Abstract
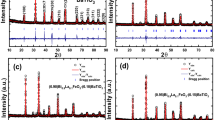
We prepared \({\text {SiO}}_{2}\), \({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\), and \({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) composites by an enhanced method and reported the result of their structural, optical and magnetic properties. In the x-ray diffraction results of the \({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\), \({\text {Fe}}_{2}{\text {O}}_{3}\) and the \({\text {SiO}}_{2}\) it is evident that these composites are crystallized in rhombohedral and trigonal structures, respectively. In the\({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) composite, \({\text {SiO}}_{2}\) transforms into a trigonal structure with the addition of Dy. The absorption bands belong to \({\text {Fe}}_{2}{\text {O}}_{3}\), and \({\text {Dy}}_{2}{\text {O}}_{3}\) were obtained using the Fourier transform infrared spectra. In ultraviolet–visible spectra, the photocatalytic properties of \({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) and \({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) were determined as a function of time at room temperature. Maximum transmittance change at 800 nm was 75% and 40% for composites \({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\), and \({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\), respectively. The photocatalytic property of \({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) composite increases gradually from short to long in the wavelength region where it exhibits a maximum value in the visible region. In magnetic measurements, a weak ferromagnetic behavior was observed in the \({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\), while \({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) exhibited paramagnetic behavior as expected. The saturation and coercivity values for \({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) were found to be 0.15 \(\hbox {Am}^2\hbox {kg}^{-1}\) and 40 mT, respectively.
Similar content being viewed by others
References
S. Soltan, H. Jafari, S. Afshar, and O. Zabihi, Water Sci. Technol. 77, 1689 (2016). https://doi.org/10.2166/wst.2016.343.
S.C. Feifel and F. Lisdat, J. Nanobiotechnol. 9, 59 (2011). https://doi.org/10.1186/1477-3155-9-59.
S.H. Huang and R.S. Juang, J. Nanopart. Res. 13, 4411 (2011). https://doi.org/10.1007/s11051-011-0551-4.
C. Pacurariu, E.A. Taculescu, R. Ianos, O. Marinica, C.V. Mihali, and V. Socoliuc, Ceram. Int. 41, 1079 (2015). https://doi.org/10.1016/j.ceramint.2014.09.031.
E. Esmaeili, M. Salavati-Niasari, F. Mohandes, F. Davar, and H. Seyghalkar, Chem. Eng. J. 170, 278 (2011). https://doi.org/10.1016/j.cej.2011.03.010.
U. Schwertmann and R.M. Cornel, The Iron Oxides in the Laboratory, Preparation and Characterization (Weinheim: VCH Publishers, 1996).
M. Mishra and D.M. Chun, Appl. Catal. A: Gen. 498, 126 (2015). https://doi.org/10.1016/j.apcata.2015.03.023.
M. Salavati-Niasari, F. Davar, and M. Mazaheri, J. Alloys Compd. 470, 502 (2009). https://doi.org/10.1016/j.jallcom.2008.03.048.
M. Salavati-Niasari, P. Salemi, and F. Davar, J. Mol. Catal. A: Chem. 238, 215 (2005). https://doi.org/10.1016/j.molcata.2005.05.026.
F. Beshkar and M. Salavati-Niasari, J. Nanostruct. 5, 17 (2015). https://doi.org/10.7508/jns.2015.01.003.
F. Motahari, M.R. Mozdianfard, F. Soofivand, and M. Salavati-Niasari, RSC Adv. 4, 27654 (2014). https://doi.org/10.1039/C4RA02697G.
F. Soofivand, F. Mohandes, and M. Salavati-Niasari, Mater. Res. Bull. 48, 2084 (2013). https://doi.org/10.1016/j.materresbull.2013.02.025.
M. Ranjbar, M. Salavati-Niasari, S.M. Hosseinpour-Mashkani, and K. Venkateswara-Rao, J. Inorg. Organomet. Polym Mater. 22, 1122 (2012). https://doi.org/10.1007/s10904-012-9704-x.
O. Amiri, M. Salavati-Niasari, S.M. Hosseinpour-Mashkani, A. Rafiei, and S. Bagheri, Mater. Sci. Semicond. Process. 27, 261 (2014). https://doi.org/10.1016/j.mssp.2014.06.020.
H. Zeynali, S. Behnam Mousavi, and S.M. Hosseinpour-Mashkani, Mater. Lett. 144, 65 (2015). https://doi.org/10.1016/j.matlet.2015.01.023.
N. Mir, M. Bahrami, E. Safari, and S.M. Hosseinpour-Mashkani, J. Cluster Sci. 26, 1103 (2015). https://doi.org/10.1007/s10876-014-0800-7.
A. Sobhani-Nasab, Z. Zahraei, M. Akbari, M. Maddahfar, and S.M. Hosseinpour-Mashkani, J. Mol. Struct. 1139, 430 (2017). https://doi.org/10.1016/j.molstruc.2017.03.069.
S. Zinatloo-Ajabshir, M.S. Morassaei, and M. Salavati-Niasari, J. Colloid Interface Sci. 497, 298 (2017). https://doi.org/10.1016/j.jcis.2017.03.031.
M. Mahdiani, F. Soofivand, F. Ansari, and M. Salavati-Niasari, J. Clean. Prod. 176, 1185 (2018). https://doi.org/10.1016/j.jclepro.2017.11.177.
J. Balbuena, M. Cruz-Yusta, A. Pastor, and L. Snchez, J. Alloys Compd. 735, 1553 (2018). https://doi.org/10.1016/j.jallcom.2017.11.259.
V.M. Bogatyrev, V.M. Gunko, M.V. Galaburda, M.V. Borysenko, V.A. Pokrovskiy, O.I. Oranska, E.V. Polshin, O.M. Korduban, R. Leboda, and J. Skubiszewska-Ziba, J. Colloid Interface Sci. 338, 376 (2009). https://doi.org/10.1016/j.jcis.2009.06.044.
Q. Sun, W. Leng, Z. Li, and Y. Xu, J. Hazard. Mater. 229230, 224232 (2012). https://doi.org/10.1016/j.jhazmat.2012.05.098.
X. Wang, C. Liu, X. Li, F. Li, and S. Zhou, J. Hazard. Mater. 153, 426433 (2008). https://doi.org/10.1016/j.jhazmat.2007.08.072.
Y.B. Saddeek, K.H.S. Shaaban, R. Elsaman, A. El-Taher, and T.Z. Amer, Radiat. Phys. Chem. 150, 182 (2018). https://doi.org/10.1016/j.radphyschem.2018.04.028.
M.S. Rao, B. Sanyal, K. Bhargavi, R. Vijay, I.V. Kityk, and N. Veeraiah, J. Mol. Struct. 1073, 174 (2014). https://doi.org/10.1016/j.molstruc.2014.04.075.
H. Huang, Y. Ou, S. Xu, G. Fang, M. Li, and X.Z. Zhao, Appl. Surf. Sci. 254, 2013 (2008). https://doi.org/10.1016/j.apsusc.2007.08.041.
G.A.S. Josephine and A. Sivasamy, Environ. Sci. Technol. Lett. 1, 172 (2014). https://doi.org/10.1021/ez4002032.
G.A.S. Josephine and A. Sivasamy, Appl. Catal. B: Environ. 150–151, 288 (2014). https://doi.org/10.1016/j.apcatb.2013.11.004.
A. Kumar, N. Yadav, M. Bhatt, N.K. Mishra, P. Chaudhary, and R. Singh, Res. J. Chem. Sci. 5, 98 (2015).
J.D. Mackenzie, J. Non-Cryst. Solids 100, 162 (1988).
A. Altomare, N. Corriero, C. Cuocci, A. Falcicchio, A. Moliterni, and R. Rizzi, J. Appl. Crystallogr. 48, 598 (2015). https://doi.org/10.1107/S1600576715002319.
S. Vasanthavel, B. Derby, and S. Kannan, Inorg. Chem. 56, 1273 (2017). https://doi.org/10.1021/acs.inorgchem.6b02361.
S. Moshtaghi, S. Zinatloo-Ajabshir, and M. Salavati-Niasari, J. Mater. Sci.: Mater. Electron. 27, 425 (2016). https://doi.org/10.1007/s10854-015-3770-0.
A. Vazquez, T. Lopez, R. Gomez, Bokhimi, A. Morales, and O. Novaro, J. Solid State Chem. 128, 161 (1997). https://doi.org/10.1006/jssc.1996.7135.
A. Aronne, S. Esposito, and P. Pernice, Mater. Chem. Phys. 51, 163 (1997). https://doi.org/10.1016/S0254-0584(97)80287-8.
W.L. Konijnendijk, Glastechn. Ber. 48, 216 (1975).
W.L. Konijnendijk and J.M. Stevels, J. Non-Cryst. Solids 21, 447 (1976). https://doi.org/10.1016/0022-3093(76)90033-8.
Y. Tsunawaki, N. Iwamoto, T. Hattori, and A. Mitsuishi, J. Non-Cryst. Solids 44, 369 (1981). https://doi.org/10.1016/0022-3093(81)90039-9.
S. Maensiri, C. Masingboon, B. Boonchom, and S. Seraphin, Scripta Mater. 56, 797 (2007). https://doi.org/10.1016/j.scriptamat.2006.09.033.
S. Zhang, Nanostructured Thin Films and Coatings: Functional Properties (Routledge: Taylor and Francis, 2010).
Y.B. Saddeek, K.H.S. Shaaban, R. Elsaman, A. El-Taher, and T.Z. Am, Proc. Indian Acad. Sci. 12A, 93 (1940).
V.C. Pierre and M.J. Allen, Contrast Agents for MRI. The Royal Society of Chemistry, London. (2018). https://doi.org/10.1039/9781788010146.
M. Arshad, A. Azam, A.S. Ahmed, S. Mollah, and A.H. Naqvi, J. Alloys Compd 509, 8378 (2011). https://doi.org/10.1016/j.jallcom.2011.05.047.
S. Muthu Kumaran and R. Gopalakrishnan, J. Sol-Gel Sci. Technol. 62, 193 (2012). https://doi.org/10.1007/s10971-012-2708-8.
J. Pisarska, J. Phys.: Condens. Matter 21, 285101 (2009). https://doi.org/10.1088/0953-8984/21/28/285101.
K. Kadono, N. Itakura, T. Akai, M. Yamashita, and T. Yazawa, J. Phys.: Condens. Matter 22, 045901 (2010). https://doi.org/10.1088/0953-8984/22/4/045901.
R. Kant, D. Kumar, and V. Dutta, RSC Adv. 5, 52945 (2015). https://doi.org/10.1039/C5RA06261F.
T.P. Raming, A.J.A. Winnubst, C.M. van Kats, and A.P. Philipse, J. Colloid Interface Sci. 249, 346 (2002). https://doi.org/10.1006/jcis.2001.8194.
D.X. Chen, V. Skumryev, and B. Bozzo, Rev. Sci. Instrum. 82, 045112 (2011). https://doi.org/10.1063/1.3581224.
D.J. Flood, Phys. Lett. A 49, 59 (1974). https://doi.org/10.1016/0375-9601(74)90668-9.
Acknowledgments
We thank Asst. Prof. Dr. C. Gökhan Ünlü for assistances of scanning electron microscopy measurement. Also, we thank Mr. Kağan Şarlar for the magnetic measurements of the composites. This work was supported by the Commission of Scientific Research Projects of Uludag University [Project Number OUAP(F)-2018/4].
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kendir, E., Tekgül, A., Küçük, İ. et al. Structural, Optical and Magnetic Properties of \(\alpha \)-\({\text {Fe}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) and \({\text {Dy}}_{2}{\text {O}}_{3}\)-\({\text {SiO}}_{2}\) Composites Produced by a Facile Method. J. Electron. Mater. 49, 798–806 (2020). https://doi.org/10.1007/s11664-019-07718-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07718-1