Abstract
Background
Patellar femoral chondropathy (FPC) is a common problem in patients undergoing anterior cruciate ligament reconstruction (ACL-R) surgery, which, if left untreated, predisposes to arthrosis. Magnetic resonance imaging (MRI) is the non-invasive gold standard for morphological evaluation of cartilage, while in recent years advanced MRI techniques (such as T2 mapping) have been developed to detect early cartilage biochemical changes. This study evaluates the different onset of early PFC between B-TP-B and HT through T2 mapping. Secondly, it aims to assess the presence of any concordance between self-reported questionnaires and qualitative MRI.
Materials and methods
19 patients enrolled were divided into two groups based on the type of intervention: B-PT-B and HT. After a median time of 54 months from surgery, patients were subjected to conventional MRI, T2 mapping, and clinical-functional evaluation through three self-reported questionnaires: Knee Injury and Osteoarthritis index (KOOS); Tegner Lysholm Knee Scoring Scale; International Knee Documentation Committee (IKDC).
Results
There is not statistically significant difference in the comparison between the two MRI techniques and the two reconstructive techniques. KOOS and Tegner Lysholm scales showed significant agreement with MRI results on the grading of chondropathy.
Conclusions
There are no differences between B-TP-B and HT techniques in the early development of PFC detectable through non-invasive methods. Due to the large reduction in the frequency of physical activity following ACL-R and the finding of mild PFC (grade I and II) in a substantial proportion of patients, after a relatively short period from ACL-R, all patients should undergo conservative treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis is a well-renowned chronic pathology that involves joints and tissues. Beginning with progressive damaging of the articular cartilage and evolving to the subchondral bone and surrounding synovial structures, it is one of the most common causes of disability worldwide [1].
In knee surgery, Anterior Cruciate Ligament Reconstruction (ACL-R) is usually linked to long term onset of arthrosis. Giorgino et al., reported that after ACL rupture there is a fourfold increase in risk of developing osteoarthritis [2].
Reconstruction is done either by bone-patellar tendon-bone (B-PT-B) or with two hamstring tendons (HT): semitendinosus and gracilis.
B-TP-B has been popularized in the ‘60 s by Erikson [3] and offers a fast procedure that can lead to a reprise of the knee functionality as before of the trauma and a reduced fracture risk. On the contrary, B-TP-B reduces the anterior knee pain, at the cost of a reduction of the extensor apparatus power [4,5,6,7,8,9].
Studies demonstrated that patello-femoral arthrosis (PFA) in ACL-R patients is almost inevitable as it has been reported in up to 80% of the patients after 10 years and up to 90% after 15 years from the surgery [10, 11].
PFA is most common in elderly, but can be found also in athletes [12, 13]. This condition is often underdiagnosed as follow-ups are usually performed by radiography in which only advanced arthrosis is recognisable. Plain radiography do not show statistically significant differences between HT and B-PT-B.
recostruction [14]. As detecting early chondropathy in a reversible phase is crucial for personalized interventions, radiography is not ideal [15,16,17,18,19,20].
Magnetic Resonance (MR) is the non-invasive gold standard imaging technique for chondral anatomy and the evaluation of early bone insults (eg presence of oedema at the spongy bone). However, MR has less sensitivity in picturing chondral biochemical alterations in early chondropathy stage, such as reduction of proteoglycans and changes in water and collagen percentage [21,22,23,24]. Different studies on cartilage detriment following ACL reconstruction report how in 1–5 years follow ups patellofemoral cartilage (PFC) is compromised way more than tibiofemoral cartilage [25, 26].
Lately, different MR sequences such as T1r, T2 and dGEMRIC has been developed for evaluating changes in cartilage composition before morphologic changes can be appreciated by standard MR imaging (MRI) [27]. In particular, studies linking variations in T2 values and variations of water and collagen in cartilage have shown significant differences between symptomatic and asymptomatic patients with arthrosis [28, 29].
T2 mapping has been validated for evaluating early degenerative changes in various joints such as knee, wrist and ankle [28, 30, 31]. During T2 mapping acquisition, the T2 relaxation time of the cartilage is measured by using fixed repetition time and multiple spin-echo times. T2 mapping values tend to increase with the aging of the patient or with disruptive processes [29]. Changes in T2 mapping values do not correlate with specific pathologies but underlines biochemical alterations within the cartilage.
This study firstly aims to assess the possible presence of differences in the onset of chondropathy between the use of B-TP-B and HT for ACL-R comparing T2 mapping evaluation and standard MR morphologic evaluation. Secondarily, it aims to understand whether there are specific zones in the PFC more prone to detriment. We choose to divide the PFC in 9 compartments following the 3-dimensional axes: superior—middle—inferior, lateral—central—medial; superficial—intermediate—deep.
Materials and methods
This prospective study has been conducted in accordance with the principles of the Declaration of Helsinki. Informed consent has been signed by patients enrolled in the study. We enrolled 19 patients sorted in two groups: patients who underwent an ACL reconstruction with B-TP-B (group 1; 9 patients) and with HT (group 2, 10 patients) between June 2016 and January 2020. All patients underwent surgery at the same Orthopaedic Center (Ospedale Maggiore della Carità—Novara—Italy). We used the following inclusion criteria: patients between 18- and 40-year-old, ACL rupture without other ligament lesions, surgery done within 6 months from trauma, surgery done in the last 6 years, no other knee traumas since the surgery. The 6-year-from-surgery cut off represents an optimal timeframe for the interception of early chondropathy [7]. The exclusion criteria were: Body Mass Index (BMI) > 30 kg/m2, presence of any kind of lesions at the cartilage at the moment of surgery, disease at the PF joint, genu valgus, genu varus, instability of the operated knee and refuse to participate. The presence of damages at the cartilage (e.g. chondral fissuring) was visualised via qualitative assessment through pre-operation MRI. Patients > 40 years were excluded to reduce the influence of age-related osteoarthritis [32].
All tests were performed on a 1.5T (Philips Ingenia Ambition) with a 16-channel coil dedicated for the knee. The sequences performed were T1-weighted on the sagittal plane (FOV 140 × 140 mm, TR/TE/NEX 525/min full/2, matrix 320 × 224, slice thickness 4 mm, gap 0.4 mm), T2-weighted on the axial plane (FOV 140 × 140 mm, TR/TE/NEX 6800/102/2, matrix 320 × 224, slice thickness 4 mm, 0.4 mm), T2 FAT-SAT on the coronal plane (FOV 140 × 140 mm, TR/TE/NEX 6800/102/2, matrix 320 × 224, slice thickness 4 mm, gap 0.4 mm) and proton density (PD) FAT-SAT on the sagittal plane (FOV 140 × 140 mm, TR/TE/NEX 1640/35/2, matrix 320 × 224, slice thickness 4 mm, gap 0.4 mm). T2 mapping was performed via multi-echo spin-echo (SE) sequence called T2-Cal (FOV 140 × 140 mm, TR/TE 1000/7.6, 15.2, 22.8, 30.4, 37.6, 45.6, 52.8 e 60.6 ms, matrix 320 × 224, slice thickness 3 mm, gap 0 mm, bandwidth 228 kHz).
Every patient enrolled has been visited the day of the MR acquisition by our orthopaedic team who registered the most valuable anamnesis data (sex, age, operated knee, date of surgery, comorbidity), and administered three self-reported questionnaires: Knee Injury and Osteoarthritis index (KOOS) (score 0–100); Tegner Lysholm Knee Scoring Scale (score 0–100); International Knee Documentation Committee (IKDC) (score 0–100). Physical activity pre- and post-surgery was evaluated via Bhapkar test and non-parametric Wilcoxon test. Results from the group’s analysis via qualitative MRI and three functional scales (KOOS, Tegner Lysholm, IKDC) were compared via non-parametric Wilcoxon Rank Sum test.
Image analysis
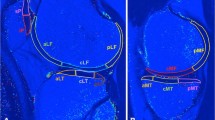
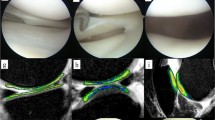
Images were anonymized, randomized and read by two musculoskeletal experts (with 5 and 30 years of experience respectively). Images were evaluated through a morphological analysis of the cartilage following the modified Outerbridge classification [33] and a quantitative analysis: 11 patients presented grade 0-I chondropathy and 8 patients presented grade II chondropathy. The quantitative analysis was performed through T2 mapping sequence evaluation via Philips IntelliSpace Portal v.93. The comparison between the groups was done on 9 segments obtained by dividing the cartilage cranio-caudally, latero-laterally and antero-posteriorly and by considering 27 segments of the PF joint. Manual segmentation was performed in the presence of both musculoskeletal experts with intra-procedural agreement. Cartilage was selected by tracing two lines: one at the bone-cartilage interface and one at the cartilage-joint interface. Cartilage was divided cranio-caudally (superior, middle, inferior), latero-laterally (lateral, central, medial) and antero-posteriorly (superficial, intermediate, deep) thus obtaining 27 volumes (Fig. 1). The software produces a colorimetric map based on T2 relaxation time ranging from 0 ms (in blue) to 80 ms (in dark red) with a relative T2 mapping mean value for every segment (Fig. 2).
Results
Population
Patients were divided into two grups: group 1 (9 patients who underwent ACL reconstruction with B-TP-B) and group 2 (10 patients who underwent ACL reconstruction with HT). Group 1 comprehended 8 males and 1 female while group 2 comprehended 8 males and 2 females. Due to the small population, we did not compare the two groups by age, BMI, and years from surgery, but instead we considered the single group as a whole. Table 1 reports general information about the two groups. Group 1 (B-TP-B) had a mean age of 26.67 years, with the youngest patient being 21 and the oldest one being 33. The mean BMI was 25.37 kg/m2. The lower BMI was 19.50 kg/m2 while the highest was 29.10 kg/m2. Mean time from surgery was 51.996 months. The shortest period from surgery was 36 months, while the longest period was 72 months.
Group 2 (HT) had a mean age of 29 years, ranging from 22 to 35 years. Mean BMI was 25.34 kg/m2, with the lower BMI being 20.50 kg/m2 and the higher 28.40 kg/m2. Mean time from surgery of the second group was 54.312 months. The shortest period from surgery was 36 months and the longest one was 72 months.
Functional assessment
The physical activity pre- and post-surgery of the 19 patients was evaluated via Bhapkar test and non-parametric Wilcoxon test (Table 2). Nearly every patient reduced physical activity. Only 7 patients who used to perform physical activity more than twice per week did not change weekly physical activity. On the contrary, 1 patient who lived a sedentary life before the surgery increased physical activity post-surgery (twice per week). The reduction of the physical activity of the patients was statistically significant (p-value < 0.05).
Qualitative and quantitative MRI assessment
The non-parametric Wilcoxon Rank Sum test regarding the results obtained by qualitative and quantitative MRI between group 1 and group 2 showed no significant differences. In Table 3 we report the comparison between qualitative MRI and quantitative T2 mapping MRI in describing two independent groups that underwent ACL-R with B-TP-B (Group 1) and HT (Group 2). Here reported is the comparison between the 9 segments.
Furthermore, we compared two independent groups: patients with grade 0/I chondropathy and patients with grade II chondropathy and their results from the three functional scales (KOOS, Tegner Lysholm, IKDC). KOOS and Tegner Lysholm scales were found concordant to qualitative MRI as reported in Table 4 with a p-value < 0.05.
Quantitative MRI evaluation of the cartilage’s segments and functional scales were compared through Spearman’s Index showing no correlation (Table 5). Table 5 shows all the data relative to the 27 segments in which the PF joint was divided. No significant concordance was found between T2 mapping MRI evaluation of the single segment and the functional scales.
Discussions
Following the rupture of the anterior cruciate ligament, its reconstruction represents the best therapeutic option especially in young patients. Unlikely, this intervention is burdened by a high rate of development of early osteoarthritis that leads to chronic pain, particularly in the patellofemoral compartment. Since its diagnosis is often delayed in routine follow-up instrumental examinations, our study aimed to use the advanced T2 mapping magnetic resonance technique to intercept early changes of the osteocartilaginous layer in this specific compartment. Zhao et al., demonstrated how MRI is capable of measuring early biochemical changes in the cartilage via T2 mapping [34]; his early detection can lead to anticipated and more personalized treatments leading to better outcomes for the patient by slowing chondropathy [2].
This study considered 19 patients (16 males and 3 females, with an average age of about 26.7 years), after an average time of about 54 months from surgery. After the intervention we found a statistically significant reduction in weekly physical activity. Patients with ACL reconstruction may not return to preinjury levels of sports participation for a variety of reasons. As Ardern et al. [35] reported, the most common cause of the reduction are: fear of reinjury (19%), reasons other than the knee (18%), knee impairments (13%), and changes in lifestyle (11%).
In our group of patients, we have found that the reduced functionality can be linked to the development of patello-femoral chondropathy. T2 mapping technique allows its detection in 57.9% of patients with grade 0/I (Outerbridge classification modified [33]) and in 42.1% with grade II. This data underlines the possibility of an early chondropathy in those patients who are not subjected to adequate follow-up and treatment.
The comparison between the B-TP-B and the HT group through qualitative and quantitative MR analysis found no significative differences between the two groups in terms of chondropathy showing that it is not the type of ACL reconstruction that influences the onset of arthrosis. Our observation results in accordance to the systematic review of Belk et al. [14] in which no significative differences were found between the use of B-TP-B or HT for ACL reconstruction at 11 years from surgery.
In this study we evaluated the concordance between qualitative and quantitative (T2 mapping) analyses for everyone of the 27 segments by which we divided the cartilage. As suggested by other studies [21, 23], T2 mapping is a useful tool for analyse the cartilage as a whole, but it loses its diagnostic value when it comes to small segments. In particular, as Surowiec et al. [21] pointed out, the subdivision of the knee cartilage into clinically relevant and reproducible subregions such as variations in the composition and structure of the healthy cartilage, proved to be incoherent and can lead to an uneven distribution of T2 cartilage values. Furthermore, as demonstrated by Slauterbeck et al. [36] cartilage degeneration can occur throughout the entire joint, highlighting the need for subregions they take this into consideration.
Comparison of the three functional scales (KOOS, Tegner Lysholm, and IKDC) and the standard MRI assessment of the PF joint showed KOOS and Tegner Lysholm scales had significant agreement between the self-report questionnaires and the MRI results on the grading of chondropathy. As reported in previous studies [37, 38], avoidance of sports activity for fear of a new injury could explain the low pain ratings in these functional scales compared to an underlying condition of early chondropathy without clinically relevant symptoms.
Furthermore, there was no significant agreement between functional scales and the quantitative assessment of the 27 subregions through T2 mapping. This may be explained by the limitations of the T2 mapping described above, whereby T2 mapping works best when considering the cartilage as a whole rather than high-number segmentation [21, 36].
This study has some limitations. The small population surely can affect the statistical power of the study and can explain the lack of significance of T2 mapping results. Another limitation is the use of a single-vendor MRI. About the non-significative difference between the two ACL reconstruction techniques, one limit is represented by the short period underwent from the surgery, but a longer period could enlight some differences. The group of patients enlisted for the study was homogeneous for indirect factors influencing the onset of chondropathy, such as BMI, smoking habits, diabetes and hypertension. Further studies should consider a more heterogenous population. The small number of patients enlisted compromises our study.
In the end, T2 mapping is a powerful tool for quantitative analyses, but it still suffers from inter-observer bias as there is no international standard for processing the data obtained from the sequences. As suggested by Surowiec et al., to improve this issue a full automatization of the segmentation is needed [21, 28].
Conclusions
After ACL reconstruction there is often a reduction in physical activity which could be determined by the onset of chondropathy. The advanced T2 mapping technique has proved to be a useful tool for detecting early cartilage alterations. However, the partial positivity of the functional scales suggests that psychological avoidance factors could play an important role in the reduction of sports activity.
The development of chondropathy would not appear to be linked to the type of intervention between the use of B-TP-B or HT. Furthermore, T2 mapping did not detect specific sectors within the cartilaginous layer that could be more exposed to the development of chondropathy, although a lack of a standardized protocol for the image processing.
This work represents a preliminary study, further investigation with larger sample size and different vendor machine are mandatory.
References
Hunter DJ, Bierma-Zeinstra S (2019) Osteoarthritis. Lancet Lond Engl 393:1745–1759. https://doi.org/10.1016/S0140-6736(19)30417-9
Giorgino R, Albano D, Fusco S, Peretti GM, Mangiavini L, Messina C (2023) Knee osteoarthritis: epidemiology, pathogenesis, and mesenchymal stem cells: what else is new? An update. Int J Mol Sci 24:6405. https://doi.org/10.3390/ijms24076405
Chambat P, Guier C, Sonnery-Cottet B, Fayard J-M, Thaunat M (2013) The evolution of ACL reconstruction over the last fifty years. Int Orthop 37:181–186. https://doi.org/10.1007/s00264-012-1759-3
Freedman KB, D’Amato MJ, Nedeff DD, Kaz A, Bach BR (2003) Arthroscopic anterior cruciate ligament reconstruction: a metaanalysis comparing patellar tendon and hamstring tendon autografts. Am J Sports Med 31:2–11. https://doi.org/10.1177/03635465030310011501
Gifstad T, Foss OA, Engebretsen L, Lind M, Forssblad M, Albrektsen G, Drogset JO (2014) Lower risk of revision with patellar tendon autografts compared with hamstring autografts: a registry study based on 45,998 primary ACL reconstructions in Scandinavia. Am J Sports Med 42:2319–2328. https://doi.org/10.1177/0363546514548164
Li S, Chen Y, Lin Z, Cui W, Zhao J, Su W (2012) A systematic review of randomized controlled clinical trials comparing hamstring autografts versus bone-patellar tendon-bone autografts for the reconstruction of the anterior cruciate ligament. Arch Orthop Trauma Surg 132:1287–1297. https://doi.org/10.1007/s00402-012-1532-5
Lien-Iversen T, Morgan DB, Jensen C, Risberg MA, Engebretsen L, Viberg B (2020) Does surgery reduce knee osteoarthritis, meniscal injury and subsequent complications compared with non-surgery after ACL rupture with at least 10 years follow-up? A systematic review and meta-analysis. Br J Sports Med 54:592–598. https://doi.org/10.1136/bjsports-2019-100765
Marques FS, Barbosa PHB, Alves PR, Zelada S, Nunes RPS, de Souza MR, Pedro MAC, Nunes JF, Alves WM, de Campos GC (2020) Anterior knee pain after anterior cruciate ligament reconstruction. Orthop J Sports Med. https://doi.org/10.1177/2325967120961082
Samuelsen BT, Webster KE, Johnson NR, Hewett TE, Krych AJ (2017) Hamstring autograft versus patellar tendon autograft for ACL reconstruction: is there a difference in graft failure rate? A meta-analysis of 47,613 patients. Clin Orthop 475:2459–2468. https://doi.org/10.1007/s11999-017-5278-9
Culvenor AG, Cook JL, Collins NJ, Crossley KM (2013) Is patellofemoral joint osteoarthritis an under-recognised outcome of anterior cruciate ligament reconstruction? A narrative literature review. Br J Sports Med 47:66–70. https://doi.org/10.1136/bjsports-2012-091490
Culvenor AG, Lai CCH, Gabbe BJ, Makdissi M, Collins NJ, Vicenzino B, Morris HG, Crossley KM (2014) Patellofemoral osteoarthritis is prevalent and associated with worse symptoms and function after hamstring tendon autograft ACL reconstruction. Br J Sports Med 48:435–439. https://doi.org/10.1136/bjsports-2013-092975
Øiestad BE, Holm I, Engebretsen L, Aune AK, Gunderson R, Risberg MA (2013) The prevalence of patellofemoral osteoarthritis 12 years after anterior cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc Off J ESSKA 21:942–949. https://doi.org/10.1007/s00167-012-2161-9
Ajuied A, Wong F, Smith C, Norris M, Earnshaw P, Back D, Davies A (2014) Anterior cruciate ligament injury and radiologic progression of knee osteoarthritis: a systematic review and meta-analysis. Am J Sports Med 42:2242–2252. https://doi.org/10.1177/0363546513508376
Belk JW, Kraeutler MJ, Carver TJ, McCarty EC (2018) Knee osteoarthritis after anterior cruciate ligament reconstruction with bone-patellar tendon-bone versus hamstring tendon autograft: a systematic review of randomized controlled trials. Arthrosc J Arthrosc Relat Surg Off Publ Arthrosc Assoc N Am Int Arthrosc Assoc 34:1358–1365. https://doi.org/10.1016/j.arthro.2017.11.032
Zheng W, Li H, Hu K, Li L, Bei M (2021) Chondromalacia patellae: current options and emerging cell therapies. Stem Cell Res Ther 12:412. https://doi.org/10.1186/s13287-021-02478-4
da Costa SR, da Mota RF, Albuquerque E, Helito CP, Camanho GL (2021) The role of viscosupplementation in patellar chondropathy. Ther Adv Musculoskelet Dis. https://doi.org/10.1177/1759720X211015005
Filardo G, Di Matteo B, Di Martino A, Merli ML, Cenacchi A, Fornasari P, Marcacci M, Kon E (2015) Platelet-rich plasma intra-articular knee injections show no superiority versus viscosupplementation: a randomized controlled trial. Am J Sports Med 43:1575–1582. https://doi.org/10.1177/0363546515582027
Bakhtiary AH, Fatemi E (2008) Open versus closed kinetic chain exercises for patellar chondromalacia. Br J Sports Med 42:99–102. https://doi.org/10.1136/bjsm.2007.038109. (discussion 102)
Fu Y, Karbaat L, Wu L, Leijten J, Both SK, Karperien M (2017) Trophic effects of mesenchymal stem cells in tissue regeneration. Tissue Eng Part B Rev 23:515–528. https://doi.org/10.1089/ten.TEB.2016.0365
Squillaro T, Peluso G, Galderisi U (2016) Clinical trials with mesenchymal stem cells: an update. Cell Transplant 25:829–848. https://doi.org/10.3727/096368915X689622
Surowiec RK, Lucas EP, Ho CP (2014) Quantitative MRI in the evaluation of articular cartilage health: reproducibility and variability with a focus on T2 mapping. Knee Surg Sports Traumatol Arthrosc Off J ESSKA 22:1385–1395. https://doi.org/10.1007/s00167-013-2714-6
Baum T, Joseph GB, Karampinos DC, Jungmann PM, Link TM, Bauer JS (2013) Cartilage and meniscal T2 relaxation time as non-invasive biomarker for knee osteoarthritis and cartilage repair procedures. Osteoarthritis Cartilage 21:1474–1484. https://doi.org/10.1016/j.joca.2013.07.012
Le J, Peng Q, Sperling K (2016) Biochemical magnetic resonance imaging of knee articular cartilage: T1rho and T2 mapping as cartilage degeneration biomarkers. Ann N Y Acad Sci 1383:34–42. https://doi.org/10.1111/nyas.13189
Patterson BE, Culvenor AG, Barton CJ, Guermazi A, Stefanik JJ, Morris HG, Whitehead TS, Crossley KM (2018) Worsening knee osteoarthritis features on magnetic resonance imaging 1–5 years after anterior cruciate ligament reconstruction. Am J Sports Med 46:2873–2883. https://doi.org/10.1177/0363546518789685
Culvenor AG, Collins NJ, Guermazi A, Cook JL, Vicenzino B, Khan KM, Beck N, van Leeuwen J, Crossley KM (2015) Early knee osteoarthritis is evident one year following anterior cruciate ligament reconstruction: a magnetic resonance imaging evaluation. Arthritis Rheumatol Hoboken NJ 67:946–955. https://doi.org/10.1002/art.39005
Theologis AA, Haughom B, Liang F, Zhang Y, Majumdar S, Link TM, Ma CB, Li X (2014) Comparison of T1rho relaxation times between ACL-reconstructed knees and contralateral uninjured knees. Knee Surg Sports Traumatol Arthrosc Off J ESSKA 22:298–307. https://doi.org/10.1007/s00167-013-2397-z
Vogrig C, Louis J-S, Avila F, Gillet R, Hossu G, Blum-Moyse A, GondimTeixeira PA (2021) Synthetic MRI is not yet ready for morphologic and functional assessment of patellar cartilage at 1.5Tesla. Diagn Interv Imaging 102:181–187. https://doi.org/10.1016/j.diii.2020.09.002
Yao W, Qu N, Lu Z, Yang S (2009) The application of T1 and T2 relaxation time and magnetization transfer ratios to the early diagnosis of patellar cartilage osteoarthritis. Skeletal Radiol 38:1055–1062. https://doi.org/10.1007/s00256-009-0769-8
Apprich S, Welsch GH, Mamisch TC, Szomolanyi P, Mayerhoefer M, Pinker K, Trattnig S (2010) Detection of degenerative cartilage disease: comparison of high-resolution morphological MR and quantitative T2 mapping at 3.0 Tesla. Osteoarthritis Cartilage 18:1211–1217. https://doi.org/10.1016/j.joca.2010.06.002
Su F, Hilton JF, Nardo L, Wu S, Liang F, Link TM, Ma CB, Li X (2013) Cartilage morphology and T1ρ and T2 quantification in ACL-reconstructed knees: a 2-year follow-up. Osteoarthritis Cartilage 21:1058–1067. https://doi.org/10.1016/j.joca.2013.05.010
Li X, Kuo D, Theologis A, Carballido-Gamio J, Stehling C, Link TM, Ma CB, Majumdar S (2011) Cartilage in anterior cruciate ligament-reconstructed knees: MR imaging T1{rho} and T2–initial experience with 1-year follow-up. Radiology 258:505–514. https://doi.org/10.1148/radiol.10101006
Osteoarthritis—PubMed, (n.d.). https://pubmed.ncbi.nlm.nih.gov/31034380/. Accessed June 26, 2023.
Slattery C, Kweon CY (2018) Classifications in brief: outerbridge classification of chondral lesions. Clin Orthop 476:2101–2104. https://doi.org/10.1007/s11999.0000000000000255
Zhao H, Li H, Liang S, Wang X, Yang F (2022) T2 mapping for knee cartilage degeneration in young patients with mild symptoms. BMC Med Imaging 22:72. https://doi.org/10.1186/s12880-022-00799-1
Ardern CL, Webster KE, Taylor NF, Feller JA (2011) Return to sport following anterior cruciate ligament reconstruction surgery: a systematic review and meta-analysis of the state of play. Br J Sports Med 45:596–606. https://doi.org/10.1136/bjsm.2010.076364
Slauterbeck JR, Kousa P, Clifton BC, Naud S, Tourville TW, Johnson RJ, Beynnon BD (2009) Geographic mapping of meniscus and cartilage lesions associated with anterior cruciate ligament injuries. J Bone Joint Surg Am 91:2094–2103. https://doi.org/10.2106/JBJS.H.00888
Leeuw M, Goossens MEJB, Linton SJ, Crombez G, Boersma K, Vlaeyen JWS (2007) The fear-avoidance model of musculoskeletal pain: current state of scientific evidence. J Behav Med 30:77–94. https://doi.org/10.1007/s10865-006-9085-0
Vlaeyen JWS, Kole-Snijders AMJ, Boeren RGB, van Eek H (1995) Fear of movement/(re)injury in chronic low back pain and its relation to behavioral performance. Pain 62:363–372. https://doi.org/10.1016/0304-3959(94)00279-N
Funding
Open access funding provided by Università degli Studi del Piemonte Orientale Amedeo Avogrado within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
D.Z. had the idea for the article. All authors performed the literature search. G.G., M.L. and M.P enrolled the patients and received the self-reported questionnaires. I.P. and A.C. did the image analysis. M.L and A.C. overviewed the study. P.C. contributed and coordinated the first draft. F.A. revised all the tables and figures. D.Z. and P.C. revised the last draft. All authors agreed on the submission of the last draft.
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript. This study was funded by “Fondo di Ateneo per la Ricerca” of Università del Piemonte Orientale.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zagaria, D., Costantini, P., Percivale, I. et al. Early patello-femoral condropathy assessment through quantitative analyses via T2 mapping magnetic resonance after anterior cruciate ligament reconstruction. Radiol med 128, 1415–1422 (2023). https://doi.org/10.1007/s11547-023-01716-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-023-01716-4