Abstract
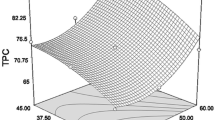
Fig fruits are widely known for its potential bioactivities but still many wild species were not phytochemically explored. The optimization of extracting conditions for these phytochemical could be useful to commercialize these wild fruits. In this study, ethanol concentration, extracting time and temperature was optimized for extracting Ficus racemosa polyphenols using response surface methodology. Central composite design was employed to investigate the effects of factors on responses such as, total phenolic content (TPC), total flavonoid content (TFC) and 2,2-diphenyl-1-picryl hydrazyl radical scavenging activity (DPPH). Based on the results 40% of ethanol concentration with 42.50 °C as extracting temperature for 80 min of extraction was optimized for extracting F. racemosa polyphenolic compounds. From the optimized conditions TPC (80.10 ± 2.51 mg GAE/g extract), TFC (79.31 ± 5.11 mg RE/g extract) were significantly found with the inhibition of 51.20 ± 3.21% on DPPH radical. The proposed model confirmed the extracting ability of phenolics and flavonoids from fruit and implies this optimization is efficient.



Similar content being viewed by others
References
S. Saravanan, T. Parimelazhagan, Food Sci. Hum. Wellness 3, 56–64 (2014)
O.M. Mosquera, Y.M. Correa, D.C. Buitrago, J. Niño, Mem. Inst. 102, 631–634 (2007)
H.E. Seifried, D.E. Anderson, E.I. Fisher, J.A. Milner, J. Nutr. Biochem. 18, 567–579 (2007)
P.M. Kris-Etherton, K.D. Hecker, A. Bonanome, S.M. Coval, A.E. Binkoski, K.F. Hilpert, T.D. Etherton, Am. J. Med. Sci. 113, 71–88 (2002)
K.B. Pandey, S.I. Rizvi, Oxid. Med. Cell Longev. 2, 270–278 (2009)
J. Richter, I. Schellenberg, Anal. Bioanal. Chem. 387, 2207–2217 (2007)
T.L. Miron, M. Herrero, E. Ibáñez, J. Chromatogr. A 1288, 1–9 (2013)
G.E.P. Box, K.B. Wilson, J. R. Stat. Soc. 13, 1–45 (1951)
C. Liyana-Pathirana, F. Shahidi, Food Chem. 93, 47–56 (2005)
S.W. Chan, C.Y. Lee, C.F. Yap, W.W. Aida, C.W. Ho, Food Res. Int. 16, 203–213 (2009)
J. Wang, B. Sun, Y. Cao, Y. Tian, X. Li, Food Chem. 106, 804–810 (2008)
X. Wang, Y. Wu, G. Chen, W. Yue, Q. Liang, Q. Wu, Ultrason. Sonochem. 20, 846–854 (2013)
B. Yang, X. Liu, Y. Gao, Innov. Food Sci. Emerg. Technol. 10, 610–615 (2009)
T. Belwal, P. Dhyani, I.D. Bhatt, R.S. Rawal, V. Pande, Food Chem. 207, 115–124 (2016)
N. Ilaiyaraja, K.R. Likhith, G.S. Babu, F. Khanum, Food Chem. 173, 348–354 (2015)
K. Ghafoor, Y.H. Choi, J.Y. Jeon, I.H. Jo, J. Agric. Food Chem. 57, 4988–4994 (2009)
T. Suman, R. Elangomathavan, M. Kasipandi, K. Chakkaravarthi, D. Tamilvendan, T. Parimelazhagan, Egypt. J. Basic Appl. Sci. 5, 130–137 (2018)
R. Chandran, S. Sathyanarayanan, M. Rajan, M. Kasipandi, T. Parimelazhagan, Bangladesh J. Pharmacol. 10, 672–680 (2015)
P.S. Sreeja, K. Arunachalam, D.T. de Oliveira Martins, J.C. da Silva Lima, S.O. Balogun, E. Pavan, T. Parimelazhagan, J. Ethnopharmacol. 225, 71–80 (2018)
K. Muniyandi, E. George, P. Thangaraj, Medicinal Plants: Promising Future for Health and New Drugs, vol 15 (CRC Press, Boca Raton, 2018)
U.B. Parveen, S. Roy, A. Kumar, J. Ethnopharmacol. 113, 387–399 (2007)
K. Arunachalam, T. Parimelazhagan, J. Ethnopharmacol. 147, 302–310 (2013)
E.S.S. Abdel-Hameed, Food Chem. 114, 1271–1277 (2009)
B. Sultana, F. Anwar, Food Chem. 108, 879–884 (2008)
A. Taskeen, I. Naeem, H. Mubeen, T. Mehmood, N Y Sci. J. 2, 32–35 (2009)
R. Murugan, K. Arunachalam, T. Parimelazhagan, Food Sci. Biotechnol. 21, 59–67 (2012)
H.P.S. Makkar, Quantification of Tannins in Tree and Shrub Foliage: A Laboratory Manual (Kluwer Academic Publishers, Dordrecht, 2003)
J. Zhishen, T. Mengecheng, W. Jianming, Food Chem. 64, 555–559 (1999)
A. Braca, N. De Tommasi, L. Di Bari, C. Pizza, M. Politi, I. Morelli, J. Nat. Prod. 64, 892–895 (2001)
K. Muniyandi, E. George, V. Mudili, N.K. Kalagatur, A.J. Anthuvan, K. Krishna, P. Thangaraj, G. Natarajan, Agric. Nat. Resour. 51, 63–73 (2017)
A. Hubaux, G. Vos, Anal. Chem. 42, 849–885 (1970)
R.H. Myers, D.C. Montgomery, C.M. Anderson-Cook, Response Surface Methodology, Process and Product Optimization Using Designed Experiments, 4th edn. (Wiley, Hoboken, 2009)
L. Quanhong, F. Caili, Food Chem. 92, 701–706 (2005)
T. Gomes, T. Delgado, A. Ferreira, J.A. Pereira, P. Baptista, S. Casal, E. Ramalhosa, Ind. Crops Prod. 44, 622–629 (2013)
H.N. Rajha, N. El Darra, Z. Hobaika, N. Boussetta, E. Vorobiev, R.G. Maroun, N. Louka, Food Nutr. Sci. 5, 397 (2014)
U.J. Vajić, J. Grujić-Milanović, J. Živković, K. Šavikin, D. Gođevac, Z. Miloradović, N. Mihailović-Stanojević, Ind. Crops Prod. 74, 912–917 (2015)
T. Madhujith, F. Shahidi, Food Chem. 117, 615–620 (2009)
M.B. Hossain, N.P. Brunton, A. Patras, B. Tiwari, C.P. O’Donnell, A.B. Martin-Diana, C. Barry-Ryan, Ultrason. Sonochem. 19, 582–590 (2012)
A. Scalbert, C. Manach, C. Morand, C. Rémésy, L. Jiménez, Crit. Rev. Food Sci. Nutr. 45, 287–306 (2005)
P. Xu, J. Bao, J. Gao, T. Zhou, Y. Wang, BioResources 7, 2431–2443 (2012)
C.Y. Guo, J. Wang, Y. Hou, Y.M. Zhao, L.X. Shen, D.S. Zhang, Pharmacogn. Mag. 9, 192–195 (2013)
A.R. Proteggente, S. Wiseman, F.H.M.M. van de Put, C.A. Rice-Evans, in Flavonoids in Health and Disease, ed. by C.A. Rice-Evans, L. Packer (Marcell Dekker Inc, New York, 2003), pp. 71–96
N.I. Bazykina, A.N. Nikolaevskii, T.A. Filippenko, V.G. Kaloerova, Pharm. Chem. J. 36, 46–49 (2002)
R. Huang, E. Mendis, S.K. Kim, Int. J. Biol. Macromol. 36, 120–127 (2005)
M.J. Simirgiotis, Molecules 18, 2061–2080 (2013)
E. Karacabey, G. Mazza, Food Chem. 119, 343–348 (2010)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jagadeesan, G., Muniyandi, K., Manoharan, A.L. et al. Optimization of phenolic compounds extracting conditions from Ficus racemosa L. fruit using response surface method. Food Measure 13, 312–320 (2019). https://doi.org/10.1007/s11694-018-9946-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-018-9946-6