optical coherence tomography parameters in morbidly obese patients who underwent laparoscopic sleeve gastrectomy. J Ophthalmol. Hindawi Limited; 2016 [cited 2018 may 20];2016:5302368. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27413543.
Posarelli C, Salvetti G, Piaggi P, et al. Ophthalmologic evaluation of severely obese patients undergoing bariatric surgery: a pilot, monocentric, prospective, open-label study. PLoS One. 2019;14:1–11.
Brynskov T, Laugesen CS, Floyd AK, et al. Thickening of inner retinal layers in the parafovea after bariatric surgery in patients with type 2 diabetes. Acta Ophthalmol. 2016;94:668–74.
Carlsson LMS, Sjöholm K, Karlsson C, et al. Long-term incidence of microvascular disease after bariatric surgery or usual care in patients with obesity, stratified by baseline glycaemic status: a post-hoc analysis of participants from the Swedish Obese Subjects study. Lancet Diabetes Endocrinol. Elsevier Ltd; 2017;5:271–9. Available from: https://doi.org/10.1016/S2213-8587(17)30061-X
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval and Informed Consent
All procedures were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Laiginhas, R., Guimarães, M., Nora, M. et al. Gastric Bypass Improves Microvascular Perfusion in Patients with Obesity. OBES SURG 31, 2080–2086 (2021). https://doi.org/10.1007/s11695-021-05223-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-021-05223-1