Abstract
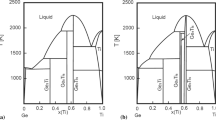
By using the CALPHAD technique, an assessment of the binary PrCl3-CaCl2 and NdCl3-CaCl2 systems have been carried out. From measured phase equilibrium data and experimental integral properties, the PrCl3-CaCl2 and NdCl3-CaCl2 phase diagrams were optimized and calculated. A set of thermodynamic functions has been optimized based on an interactive computer-assisted analysis. The calculated results by present method agree well with the experimental data.




Similar content being viewed by others
References
Y. Wang, G. Shao, S. Li, Y. Sun, and Z. Qiao, Phase Equilibria Calculation of the LaI3-MI (M = Na, K, Cs) Binary Systems, J. Rare Earths, 2009, 27(2), p 300-303
Y. Sun, Y. Wang, Z. Ma, X. Meng, X. Ye, and Z. Qiao, Thermodynamic Optimization of the Binary YbCl3-AECl2 (AE = Mg, Ca, Sr, Ba) Systems, J. Phase Equilib. Diffus., 2005, 26(6), p 616-621
Y. Wang, Y. Sun, Z. Qiao, X. Ye, Z. Ma, and X.N. Meng, Thermodynamic Optimization and Calculation of the SmCl3-MCl (M = Na, K, Rb, Cs) Phase Diagrams, CALPHAD, 2005, 29(4), p 317-322
T. Hattori, H. Ikezawa, R. Hirano, and J. Mochinaga, Phase diagram of Ternary Praseodymium Chloride-Calcium Chloride-Sodium Chloride System, Bull. Chem. Soc. Jpn., 1982, 6, p 952-955
I. Trifonov, G. Lebedev, T. Pobirchenko, V. Kuzmina, and B. Lyazgin, Sodium Chloride-Calcium Chloride-Praseodymium (III) Chloride System, Zh. Neorg. Khim., 1985, 30(6), p 1536-1537
C. Zheng, H. Liu, Z. Qiao, and Y. Ye, Phase Diagram of Ternary System PrCl3-CaCl2-MgCl2, Acta Metall. Sin., 1990, 26(1), p 68-70
R. Sharma and R. Rogers, Phase Equilibria and Structural Species in NdCl3-NaCl, NdCl3-CaCl2, PrCl3-NaCl, and PrCl3-CaCl2 Systems, J. Am. Ceram. Soc., 1992, 75(9), p 2484-2490
M. Gaune-Escard, L. Rycerz, W. Szezepaniak, and A. Bognacz, Enthalpies of Mixing in PrCl3-CaCl2 and NdCl3-CaCl2 Liquid Systems, Thermochim. Acta, 1994, 236, p 51-58
E. Guggenheim, The Statistical Mechanics of Regular Solutions, Proc. R. Soc., 1935, 148, p 304-312
R.H. Fowler and E.A. Guggenheim, Statistical Thermodynamics, Cambridge University Press, Cambridge, UK, 1939, p 350-366
A.D. Pelton and M. Blander, Analysis and Predictions of the Thermodynamic Properties of Multicomponent Silicates, Proc. Warrendale, AIME Symposium on Molten Salts and Slags, Lake Tahoe, Nevada, TMS-AIME, 1984, p 281-294
A.D. Pelton and M. Blander, Thermodynamic Analysis of Ordered Liquid Solutions by a Modified Quasichemical Approach Application to Silicate Slage, Metall. Trans., 1986, 17(4), p 805-815
M. Blander and A.D. Pelton, Thermodynamic Analysis of Binary Liquid Silicates and Prediction of Ternary Solution Properties by Modified Quasichemical Equations, Geochim. Cosmochim. Acta, 1987, 51(1), p 85-95
A.D. Pelton, S.A. Degterov, G. Eriksson, C. Robelin, and Y. Dessureault, The Modified Quasichemical Model I-Binary Solutions, Metall. Mater. Trans., 2000, 31, p 651-659
C.W. Bale, A.D. Pelton, and W.T. Thompson, F*A*C*T—Facility for the Analysis of Chemical Thermodynamics, User’s Guide and Supplement, McGill University and Ecole Polytechnique, Montreal, 2003
Y. Sun, “Measurement, Calculation and Evaluation by Pattern Recognition of the Rare Earth Halide Phase Diagrams,” Dissertation of Doctor’s Degree, Department of Physical Chemistry, University of Science and Technology Beijing, People’s Republic of China, 1999, in Chinese
Acknowledgments
The project is supported by Foundation of Nature Science of Hefei University (08KY019ZR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Y., Sun, Y. Thermodynamic Assessment of PrCl3-CaCl2 and NdCl3-CaCl2 Systems. J. Phase Equilib. Diffus. 31, 421–424 (2010). https://doi.org/10.1007/s11669-010-9780-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-010-9780-y