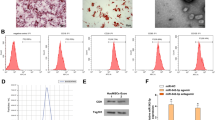
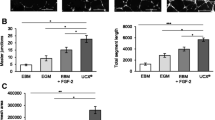
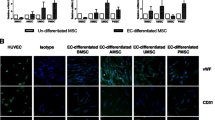
Abstract
Deep venous thrombosis (DVT) therapy during pregnancy warrants special consideration for the woman and the fetus. This study aimed to evaluate the impact of umbilical cord–derived mesenchymal stem cells (UC-MSCs) and bone marrow–derived mesenchymal stem cells (BM-MSCs) in terms of pro-angiogenic capacity and amelioration of pregnancy outcomes. The pregnant DVT rat model was successfully established by the “stenosis” method. Three consecutive injections of both UC-MSCs and BM-MSCs improved angiogenesis and ameliorated the embryo absorption rate in pregnant SD rats with DVT, in which UC-MSCs promoted angiogenesis more significantly. Furthermore, the levels of serum vascular endothelial growth factor-A (VEGF-A) and epidermal growth factor (EGF) were significantly higher in the UC-MSC group compared to those of the BM-MSC group. Thereafter, differentially expressed genes (DEGs) in thrombosed inferior vena cava tissues in the UC-MSC and BM-MSC groups were identified using transcriptome sequencing and further assessed by RT-qPCR and western blotting. The bioinformatics analysis indicated that the enriched DEG terms occurred in the cytokine activity, and the DEG pathways were significantly enriched in the cytokine-cytokine receptor interaction. In addition, both the mRNA and protein levels of angiogenic genes and their receptors, including VEGF-A, VEGF receptor-1, EGF, and EGF receptor, were significantly higher in the UC-MSC group. In conclusion, the BM-MSCs and UC-MSCs both significantly stimulate angiogenesis and ameliorate the embryo absorption rate in pregnant SD rats with DVT, but the difference in cytokine secretion causes UC-MSCs to have more potent angiogenic effects than BM-MSCs.







Similar content being viewed by others
References
Abbaspanah B, Reyhani S, Mousavi SH (2021) Applications of umbilical cord derived mesenchymal stem cells in autoimmune and immunological disorders: from literature to clinical practice. Curr Stem Cell Res Ther 16:454–464
Altmann J, Sharma S, Lang IM (2016) Advances in our understanding of mechanisms of venous thrombus resolution. Expert Rev Hematol 9:69–78
Bates SM, Middeldorp S, Rodger M, James AH, Greer I (2016) Guidance for the treatment and prevention of obstetric-associated venous thromboembolism. J Thromb Thrombolysis 41:92–128
Cheng X, Sun B, Liu S, Li D, Yang X, Zhang Y (2021) Identification of thrombomodulin as a dynamic monitoring biomarker for deep venous thrombosis evolution. Exp Ther Med 21:142
Dilli D, Kilic E, Yumusak N, Beken S, Uckan Cetinkaya D, Karabulut R, Zenciroglu AL (2017) Additive effect of mesenchymal stem cells and defibrotide in an arterial rat thrombosis model. Arch Argent Pediatr 115:249–256
Du WJ, Chi Y, Yang ZX, Li ZJ, Cui JJ, Song BQ, Li X, Yang SG, Han ZB, Han ZC (2016) Heterogeneity of proangiogenic features in mesenchymal stem cells derived from bone marrow, adipose tissue, umbilical cord, and placenta. Stem Cell Res Ther 7:163
Du X, Zhuang H, Hong L, Zhang Y, Li C, Qian A, Sang H, Li X (2019) Long-term outcome of catheter-directed thrombolysis in pregnancy-related venous thrombosis. Med Sci Monit 25:3771–3777
Ebrahim N, Mostafa O, El Dosoky RE, Ahmed IA, Saad AS, Mostafa A, Sabry D, Ibrahim KA, Farid AS (2018) Human mesenchymal stem cell-derived extracellular vesicles/estrogen combined therapy safely ameliorates experimentally induced intrauterine adhesions in a female rat model. Stem Cell Res Ther 9:175
Gee E, Roberts L, Arya R, Tsianakas V (2019) Patient experience of pregnancy-related venous thrombosis: a phenomenological study. Thromb Res 183:86–90
Han ZC, Du WJ, Han ZB, Liang L (2017) New insights into the heterogeneity and functional diversity of human mesenchymal stem cells. Biomed Mater Eng 28:S29–S45
James AH (2015) Thrombosis in pregnancy and maternal outcomes. Birth Defects Res C Embryo Today 105:159–166
Jo H, Brito S, Kwak BM, Park S, Lee MG, Bin BH (2021) Applications of mesenchymal stem cells in skin regeneration and rejuvenation. Int J Mol Sci 22(5):2410. https://doi.org/10.3390/ijms22052410
Mebarki M, Abadie C, Larghero J, Cras A (2021) Human umbilical cord-derived mesenchymal stem/stromal cells: a promising candidate for the development of advanced therapy medicinal products. Stem Cell Res Ther 12:152
Melief SM, Zwaginga JJ, Fibbe WE, Roelofs H (2013) Adipose tissue-derived multipotent stromal cells have a higher immunomodulatory capacity than their bone marrow-derived counterparts. Stem Cells Transl Med 2:455–463
Meng K, Hu X, Peng X, Zhang Z (2015) Incidence of venous thromboembolism during pregnancy and the puerperium: a systematic review and meta-analysis. J Matern Fetal Neonatal Med 28:245–253
Mukhopadhyay S, Johnson TA, Sarkar R, Antalis TM (2018) Serpins in venous thrombosis and venous thrombus resolution. Methods Mol Biol 1826:197–211
Nuzzo AM, Moretti L, Mele P, Todros T, Eva C, Rolfo A (2022) Effect of placenta-derived mesenchymal stromal cells conditioned media on an LPS-induced mouse model of preeclampsia. Int J Mol Sci 23(3):1674. https://doi.org/10.3390/ijms23031674
Paz T, Bloom A, Roth B, Kalish Y, Rottenstreich A, Elchalal U, Da’as N, Varon D, Spectre G (2021) Pharmacomechanical catheter thrombolysis for pregnancy-related proximal deep venous thrombosis: prevention of post-thrombotic syndrome. J Matern Fetal Neonatal Med 34:1441–1447
Reyhani S, Abbaspanah B, Mousavi SH (2020) Umbilical cord-derived mesenchymal stem cells in neurodegenerative disorders: from literature to clinical practice. Regen Med 15:1561–1578
Santos EDS, Nogueira KAB, Fernandes LCC, Martins JRP, Reis AVF, Neto JBV, Junior I, Pessoa C, Petrilli R, Eloy JO (2021) EGFR targeting for cancer therapy: pharmacology and immunoconjugates with drugs and nanoparticles. Int J Pharm 592:120082
Suvakov S, Richards C, Nikolic V, Simic T, McGrath K, Krasnodembskaya A, McClements L (2020) Emerging therapeutic potential of mesenchymal stem/stromal cells in preeclampsia. Curr Hypertens Rep 22:37
Urasoko Y, He XJ, Ebata T, Kinoshita Y, Kobayashi J, Mochizuki M, Ikeya M (2009) Changes in blood parameters and coagulation-related gene expression in pregnant rats. J Am Assoc Lab Anim Sci 48:272–278
Vedantham S, Goldhaber SZ, Julian JA, Kahn SR, Jaff MR, Cohen DJ, Magnuson E, Razavi MK, Comerota AJ, Gornik HL, Murphy TP, Lewis L, Duncan JR, Nieters P, Derfler MC, Filion M, Gu CS, Kee S, Schneider J, Saad N, Blinder M, Moll S, Sacks D, Lin J, Rundback J, Garcia M, Razdan R, VanderWoude E, Marques V, Kearon C, Investigators AT (2017) Pharmacomechanical catheter-directed thrombolysis for deep-vein thrombosis. N Engl J Med 377:2240–2252
Waltham M, Burnand KG, Collins M, McGuinness CL, Singh I, Smith A (2003) Vascular endothelial growth factor enhances venous thrombus recanalisation and organisation. Thromb Haemost 89:169–176
Waltham M, Burnand KG, Collins M, Smith A (2000) Vascular endothelial growth factor and basic fibroblast growth factor are found in resolving venous thrombi. J Vasc Surg 32:988–996
Wang Z, Zheng L, Lian C, Qi Y, Li W, Wang S (2019) Human umbilical cord-derived mesenchymal stem cells relieve hind limb ischemia by promoting angiogenesis in mice. Stem Cells Dev 28:1384–1397
Yumusak N, Yavuz U, Sarikaya B, Yucel G (2017) Effects of epidermal growth factor on reduction of the formation of thrombus and vessel wall healing in an experimental rat model. Bratisl Lek Listy 118:752–758
Zhang D, Fu L, Wang L, Lin L, Yu L, Zhang L, Shang T (2017) Therapeutic benefit of mesenchymal stem cells in pregnant rats with angiotensin receptor agonistic autoantibody-induced hypertension: implications for immunomodulation and cytoprotection. Hypertens Pregnancy 36:247–258
Zhang J, Kong X, Jin X, Gao P, Wang M, Yang L (2019) Bone marrow stromal cells transplantation promotes the resolution and recanalization of deep vein thrombosis in rabbits through regulating macrophage infiltration and angiogenesis. J Cell Biochem. https://doi.org/10.1002/jcb.28447
Funding
This work was supported by the Scientific Research Project of Jiangsu Provincial Health Committee (Grant No. Z2019003), the Nantong Science and Technology Plan Project (MS12021003), and the Postgraduate Research and Practice Innovation Program of Jiangsu Province (Grant Nos. KYCX20_2799, KYCX21_3118).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Supplementary Information
Below is the link to the electronic supplementary material.

11626_2022_733_Fig8_ESM.png
Figure S1. Rat DVT model diagram (stenosis). (A) A 2-cm incision in the middle of the abdomen and full exposure and separation of the inferior vena cava branches below the left renal vein to the level of iliac vein. (B) Ligation of the visible branches one by one with 5-0 silk suture. (C) Placement of 5-0 silk suture tightly around the IVC together with 4-0 silk suture. (D) Ligation of IVC and removal of 4-0 silk suture.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheng, X., Wang, W., Du, R. et al. Impact of repeated intravenous infusions of umbilical cord–derived versus bone marrow–derived mesenchymal stem cells on angiogenesis in a pregnant experimentally induced deep venous thrombosis rat model. In Vitro Cell.Dev.Biol.-Animal 58, 957–969 (2022). https://doi.org/10.1007/s11626-022-00733-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11626-022-00733-3