Abstract
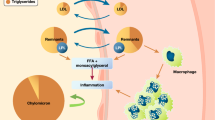
Chiglitazar sodium is a new peroxisome proliferator-activated receptor (PPAR) pan-agonist with independent intellectual property rights in China. It can treat type 2 diabetes mellitus and regulate metabolism by modestly activating PPARα, PPARγ, and PPARδ to improve insulin sensitivity, regulate blood glucose, and promote fatty acid oxidation and utilization. Chiglitazar sodium has a significant insulin-sensitizing effect and is advantageous in reducing fasting and postprandial blood glucose levels, particularly at the 48 mg dose in patients with concomitant high triglycerides in terms of blood glucose and triglyceride level control.
Similar content being viewed by others
References
Athyros VG, Doumas M, Imprialos KP, et al. Diabetes and lipid metabolism. Hormones (Athens), 2018,17(1):61–67
Javeed N, Matveyenko AV. Circadian Etiology of Type 2 Diabetes Mellitus. Physiology (Bethesda), 2018,33(2):138–150
Mayer-Davis EJ, Lawrence JM, Dabelea D, et al. Incidence Trends of Type 1 and Type 2 Diabetes among Youths, 2002–2012. N Engl J Med, 2017,376(15):1419–1429
Kahn SE, Haffner SM, Heise MA, et al. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N Engl J Med, 2006,355(23):2427–2443
Soccio RE, Chen ER, Lazar MA. Thiazolidinediones and the promise of insulin sensitization in type 2 diabetes. Cell Metab, 2014,20 (4):573–591
Li PP, Shan S, Chen YT, et al. The PPAR alpha/gamma dual agonist chiglitazar improves insulin resistance and dyslipidemia in MSG obese rats. Brit J Pharmacol, 2006,148(5):610–618
Pan DS, Wang W, Liu NS, et al. Chiglitazar Preferentially Regulates Gene Expression via Configuration-Restricted Binding and Phosphorylation Inhibition of PPARγ. PPAR Res, 2017,2017:4313561
He BK, Ning ZQ, Li ZB, et al. In Vitro and In Vivo Characterizations of Chiglitazar, a Newly Identified PPAR Pan-Agonist. PPAR Res, 2012,2012:546548
Gross B, Pawlak M, Lefebvre P, et al. PPARs in obesity-induced T2DM, dyslipidaemia and NAFLD. Nat Rev Endocrinol, 2016,13 (1):36–49
Deeks ED. Chiglitazar: First Approval. Drugs, 2022, 82(1):87–92
Haluzík MM, Haluzík M. PPAR-alpha and insulin sensitivity. Physiol Res, 2006,55(2):115–122
Cabrero A, Alegret M, Sánchez RM, et al. Bezafibrate reduces mRNA levels of adipocyte markers and increases fatty acid oxidation in primary culture of adipocytes. Diabetes, 2001,50(8):1883–1890
Chen T, Zhang Y, Liu Y, et al. MiR-27a promotes insulin resistance and mediates glucose metabolism by targeting PPAR-γ-mediated PI3K/AKT signaling. Aging (Albany NY). 2019,11(18):7510–7524
Kamenecka TM, Busby SA, Kumar N, et al. Potent Anti-Diabetic Actions of a Novel Non-Agonist PPARγ Ligand that Blocks Cdk5-Mediated Phosphorylation. In: Probe Reports from the NIH Molecular Libraries Program. Bethesda (MD): National Center for Biotechnology Information (US). 2011
Yessoufou A, Wahli W. Multifaceted roles of peroxisome proliferator-activated receptors (PPARs) at the cellular and whole organism levels. Swiss Med Wkly, 2010,140:w13071
Han L, Shen WJ, Bittner S, et al. PPARs: regulators of metabolism and as therapeutic targets in cardiovascular disease. Part III: PPAR-β/δ and PPAR-γ. Future Cardiol, 2017,13(3):279–296
Montaigne D, Butruille L, Staels B. PPAR control of metabolism and cardiovascular functions. Nat Rev Cardiol, 2021,18(12):809–823
Sullivan HJ, Wang X, Nogle S, et al. To Probe Full and Partial Activation of Human Peroxisome Proliferator-Activated Receptors by Pan-Agonist Chiglitazar Using Molecular Dynamics Simulations. PPAR Res, 2020,2020:5314187
Yan LP, Dou GF. In vitro metabolism of chiglitazar. J Chin J Pharmacol Toxicol (Chinese), 2007,21(2):124–130
Choi JH, Banks AS, Kamenecka TM, et al. Antidiabetic actions of a non-agonist PPARγ ligand blocking Cdk5-mediated phosphorylation. Nature, 2011,477(7365):477–481
Xu HR, Zhang JW, Chen WL, et al. Pharmacokinetics, Safety and Tolerability of Chiglitazar, A Novel Peroxisome Proliferator-Activated Receptor (PPAR) Pan-Agonist, in Healthy Chinese Volunteers: A Phase I Study. Clin Drug Invest, 2019,39(6):553–563
Li X, Yu J, Wu M, et al. Pharmacokinetics and Safety of Chiglitazar, a Peroxisome Proliferator-Activated Receptor Pan-Agonist, in Patients < 65 and ≥ 65 Years With Type 2 Diabetes. Clin Pharmacol Drug Dev, 2020,10(7):789–796
Yi L, Zhang H, Zhang JW, et al. Study on Drug-Drug Interactions Between Chiglitazar, a Novel PPAR Pan-Agonist and Metformin Hydrochloride in Healthy Subjects. Clin Pharmacol Drug Dev, 2019,8(7): 934–941
Lyu X, Gao Y, Zhu L, et al. Efcacy and safety of chiglitazar in type 2 diabetes: a multi-center, randomized, double-blind andparallel group clinical trial. J China Diabetes Mellitus (Chinese), 2019,11(5):334–340
Ji L, Song W, Fang H, et al. Efficacy and safety of chiglitazar, a novel peroxisome proliferator-activated receptor pan-agonist, in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled, phase 3 trial (CMAP). Sci Bull (Beijing), 2021,66(15):1571–1580
Jia W, Ma J, Miao H, et al. Chiglitazar monotherapy with sitagliptin as an active comparator in patients with type 2 diabetes: a randomized, double-blind, phase 3 trial (CMAS). Sci Bull (Beijing), 2021,66(15): 1581–1590
Zhou Y, Wang H, Wang Y, et al. Comparative Evaluation of Chiglitazar and Sitagliptin on the Levels of Retinol-Binding Protein 4 and Its Correlation With Insulin Resistance in Patients With Type 2 Diabetes. Front Endocrinol (Lausanne), 2022,13:801271
Wang Y, Li H, Gao H, et al. Effect of chiglitazar and sitagliptin on glucose variations, insulin resistance and inflammatory-related biomarkers in untreated patients with type 2 diabetes. Diabetes Res Clin Pract, 2021,183:109171
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author declare no conflicts of interest.
Rights and permissions
About this article
Cite this article
Zhang, Xh., Tian, Yf., Huang, Gl. et al. Advances in Studies of Chiglitazar Sodium, a Novel PPAR Pan-Agonist, for the Treatment of Type 2 Diabetes Mellitus. CURR MED SCI 43, 890–896 (2023). https://doi.org/10.1007/s11596-023-2760-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-023-2760-3