Abstract
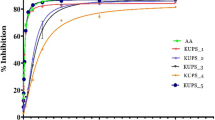
This research aimed to extract and profile phenolic compounds from seed and fruit of Diospyros lotus, evaluate and compare the antioxidants and cytotoxic potential of the extracts. Seven phenolic acids and four other flavonoids extracts were subsequently obtained following different extraction methods. The extracts were evaporated, lyophilized, and prepared for RP-HPLC-DAD, MTT assay, and antioxidant evaluation by the ferrous ion chelating, ferric ion reducing power, FRAP, DPPH•, •OH and NO• scavenging methods. The RP-HPLC-DAD revealed an epigallocatechin, a flavonoid, and fourteen phenolic acids. Phenolic compounds with the highest concentrations are epigallocatechin (1460.80 ± 10.74 µg/gDW), gallic acid (1167.11 ± 4.04 µg/gDW) and t-cinnamic acid (244.45 ± 3.61 µg/gDW). The bound phenolic acids from acid hydrolysis-1 (BPAH-1) extract of the fruit sample exhibit the highest DPPH• activity, reducing power, and FRAP. The flavonol extract of the fruit sample unveils the highest Fe2+ chelating effect. The BPAH-1 and BPAH-2 extracts of the seed reveal the highest •OH and NO• activities, respectively. Seed sample gave the highest total phenolic/flavonoid/tannin contents. The cytotoxicity of the extracts on HepG2 cell lines > HeLa cell lines and the effects are more pronounced 48 h after treatments. Specifically, flavan-3-ol methanolic and BPAH-2 extracts of the seed, and flavan-3-ol methanolic and BPAH-1 extracts of the fruit show good cytotoxic effect against HepG2 cell lines (IC50 < 37 µg/mL). This research unveils the potential of D. lotus seed and fruit as possible antioxidant and anticancer agent. The antioxidant and cytotoxicity properties observed from the samples might be due to the phenolic compounds determined.

Graphical abstract



Similar content being viewed by others
References
M. Naeem, I.S. Sheikh, M. Rafiq, J. Ahmad, H. Batool, M. Khan, N. Khan, H.K. Baloch, I. Saba, Pak-Euro J. Med. Life Sci. 5, 257 (2022)
M. Sołtan, D. Bartusik-Aebisher, D. Aebisher, Front. Bioeng. Biotechnol. 10, (2022)
A.J. Doman, S. Tommasi, M.V. Perkins, R.A. McKinnon, A.A. Mangoni, P.C. Nair, Bioorg. Med. Chem., 116970 (2022)
H.A. Hussein, M.A. Abdullah, Mar. Drugs 18, 356 (2020)
A. Ashok, S.S. Andrabi, S. Mansoor, Y. Kuang, B.K. Kwon, V. Labhasetwar, Antioxidants 11, 408 (2022)
C.M. Ma, J.R. Zhao, F.F. Wu, Q. Zhang, X.H. Zhao, J. Food Meas. Charact. 16, 114 (2022)
A.N. Panche, A.D. Diwan, S.R. Chandra, J. Nutr. Sci. 5, e47 (2016)
J. Chen, J. Yang, L. Ma, J. Li, N. Shahzad, C.K. Kim, Sci. Rep. 10, 1 (2020)
R. Dhalaria, R. Verma, D. Kumar, S. Puri, A. Tapwal, V. Kumar, E. Nepovimova, K. Kuca, Antioxidants 9, 1123 (2020)
G.R. Velderrain-Rodríguez, H. Palafox-Carlos, A. Wall-Medrano, J.F. Ayala-Zavala, C.Y.O. Chen, M. Robles-Sánchez, H. Astiazaran-García, E. Alvarez-Parrilla, G.A. González-Aguilar, Food Funct. 5, 189 (2014)
A. Rauf, G. Uddin, B.S. Siddiqui, A. Khan, U. Farooq, F.A. Khan, S.M. Bukhari, S.B. Khan, Chin. J. Nat. Med. 15, 865 (2017)
K. Gasimova, E. Breman, A. Faruk, V.M. Ali-zade, Plant. Fungal Res. 2, 52 (2020)
E. Aydin, Gıda 46, 669 (2021)
B.O. Cho, H.H. Yin, C.Z. Fang, S.J. Kim, S.I. Jeong, S.I. Jang, Food Sci. Biotechnol. 24, 2205 (2015)
Z. Akar, A. Karakurt, F. Okumuş, S. Cinemre, A. Düzgün, B. Akar, Z. Can, Int. J. Second Metab. 7, 237 (2020)
Z.T. Murathan, Acta Sci. Pol. Hortorum Cultus 19, 49 (2020)
A.M.A. Hassan, O. Zannou, H. Pashazadeh, A. Ali Redha, I. Koca, J. Food Sci. (2022)
Y. Hu, H. Yan, Y. Yin, X. Li, H. Li, LWT 154, 112642 (2022)
N.B. Türkmen, D.A. Özek, A. Taşlidere, O. Çiftçi, Ö Saral, C.C. Gül, Turkish J. Pharm. Sci. 19, 132 (2022)
A.H. Moghaddam, S.M. Nabavi, S.F. Nabavi, R. Bigdellou, S. Mohammadzadeh, M.A. Ebrahimzadeh, Acta Pol. Pharm. - Drug Res. 69, 687 (2012)
P. Valentão, P.B. Andrade, F. Areias, F. Ferreres, R.M. Seabra, J. Agric. Food Chem. 47, 4579 (1999)
M. Olszewska, J. Pharm. Biomed. Anal. 48, 629 (2008)
A. de Villiers, F. Lynen, A. Crouch, P. Sandra, Chromatographia 59, 403 (2004)
K. Kim, R. Tsao, R. Yang, S. Cui, Food Chem. 95, 466 (2006)
V.L. Singleton, R. Orthofer, R.M. Lamuela-Raventós, Methods Enzymol (Elsevier, Amsterdam, 1999), pp. 152–178
L.E. Dowd, Anal. Chem. 31, 1184 (1959)
R.B. Broadhurst, W.T. Jones, J. Sci. Food Agric. 29, 788 (1978)
W. Brand-Williams, M.E. Cuvelier, C. Berset, LWT - Food Sci. Technol. 28, 25 (1995)
B. Halliwell, J.M.C. Gutteridge, O.I. Aruoma, Anal. Biochem. 165, 215 (1987)
M.N.A. Rao, J. Pharm. Pharmacol. 49, 105 (1997)
T.C.P. Dinis, V.M.C. Madeira, L.M. Almeida, Arch. Biochem. Biophys. 315, 161 (1994)
M. Oyaizu, Japanese J. Nutr. Diet. 44, 307 (1986)
K. Thaipong, U. Boonprakob, K. Crosby, L. Cisneros-Zevallos, D.H. Byrne, J. Food Compos. Anal. 19, 669 (2006)
T. Mosmann, J. Immunol. Methods 65, 55 (1983)
P.S. Kaparekar, S.K. Anandasadagopan, Curr. Pharmacol. Reports, 1 (2022)
P.J. White, Y. Xing, Antioxidants from Cereals and Legumes (AOCS Press, Champaign, Illinois, 1997), pp. 25–63
S. Kaur, M.K. Samota, M. Choudhary, M. Choudhary, A.K. Pandey, A. Sharma, J. Thakur, Physiol. Mol. Biol. Plants 1 (2022)
S. Kumar, A.K. Pandey, Sci. World J. 2013, 1 (2013)
V.M. Patil, N. Masand, Stud. Nat. Prod. Chem (Elsevier, Amsterdam, 2018), pp. 401–430
J. Côté, S. Caillet, G. Doyon, J.F. Sylvain, M. Lacroix, Crit. Rev. Food Sci. Nutr. 50, 666 (2010)
C. Chu, J. Deng, Y. Man, Y. Qu, Biomed. Res. Int. 2017, 1 (2017)
H. Slika, H. Mansour, N. Wehbe, S.A. Nasser, R. Iratni, G. Nasrallah, A. Shaito, T. Ghaddar, F. Kobeissy, A.H. Eid, Biomed. Pharmacother 146, 112442 (2022)
F. Ahmed, M. Iqbal, Org. Med. Chem. Int. J. 5, 107 (2018)
M. Safari, S. Ahmady-Asbchin, Biotechnol. Biotechnol. Equip. 33, 372 (2019)
J.A. Jeong, S.H. Kwon, Y.J. Kim, C.S. Shin, C.H. Lee, Korean J. Plant. Resour. 20, 177 (2007)
H. Gao, N. Cheng, J. Zhou, B. Wang, J. Deng, W. Cao, J. Food Sci. Technol. 51, 950 (2014)
F. Chen, B. Wang, G. Zhao, X. Liang, S. Liu, J. Liu, J. Food Meas. Charact. 1 (2022)
M.I.N. Manik, M.H. Ali, M.M. Islam, A. Zobayed, A. Khan, Biomed. Pharmacol. J. 15, 911 (2022)
S.M. Nabavi, M.A. Ebrahimzadeh, S.F. Nabavi, M. Fazelian, B. Eslami, Pharmacogn. Mag. 5, 122 (2009)
F. Akdeniz, İ Dursun, K. Tepebaş, H. Özbay, L. Kekeç, S. Yildiz, Turkish J. Anal. Chem. 3, 64 (2021)
M. Rudrapal, S. Maji, S.K. Prajapati, P. Kesharwani, P.K. Deb, J. Khan, R. Mohamed, R.S. Ismail, R.K. Kankate, S.J. Sahoo, Khairnar, Antioxidants 11, 1217 (2022)
X. Li, Q. Zhou, A.M.M. Japir, D. Dutta, N. Lu, Z. Ge, ACS Nano 16, 14982 (2022)
L. Marcocci, J.J. Maguire, M.T. Droylefaix, L. Packer, Biochem. Biophys. Res. Commun. 201, 748 (1994)
L. Marcocci, L. Packer, M.T. Droy-Lefaix, A. Sekaki, M. Gardès-Albert, Methods Enzymol. (Elsevier, Amsterdam, 1994), pp. 462–475
A.A. Shahat, A.Y. Ibrahim, M.S. Alsaid, Indian J. Tradit Knowl. 1, 28 (2015)
A. Grzelakowska, J. Modrzejewska, J. Kolińska, M. Szala, M. Zielonka, K. Dębowska, M. Zakłos-Szyda, A. Sikora, J. Zielonka, R. Podsiadły, Free Radic. Biol. Med. 179, 34 (2022)
J.P. Adjimani, P. Asare, Toxicol Rep. 2, 721 (2015)
V.T. Aparadh, V.V. Naik, B.A. Karadge, Ann. Di Bot. 2, 49 (2012)
M.R. Loizzo, A. Said, R. Tundis, U.W. Hawas, K. Rashed, F. Menichini, N.G. Frega, F. Menichini, Plant. Foods Hum. Nutr. 64, 264 (2009)
F. Benchikh, H. Benabdallah, H. Amira, I. Amira, W. Mamache, S. Amira, Turkish J. Agric. Sci. Technol. 10, 1089 (2022)
M.A. Ebrahimzadeh, S. Eslami, S.M. Nabavi, S.F. Nabavi, B. Eslami, Biotechnol. Biotechnol. Equip. 24, 2127 (2010)
İ Gülçin, Z. Bingöl, P. Taslimi, A.C. Gören, S.H. Alwasel, A.Z. Tel, Chem. Biodivers. 19, e202100775 (2022)
P.W. Harlina, M. Ma, R. Shahzad, I. Khalifa, Food Sci. Anim. Resour. 42, 689 (2022)
L.P. Kose, İ Gulcin, Molecules 26, (2021)
S. Kamiloglu, G. Sari, T. Ozdal, E. Capanoglu, Food Front. 1, 332 (2020)
Z. Baharum, A.M. Akim, Y.H. Taufiq-Yap, R.A. Hamid, R. Kasran, Molecules 19, 18317 (2014)
E. Puspitasari, D.A. Pangaribowo, I.Y. Isparnaning, Y. Utami, in Proceeding 1st Univ. Muhammadiyah Purwokerto - Pharm. Int. Conf., ed. By D.D. Asmiyenti, H. Dwi, I.U. Pri (Faculty of Pharmacy University of Muhammadiyah Purwokerto, Central Java, 2015), pp. 82–86
X. Yue, H. Yang, T. Wang, S. Dong, Z. Sun, Y. Wang, X. Luo, B. Chen, G. Yao, Y. Gao, C. Lv, D. Zheng, Y. Zhao, T. Wang, S. Yan, W. Peng, Therm. Sci. 24, 1705 (2020)
Acknowledgements
In this study, Umar Muazu Yunusa was supported with scholarship by the Presidency for Turks Abroad and Related Communities and the scientific research project coordination unit of Dokuz Eylül University for project number 2019.KB.FEN.010.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors have no competing interests to declare that are relevant to the content of this article.
Research involving human participants and/or animals.
Not applicable.
Informed consent
All authors read the manuscript and given their consent for publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yunusa, U.M., Urek, R.O. Determination of phenolic compounds in Diospyros lotus by RP-HPLC-DAD and evaluation of antioxidant and cytotoxic properties. Food Measure 17, 1413–1427 (2023). https://doi.org/10.1007/s11694-022-01696-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-022-01696-5