Abstract
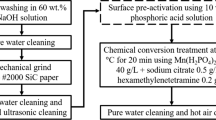
In this investigation, the effect of variations of phosphating bath (magnesium carbonate and phosphoric acid) concentration was studied on the properties of magnesium phosphate coating. The formation of the coating and morphological evolution were examined by XRD and SEM, respectively. The coating thickness was measured using a magnetic thickness gauge. Potentiodynamic polarization curves were used to investigate corrosion behavior. The findings revealed that optimizing both the phosphoric acid and magnesium carbonate concentration affects the nucleation and growth of the phosphate crystals and considerably affects the coating thickness and porosity. Therefore, optimizing these constituents is essential to decrease the corrosion rate. The heaviest coating thickness, lowest porosity and the lowest corrosion rate were observed at 23 mL/L of phosphoric acid and 9 g/L of magnesium carbonate concentrations. Variations of magnesium carbonate concentration was a more effective factor than the phosphoric acid.
Graphical Abstract


















Similar content being viewed by others
References
M. Manna, Characterisation of Phosphate Coatings Obtained Using Nitric Acid Free Phosphate Solution on Three Steel Substrates: An Option to Simulate TMT Rebars Surfaces, Surf. Coat. Technol., 2009, 203, p 1913. https://doi.org/10.1016/j.surfcoat.2009.01.024
T.K. Rout, H.K. Pradhan, and T. Venugopalan, Enhanced Forming Properties of Galvannealed Steel Sheet by Polymanganese Phosphate Coating, Surf. Coat. Technol., 2006, 201, p 3496. https://doi.org/10.1016/j.surfcoat.2006.07.260
M. Zubielewicz, E.K. Tarnawska, and A. Kozłowska, Protective Properties of Organic Phosphate-Pigmented Coatings on Phosphated Steel Substrates, Prog. Org. Coat., 2005, 53, p 276–285. https://doi.org/10.1016/j.porgcoat.2005.02.008
P.E. Tegehall and N.G. Vannerberg, Nucleation and Formation of Zinc Phosphate Conversion Coating on Cold-Rolled Steel, Corros. Sci., 1991, 32, p 635–652. https://doi.org/10.1016/0010-938X(91)90112-3
J. Flis, J. Mankowski, T. Zakroczymski, and T. Bell, The Formation of Phosphate Coatings on Nitrided Stainless Steel, Corros. Sci., 2001, 43, p 1711–1725. https://doi.org/10.1016/S0010-938X(00)00179-7
W. Zhou, D. Shan, E.H. Han, and W. Ke, Structure and Formation Mechanism of Phosphate Conversion Coating on Die-Cast AZ91D Magnesium Alloy, Corros. Sci., 2008, 50, p 329–337. https://doi.org/10.1016/j.corsci.2007.08.007
K. Ogle, A. Tomandl, N. Meddahi, and M. Wolpers, The Alkaline Stability of Phosphate Coatings I: ICP Atomic Emission Spectroelectrochemistry, Corros. Sci., 2004, 46, p 979–995. https://doi.org/10.1016/S0010-938X(03)00182-3
G.Y. Li, J.S. Lian, L.Y. Niu, Z.H. Jiang, and Q. Jiang, Growth of Zinc Phosphate Coatings on AZ91D Magnesium Alloy, Surf. Coat. Technol., 2006, 201, p 1814. https://doi.org/10.1016/j.surfcoat.2006.03.006
L.Y. Niu, Z.H. Jiang, G.Y. Li, C.D. Gu, and J.S. Lian, A Study and Application of Zinc Phosphate Coating on AZ91D Magnesium Alloy, Surf. Coat. Technol., 2006, 200, p 3021–3026. https://doi.org/10.1016/j.surfcoat.2004.10.119
D. Weng, P. Jokiel, A. Uebleis, and H. Boehni, Corrosion and Protection Characteristics of Zinc and Manganese Phosphate Coatings, Surf. Coat. Technol., 1996, 88, p 147. https://doi.org/10.1016/S0257-8972(96)02860-5
S.H.L. Zhang, H.H. Chen, X.L. Zhang, and M.M. Zhang, The Growth of Zinc Phosphate Coatings on 6061-Al Alloy, Surf. Coat. Technol., 2008, 202, p 1674. https://doi.org/10.1016/j.surfcoat.2007.07.037
M.C.M. Farias, C.A.L. Santos, Z. Panossian, and A. Sinatora, Friction Behavior of Lubricated Zinc Phosphate Coatings, Wear, 2009, 266, p 873–877. https://doi.org/10.1016/j.wear.2008.10.002
S. Jegannathan, T.S.N.S. Narayanan, K. Ravichandran, and S. Rajeswari, Performance of Zinc Phosphate Coatings Obtained by Cathodic Electrochemical Treatment in Accelerated Corrosion Tests, Electrochim. Acta, 2005, 51, p 247–256. https://doi.org/10.1016/j.electacta.2005.04.020
G. Li, L. Niu, J. Lian, and Z. Jiang, A Black Phosphate Coating for C1008 Steel, Surf. Coat. Technol., 2004, 176, p 215–221. https://doi.org/10.1016/S0257-8972(03)00736-9
B.L. Lin, J.T. Lu, and G. Kong, Effect of Molybdate Post-sealing on the Corrosion Resistance of Zinc Phosphate Coatings on Hot-Dip Galvanized Steel, Corros. Sci., 2008, 50, p 962–967. https://doi.org/10.1016/j.corsci.2007.12.002
X. Sun, D. Susac, R. Li, K.C. Wong, T. Foster, and K.A.R. Mitchell, Some Observations for Effects of Copper on Zinc Phosphate Conversion Coatings on Aluminum Surfaces, Surf. Coat. Technol., 2002, 155, p 46–50. https://doi.org/10.1016/S0257-8972(02)00027-0
Q. Li, Sh. Xu, J. Hu, Sh. Zhang, X. Zhong, and X. Yang, The Effects to the Structure and Electrochemical Behavior of Zinc Phosphate Conversion Coatings with Ethanolamine on Magnesium Alloy AZ91D, Electrochim. Acta, 2010, 55, p 887–894. https://doi.org/10.1016/j.electacta.2009.06.048
C.Y. Tsai, J.S. Liu, P.L. Chen, and C.S. Lin, A Two-Step Roll Coating Phosphate/Molybdate Passivation Treatment for Hot-Dip Galvanized Steel Sheet, Corros. Sci., 2010, 52, p 3385–3393. https://doi.org/10.1016/j.corsci.2010.06.020
V.S. Kathavate and P.P. Deshpande, Role of Nano TiO2 and Nano ZnO Particles on Enhancing the Electrochemical and Mechanical Properties of Electrochemically Deposited Phosphate Coatings, Surf. Coat. Technol., 2020, 394, p 12590. https://doi.org/10.1016/j.surfcoat.2020.125902
M. Tamilselvi, P. Kamaraj, M. Arthanareeswari, and S. Devikala, Nano Zinc Phosphate Coatings for Enhanced Corrosion Resistance of Mild Steel, Appl. Surf. Sci., 2015, 327, p 218–225. https://doi.org/10.1016/j.apsusc.2014.11.081
M. Arthanareeswaria, P. Kamarajb, M. Tamilselvic, and S. Devikala, A Low Temperature Nano TiO2 Incorporated Nano Zinc Phosphate Coating on Mild Steel with Enhanced Corrosion Resistance, Mater. Today: Proc., 2018, 5, p 9012–9025. https://doi.org/10.1016/j.matpr.2017.12.349
B.I. Lin, J.T. Lu, K.O. Gang, and L. Jun, Growth and Corrosion Resistance of Molybdate Modified Zinc Phosphate Conversion Coatings on Hot-Dip Galvanized Steel, Trans. Nonferrous Met. Soc. China, 2007, 17, p 755–761. https://doi.org/10.1016/S1003-6326(07)60169-1
Y. Tian, H. Huang, H. Wang, Y. Xie, X. Sheng, L. Zhong, and X. Zhang, Accelerated Formation of Zinc Phosphate Coatings with Enhanced Corrosion Resistance on Carbon Steel by Introducing α-Zirconium Phosphate, J. Alloys Compd., 2020, 831, p 15490. https://doi.org/10.1016/j.jallcom.2020.154906
T. Li, Z.J. Leng, S. Wang, X. Wang, R. Ghomashchi, Y. Yang, and J. Zhou, Comparison of the Effects of Pre-activators on Morphology and Corrosion Resistance of Phosphate Conversion Coating on Magnesium Alloy, J. Magnes. Alloy, 2021 https://doi.org/10.1016/j.jma.2021.03.012
S. Hu, M. Muhammad, M. Wang, R. Ma, A. Du, Y. Fan, X. Cao, and X. Zhao, Corrosion Resistance Performance of Nano-MoS2-Containing Zinc Phosphate Coating on Q235 Steel, Mater. Lett., 2020, 265, p 127256. https://doi.org/10.1016/j.matlet.2019.127256
W. Wang, Y. Tian, A. Ke, H. Huang, H. Shen, and X. Zhang, Enalapril Maleate as a Green Accelerator for Zinc Phosphating Coating on Low-Carbon Steel, J. Ind. Eng. Chem., 2023, 120, p 477–486. https://doi.org/10.1016/j.jiec.2022.12.056
Y. Tian, H. Huang, H. Wang, Y. Xie, X. Sheng, L. Zhong, and X. Zhang, Accelerated Formation of Zinc Phosphate Coatings with Enhanced Corrosion Resistance on Carbon Steel by Introducing A-Zirconium Phosphate, J. Alloys Compd., 2020, 831, p 15490. https://doi.org/10.1016/j.jallcom.2020.154906
M. Wang, R. Ma, A. Du, S. Hu, M. Muhammad, X. Cao, Y. Fan, X. Zhao, and J. Wu, Corrosion Resistance of Black Phosphorus Nanosheets Composite Phosphate Coatings on Q235 Steel, Mater. Chem. Phys., 2020, 250, p 123056. https://doi.org/10.1016/j.matchemphys.2020.123056
X. He, J. Wu, Y. Chen, L. Zhang, and X. Sheng, A Trace Amount of MXene@PDA Nanosheets for Low-Temperature Zinc Phosphating Coatings with Superb Corrosion Resistance, Appl. Surf. Sci., 2022, 603, p 154455. https://doi.org/10.1016/j.apsusc.2022.154455
A. Anawati, E. Hidayati, and H. Labibah, Characteristics of Magnesium Phosphate Coatings Formed on AZ31 Mg Alloy by Plasma Electrolytic Oxidation with Improved Current Efficiency, Mater. Sci. Eng. B, 2021, 272, p 115354. https://doi.org/10.1016/j.mseb.2021.115354
Q. Wu, B. Yu, P. Zhou, T. Zhang, and F. Wang, Fabrication of Phosphate Conversion Coatings on Rolled AZ31 Magnesium Alloy: Variation of Corrosion Resistance on Different Planes Induced by the Crystallographic Texture, Mater. Chem. Phys., 2021, 273, p 125121. https://doi.org/10.1016/j.matchemphys.2021.125121
E.P. Banczeka, P.R.P. Rodriguesb, and I. Costa, Evaluation of Porosity and Discontinuities in Zinc Phosphate Coating by Means of Voltametric Anodic Dissolution (VAD), Surf. Coat. Technol., 2009, 203, p 1213. https://doi.org/10.1016/j.surfcoat.2008.10.026
M. Fouladi and A. Amadeh, Comparative Study Between Novel Magnesium Phosphate and Traditional Zinc Phosphate Coatings, Mater. Lett., 2004, 58, p 3316. https://doi.org/10.1016/j.matlet.2013.01.061
M. Fouladi and A. Amadeh, Effect of Phosphating Time and Temperature on Microstructure and Corrosion Behavior of Magnesium Phosphate Coating, Electrochim. Acta, 2013, 106, p 1–12. https://doi.org/10.1016/j.electacta.2013.05.041
M.R. Dayyari, A. Amadeh, and S. Sadreddini, The Influence of Direct Current Density on Microstructure and Corrosion Resistance of Magnesium Phosphate Coating via Cathodic Electrochemical Treatment, Mater. Chem. Phys., 2017, 199, p 537–542. https://doi.org/10.1016/j.matchemphys.2017.07.040
M.R. Dayyari, A. Amadeh, and S. Sadreddini, Application of Magnesium Phosphate Coating on Low Carbon Steel via Electrochemical Cathodic Method and Investigation of Its Corrosion Resistance, J. Alloys Compd., 2015, 647, p 956–958. https://doi.org/10.1016/j.jallcom.2015.06.063
H. Liu, H. Yang, Z. Tong, D. Yang, Y. Ge, and X. Ren, Effects of Laser Shock Peening Post-treatment on Microstructure and Corrosion Resistance of Phosphate Conversion Coatings, Opt. Laser Technol., 2023, 162, p 10928. https://doi.org/10.1016/j.optlastec.2023.109289
A.A. Ayoola, B.M. Durodola, R. Babalola, O.D. Adeniyi, and C.E. Ilobinso, Corrosion Inhibitive Effects of Calcium-Modified Zinc Phosphate Coating on A36 Mild Steel, Results Eng., 2023, 17, p 10088. https://doi.org/10.1016/j.rineng.2023.100880
D. Zhang, Y. Ding, and X. Song, Preparation of Zn-Ca Phosphate Coating in Presence of PTFE on 45 Steel and Its Corrosion Properties in Simulated Seawater, Int. J. Electrochem. Sci., 2022, 17, p 220974. https://doi.org/10.20964/2022.09.42
M.F. Morks, Magnesium Phosphate Treatment for Steel, Mater. Lett., 2004, 58, p 3316. https://doi.org/10.1016/j.matlet.2004.06.027
E. E. Stansbury, Fundamentals of Electrochemical Corrosion, ASM International, Ohio, 2000, pp. 233–363
ASTM standard G 102-89, ASTM International, 2015, 03.02
D. Halliday, R. Resnick, and J. Walker, "Fundamental of Physics", 9th Ed., John Wiley and Sons, 2010, pp. 645
V.F.C. Lins and G.F.A. Reis, Electrochemical Impedance Spectroscopy and Linear Polarization Applied to Evaluation of Porosity of Phosphate Conversion Coatings on Electrogalvanized Steels, Surf. Coat. Technol., 2006, 253, p 2875–2884.
J. Creus, H. Mazille, and H. Idrissi, Porosity Evaluation of Protective Coatings Onto Steel, through Electrochemical Techniques, Surf. Coat. Technol., 2000, 130, p 224–232. https://doi.org/10.1016/j.apsusc.2006.06.030
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fouladi, M., Amadeh, A.A. Effect of Magnesium Carbonate and Phosphoric Acid Concentration on Microstructure and Corrosion Behavior of Magnesium Phosphate Coating. J. of Materi Eng and Perform (2023). https://doi.org/10.1007/s11665-023-08604-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11665-023-08604-y