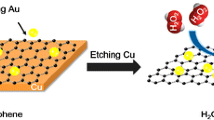
Abstract
Electrode surface modification is a common method for designing sensitive electrochemical sensors. However, a long time fabrication process is a key issue for their practical applications. In this work, we proposed a simple in situ electroless dipping deposition process for commercial electrode surface modification. Graphene and Ag NPs have been used as examples and stepwise fabricated on a glassy carbon electrode (GCE) surface. Each fabrication cycle only takes 1 min. Besides the fast fabrication process, the performance of the electrode can be easily adjusted by changing dipping cycle numbers. Based on the proposed procedure, a H2O2 electrochemical sensor was fabricated as an example with excellent performance. We believe the proposed approach could be extended for further nanomaterial-modified electrode design.






Similar content being viewed by others
References
Baghayeri M, Zare EN, Lakouraj MM (2015) Monitoring of hydrogen peroxide using a glassy carbon electrode modified with hemoglobin and a polypyrrole-based nanocomposite. Microchim Acta 182(3–4):771–779. https://doi.org/10.1007/s00604-014-1387-2
Lin H, Li M, Mihailovič D (2015) Simultaneous determination of copper, lead, and cadmium ions at a Mo 6 S 9-x I x nanowires modified glassy carbon electrode using differential pulse anodic stripping voltammetry. Electrochim Acta 154:184–189. https://doi.org/10.1016/j.electacta.2014.12.087
Pandikumar A, Yusoff N, Huang NM, Lim HN (2015) Electrochemical sensing of nitrite using a glassy carbon electrode modified with reduced functionalized graphene oxide decorated with flower-like zinc oxide. Microchim Acta 182(5–6):1113–1122
Prabakaran E, Pandian K (2015) Amperometric detection of Sudan I in red chili powder samples using Ag nanoparticles decorated graphene oxide modified glassy carbon electrode. Food Chem 166:198–205. https://doi.org/10.1016/j.foodchem.2014.05.143
Xing L, Ma Z (2016) A glassy carbon electrode modified with a nanocomposite consisting of MoS2 and reduced graphene oxide for electrochemical simultaneous determination of ascorbic acid, dopamine, and uric acid. Microchim Acta 183(1):257–263. https://doi.org/10.1007/s00604-015-1648-8
Tristão JC, de Mendonça FG, Lago RM, Ardisson JD (2015) Controlled formation of reactive Fe particles dispersed in a carbon matrix active for the oxidation of aqueous contaminants with H2O2. Environ Sci Pollut Res 22(2):856–863. https://doi.org/10.1007/s11356-014-2554-z
Goran JM, Phan EN, Favela CA, Stevenson KJ (2015) H2O2 detection at carbon nanotubes and nitrogen-doped carbon nanotubes: oxidation, reduction, or disproportionation? Anal Chem 87:5989–5996
Chang G, Luo Y, Lu W, Qin X, Sun X (2014) Carbon nanoparticles-induced formation of polyaniline nanofibers and their subsequent decoration with Ag nanoparticles for nonenzymatic H2O2 detection. Russ J Electrochem 50(1):95–99. https://doi.org/10.1134/S1023193513020043
Leonardi SG, Aloisio D, Donato N, Russo PA, Ferro MC, Pinna N, Neri G (2014) Amperometric sensing of H2O2 using Pt–TiO2/reduced graphene oxide nanocomposites. ChemElectroChem 1(3):617–624. https://doi.org/10.1002/celc.201300106
Wang Q, Li M, Szunerits S, Boukherroub R (2014) Environmentally friendly reduction of graphene oxide using tyrosine for nonenzymatic amperometric H2O2 detection. Electroanalysis 26(1):156–163. https://doi.org/10.1002/elan.201300356
Liu H, Chen X, Huang L, Wang J, Pan H (2014) Palladium nanoparticles embedded into graphene nanosheets: preparation, characterization, and nonenzymatic electrochemical detection of H2O2. Electroanalysis 26(3):556–564. https://doi.org/10.1002/elan.201300428
Shang L, Zeng B, Zhao F (2014) Fabrication of novel nitrogen-doped graphene–hollow AuPd nanoparticle hybrid films for the highly efficient electrocatalytic reduction of H2O2. ACS Appl Mater Interfaces 7(1):122–128. https://doi.org/10.1021/am507149y
Guo Q, Li M (2016) Electrodeposition of poly (sodium 4-Styrenesulfonate)-silver nanocomposites for electrochemical detection of H2O2. Int J Electrochem Sci 11(9):7705–7713
Li S-J, Zhang J-C, Li J, Wang W-T, Liu R-T (2017) One-step electrochemically modulated synthesis of reduced graphene oxide-silver nanocomposites as efficient nonenzymatic H2O2 sensor. Int J Electrochem Sci 12:5692–5703
Al Nafiey A, Subramanian P, Addad A, Sieber B, Szunerits S, Boukherroub R (2016) Green synthesis of reduced graphene oxide-silver nanoparticles using environmentally friendly L-arginine for H2O2 detection. ECS J Solid State Sci Technol 5(8):M3060–M3066
Zhao B, Liu Z, Fu W, Yang H (2013) Construction of 3D electrochemically reduced graphene oxide–silver nanocomposite film and application as nonenzymatic hydrogen peroxide sensor. Electrochem Commun 27(Supplement C):1–4. https://doi.org/10.1016/j.elecom.2012.10.040
Zheng Y, Wang A, Cai W, Wang Z, Peng F, Liu Z, Fu L (2016) Hydrothermal preparation of reduced graphene oxide–silver nanocomposite using Plectranthus amboinicus leaf extract and its electrochemical performance. Enzym Microb Technol 95:112–117. https://doi.org/10.1016/j.enzmictec.2016.05.010
Lou X, Zhu C, Pan H, Ma J, Zhu S, Zhang D, Jiang X (2016) Cost-effective three-dimensional graphene/ag aerogel composite for high-performance sensing. Electrochim Acta 205(Supplement C):70–76. https://doi.org/10.1016/j.electacta.2016.04.102
Zheng Y, Wang A, Cai W, Wang Z, Peng F, Liu Z, Fu L (2016) Hydrothermal preparation of reduced graphene oxide–silver nanocomposite using Plectranthus amboinicus leaf extract and its electrochemical performance. Enzym Microb Technol 95(Supplement C):112–117. https://doi.org/10.1016/j.enzmictec.2016.05.010
Si W, Lei W, Zhang Y, Xia M, Wang F, Hao Q (2012) Electrodeposition of graphene oxide doped poly(3,4-ethylenedioxythiophene) film and its electrochemical sensing of catechol and hydroquinone. Electrochim Acta 85:295–301. https://doi.org/10.1016/j.electacta.2012.08.099
Chen L, Yuan C, Dou H, Gao B, Chen S, Zhang X (2009) Synthesis and electrochemical capacitance of core-shell poly (3,4-ethylenedioxythiophene)/poly (sodium 4-styrenesulfonate)-modified multiwalled carbon nanotube nanocomposites. Electrochim Acta 54(8):2335–2341. https://doi.org/10.1016/j.electacta.2008.10.071
Ensafi AA, Abarghoui MM, Rezaei B (2014) Electrochemical determination of hydrogen peroxide using copper/porous silicon based non-enzymatic sensor. Sensors Actuators B Chem 196:398–405. https://doi.org/10.1016/j.snb.2014.02.028
Li X, Liu X, Wang W, Li L, Lu X (2014) High loading Pt nanoparticles on functionalization of carbon nanotubes for fabricating nonenzyme hydrogen peroxide sensor. Biosens Bioelectron 59:221–226. https://doi.org/10.1016/j.bios.2014.03.046
Lu X, Xiao X, Li Z, Xu F, Tan H, Sun L, Wang L (2014) A novel nonenzymatic hydrogen peroxide sensor based on three-dimensional porous Ni foam modified with a Pt electrocatalyst. Anal Methods 6(1):235–241. https://doi.org/10.1039/C3AY41566J
Han Y, Zheng J, Dong S (2013) A novel nonenzymatic hydrogen peroxide sensor based on Ag–MnO2–MWCNTs nanocomposites. Electrochim Acta 90:35–43. https://doi.org/10.1016/j.electacta.2012.11.117
Qi C, Zheng J (2016) Novel nonenzymatic hydrogen peroxide sensor based on Ag/Cu2O nanocomposites. Electroanalysis 28(3):477–483. https://doi.org/10.1002/elan.201500296
He S, Chen Z, Yu Y, Shi L (2014) A novel non-enzymatic hydrogen peroxide sensor based on poly-melamine film modified with platinum nanoparticles. RSC Adv 4(85):45185–45190. https://doi.org/10.1039/C4RA06925K
Kamyabi MA, Hajari N (2016) Preparation of mesoporous silica templated metal nanostructure on Ni foam substrate and its application for the determination of hydrogen peroxide. J Appl Electrochem 46(9):951–962. https://doi.org/10.1007/s10800-016-0986-5
Wu W, Li Y, Jin J, Wu H, Wang S, Xia Q (2016) A novel nonenzymatic electrochemical sensor based on 3D flower-like Ni7S6 for hydrogen peroxide and glucose. Sensors Actuators B Chem 232(Supplement C):633–641. https://doi.org/10.1016/j.snb.2016.04.006
Noorbakhsh A, Salimi A (2009) Amperometric detection of hydrogen peroxide at nano-nickel oxide/thionine and celestine blue nanocomposite-modified glassy carbon electrodes. Electrochim Acta 54(26):6312–6321. https://doi.org/10.1016/j.electacta.2009.05.078
Rani KK, Devasenathipathy R, Wang S-F, Yang C (2017) Simple preparation of birnessite-type MnO2 nanoflakes with multi-walled carbon nanotubes for the sensitive detection of hydrogen peroxide. Ionics 23(11):3219–3226. https://doi.org/10.1007/s11581-017-2117-7
Moozarm Nia P, Lorestani F, Meng WP, Alias Y (2015) A novel non-enzymatic H2O2 sensor based on polypyrrole nanofibers–silver nanoparticles decorated reduced graphene oxide nano composites. Appl Surf Sci 332(Supplement C):648–656. https://doi.org/10.1016/j.apsusc.2015.01.189
Golsheikh AM, Huang NM, Lim HN, Zakaria R (2014) One-pot sonochemical synthesis of reduced graphene oxide uniformly decorated with ultrafine silver nanoparticles for non-enzymatic detection of H2O2 and optical detection of mercury ions. RSC Adv 4(69):36401–36411. https://doi.org/10.1039/C4RA05998K
Noor AAM, Shahid MM, Rameshkumar P, Huang NM (2016) A glassy carbon electrode modified with graphene oxide and silver nanoparticles for amperometric determination of hydrogen peroxide. Microchim Acta 183(2):911–916. https://doi.org/10.1007/s00604-015-1679-1
Funding
This work has been financially supported by Research Foundation from Hangzhou Dianzi University (KYS205617071) and Zhejiang Province Natural Science Foundation of China (LQ18E010001).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
(DOCX 958 kb)
Rights and permissions
About this article
Cite this article
Fu, L., Wang, A., Su, W. et al. A rapid electrochemical sensor fabricated using silver ions and graphene oxide. Ionics 24, 2821–2827 (2018). https://doi.org/10.1007/s11581-017-2413-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-017-2413-2