Abstract
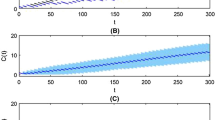
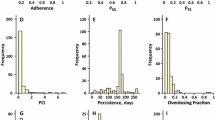
Poor drug adherence is considered one of major barriers to achieving the clinical and public health benefits of many pharmacotherapies. In the current paper, we aim to investigate the impact of dose omission on the plasma concentrations of two-compartment pharmacokinetic models with two typical routes of drug administration, namely the intravenous bolus and extravascular first-order absorption. First, we reformulate the classical two-compartment pharmacokinetic models with a new stochastic feature, where a binomial random model of dose intake is integrated. Then, we formalize the explicit expressions of expectation and variance for trough concentrations and limit concentrations, with the latter proved of the existence and uniqueness for steady-state distribution. Moreover, we mathematically demonstrate the strict stationarity and ergodicity of trough concentrations as a Markov chain. In addition, we numerically simulate the impact of drug non-adherence to different extents on the variability and regularity of drug concentration and compare the drug pharmacokinetic preference between one and two compartment pharmacokinetic models. The results of sensitivity analysis also suggest the drug non-adherence as one of the most sensitive model parameters to the expectation of limit concentration. Our modelling and analytical approach can be integrated into the chronic disease models to estimate or quantitatively predict the therapy efficacy with drug pharmacokinetics presumably affected by random dose omissions.












Similar content being viewed by others
References
Abramson F (1980) Two-compartment pharmacokinetic models: computer simulations of their characteristics and clinical consequences. J Pharm Sci 70(2):141–146
Bouwman L, Eeltink CM, Visser O et al (2017) Prevalence and associated factors of medication non-adherence in hematological-oncological patients in their home situation. BMC Cancer 17(1):1–8
Brown MT, Bussell JK (2011) Medication adherence: Who cares? Mayo Clin Proc 86(4):304–314
Burnier M (2019) Is there a threshold for medication adherence? Lessons learnt from electronic monitoring of drug adherence. Front Pharmacol 9:1540
Chan A, Cooper V, Lycett H et al (2020) Practical barriers to medication adherence: What do current self- or observer-reported instruments assess? Front Pharmacol 11:1–11
Counterman ED, Lawley SD (2021) Designing drug regimens that mitigate nonadherence. Bull Math Biol 84(1):1–36
Counterman ED, Lawley SD (2021) What should patients do if they miss a dose of medication? A theoretical approach. J Pharmacokinet Pharmacodyn 48(5):873–892
Darling R (1989) Infinite-dimensional stochastic difference equations for particle systems and network flows. J Appl Probab 26(2):325–344
Donnet S, Samson A (2013) A review on estimation of stochastic differential equations for pharmacokinetic/pharmacodynamic models. Adv Drug Deliv Rev 65(7):929–939
Fermín LJ, Lévy-Véhel J (2017) Variability and singularity arising from poor compliance in a pharmacodynamical model ii: The multi-oral case. J Math Biol 74:809–841
Fermín LJ, Lévy-Véhel J (2020) Variability and singularity arising from a piecewise-deterministic markov process applied to model poor patient compliance in the multi-iv case. J Appl Stat 47(13–15):2525–2545
Ferrante L, Bompadre S, Leone L (2003) A stochastic compartmental model with long lasting infusion. Biom J 45:182–194
Foley L, Larkin J (2021) Prevalence and predictors of medication non-adherence among people living with multimorbidity: A systematic review and meta-analysis. BMJ Open 11(9):1–13
Garreau R, Falquet B (2021) Population pharmacokinetics and dosing simulation of vancomycin administered by continuous injection in critically ill patient. Antibiotics 10(10):1–12
Geof G, Jennifer H (2013) Comutational Statictics, 2nd edn. Wiley, London
Gharouni A, Wang L, Watmough J (2017) Stochastic dispersal increases the rate of upstream spread: a case study with green crabs on the northwest atlantic coast. PLoS One 12(9):1–17
Gibaldi M, Perrier D (2007) Pharmacokinetics, 2nd edn. Informa Healthcare, New York
Goto A, Abe S, Koshiba S (2019) Current status and future perspective on preclinical pharmacokinetic and pharmacodynamic (pk/pd) analysis: survey in japan pharmaceutical manufacturers association (jpma). Drug Metab Pharmacokinet 34(2):148–154
Grabowski T, Reijers J, Burmańczuk A et al (2019) Fluctuations in pharmacokinetics profiles of monoclonal antibodies. Eur J Drug Metab Pharmacokinet 44(8):585–589
Hardwick RJ, Truscott JE, Oswald WE (2021) Individual adherence to mass drug administration in neglected tropical disease control: a probability model conditional on past behaviour. PLoS Negl Trop Dis 15(1):1–15
Harry K, Frank S (1984) Convergence in distribution of products of random matrices. Probab Theory Relat Fields 67(4):363–386
Jimmy B, Jose J (2011) Patient medication adherence: measures in daily practice. Oman Med J 26(3):155–159
Kiang TK, Sherwin CM, Spigarelli MG (2012) Fundamentals of population pharmacokinetic modelling : modelling and software. Clin Pharmacokinet 51(8):515–525
Lévy-Véhel J, Lévy-Véhel PE (2013) Variability and singularity arising from poor compliance in a pharmacodynamical model i: the multi-iv case. J Pharmacokinet Pharmacodyn 40(1):15–39
Li J, Nekka F (2007) A pharmacokinetic formalism explicitly integrating the patient drug compliance. J Pharmacokinet Pharmacodyn 34(1):115–139
Liu J, Sun J, Zhang N (2016) Biopharmaceutics and Pharmacokinetics, 5th edn. People’s Medical Publishing House, Beijing
Liu Y, He D, Wang W (2021) An analysis on the trend of aids/hiv incidence in chongqing and shenzhen, china from 2005–2015 based on age-period-cohort model. Math Biosci Eng 18(5):6961–6977
Mahmood S, Jalal Z, Hadi AM (2021) Prevalence of non-adherence to antihypertensive medication in asia: a systematic review and meta-analysis. Int J Clin Pharm 43(9859):486–501
Makovec U, Locatelli I, Kos M (2021) Improved adherence with medicines use review service in slovenia: a randomized controlled trial. BMC Health Serv Res 21(1):1–12
Marione S, Hogue I, Ray C (2008) A methodology for performing global uncertainty and sensitivity analysis in systems biology. J Theor Biol 254(1):178–196
McAllister N, Lawley S (2022) A pharmacokinetic and pharmacodynamic analysis of drug forgiveness. J Pharmacokinet Pharmacodyn 49(5):363–379
Mehdi B (2015) Pharmacokinetics and Toxicokinetics. CRC Press, Taylor & Francis Group
Osterberg L, Blaschke T (2005) Adherence to medicine. N Engl J Med 353:487–497
Patricia B, Eduardo S (2003) Adherence to long-term therapies: evidence for action. J Nurs Scholarsh 35(3):207
Simon L, Kanneganti K, Kim KS (2010) Drug transport and pharmacokinetics for chemical engineers. Chem Eng Edu 44(4):262–266
Tang SY, Xiao YN, Liang JH et al (2019) Mathematical biology. Science Press, Beijing
Wu XT, Nekka F, Li J (2019) Analytical solution and exposure analysis of a pharmacokinetic model with simultaneous elimination pathways and endogenous production: The case of multiple dosing administration. Bull Math Biol 81(12):3436–3459
Yan DD, Wu XT, Tang SY (2022) Statistical analysis of one-compartment pharmacokinetic models with drug adherence. J Pharmacokinet Pharmacodyn 49(3):209–225
Zhang B, Shang H (2014) Applied stochastic processes, 3rd edn. China Renmin University Press, Beijing
Acknowledgements
The authors thank the financial support by National Natural Science Foundation of China (Nos. 12031010, 12271346, 12071300). We would also like to thank two anonymous reviewers for their helpful and insightful comments which lead to the high improvement of the article’s quality.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Data Availability
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A: Derivation of Eqs. (59)
Appendix A: Derivation of Eqs. (59)
We claim the relationships (Eqs. (59)) in the case of IV bolus administrations, the similar procedure can be used for the case of first-order absorption. The one-compartment IV bolus model is
In order to obtain indistinguishable models, we assume that \({\widetilde{C}}(t)=C_1(t)\) for all time \(t\ge 0\). Firstly, the equation \({\widetilde{C}}(0^+)=C_1(0^+)\) directly implies \(D/\widetilde{V_1}=D/V_1\), which means
Since the functions of \({\widetilde{C}}(t)\) and \(C_1(t)\) are infinite differentiable at the right hand side of time zero, then we have \({\widetilde{C}}'(0^+)=C'_1(0^+)\) which is equivalent to
This implies
because of \(C_2(0^+)=0\).
Taking derivatives of \({\widetilde{C}}(t)\) and \(C'_1(t)\) with respect to time t give rise to
and
Substituting \(t=0^+\) into above two equations and using the identity \({\widetilde{C}}''(0^+)=C''_1(0^+)\) yields
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, D., Wu, X., Li, J. et al. Statistical Analysis of Two-Compartment Pharmacokinetic Models with Drug Non-adherence. Bull Math Biol 85, 65 (2023). https://doi.org/10.1007/s11538-023-01173-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-023-01173-0
Keywords
- Two-compartment pharmacokinetic model
- Drug non-adherence
- Stochastic differential equations
- Limit distribution
- Variability and regularity