Abstract
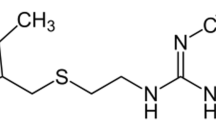
Metformin (MF) is one of the most important medicaments in the market and has been extensively employed in treating type 2 diabetes. In this work, we have observed that, because of its positive charge, MF interacts with negatively charged gold nanoparticles, leading to agglomeration even at low concentrations (< 0.01 mmol L−1). This is accompanied by the rise of a plasmon coupling band at 645 nm, allowing its colorimetric monitoring with a limit of detection, LOD, of 1.9 µmol L−1. However, above 0.01 mmol L−1, aggregation takes place, shifting the plasmonic band to 700 nm. Consequently, in this range of concentration, the optical correlation departs from that observed in the agglomeration regime. Therefore, for a critical evaluation, a systematic monitoring of the spectral changes is required to differentiate between the agglomeration and aggregation regimes, as reported in this work. The interaction of metformin with gold nanoparticles has also been monitored by Raman spectroscopy, through the SERS effect. The large enhancement of the Raman signals promoted by the plasmonic nanoparticles improved the detection limit to 0.093 µmol L−1. While monitoring the plasmonic band has inherently a low specificity, the Raman technique provides an unequivocal detection of metformin, based on its characteristic vibrational profiles.








Similar content being viewed by others
Data Availability
No datasets were generated or analyzed during the current study.
References
Ungurianu A, Seremet O, Gagniuc E et al (2019) Preclinical and clinical results regarding the effects of a plant-based antidiabetic formulation versus well-established antidiabetic molecules. Pharmacol Res 150:104522. https://doi.org/10.1016/j.phrs.2019.104522
Ambrosio-Albuquerque EP, Cusioli LF, Bergamasco R et al (2021) Metformin environmental exposure: a systematic review. Environ Toxicol Pharmacol 83:103588. https://doi.org/10.1016/j.etap.2021.103588
Foretz M, Guigas B, Viollet B (2023) Metformin: update on mechanisms of action and repurposing potential. Nat Rev Endocrinol 19:460–476. https://doi.org/10.1038/s41574-023-00833-4
Bailey CJ (2017) Metformin: historical overview. Diabetologia 60:1566–1576. https://doi.org/10.1007/s00125-017-4318-z
Shurrab NT, Arafa ESA (2020) Metformin: a review of its therapeutic efficacy and adverse effects. Obes Med 17:100186. https://doi.org/10.1016/j.obmed.2020.100186
Balakrishnan A, Sillanpää M, Jacob MM, Vo DVN (2022) Metformin as an emerging concern in wastewater: occurrence, analysis and treatment methods. Environ Res. https://doi.org/10.1016/j.envres.2022.113613
Shahabadi N, Heidari L (2014) Synthesis, characterization and multi-spectroscopic DNA interaction studies of a new platinum complex containing the drug metformin. Spectrochim Acta - Part Mol Biomol Spectrosc 128:377–385. https://doi.org/10.1016/j.saa.2014.02.167
Gopalakrishnan D, Ganeshpandian M, Loganathan R et al (2017) Water soluble Ru(II)-arene complexes of the antidiabetic drug metformin: DNA and protein binding, molecular docking, cytotoxicity, and apoptosis-inducing activity. RSC Adv 7:37706–37719. https://doi.org/10.1039/c7ra06514k
Pundir CS, Deswal R, Narwal V, Narang J (2017) Quantitative analysis of metformin with special emphasis on sensors: a review. Curr Anal Chem 14:438–445. https://doi.org/10.2174/1573411013666170907150509
Tallam AK, Sahithi A, Nuli MV (2023) A review of analytical techniques for determination of metformin: present and perspectives. Int J Heal Care Biol Sci 4:18–24
Turkevich J, Stevenson PC, Hillier J (1951) A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss Faraday Soc 11:55–75
Frens G (1973) Controlled nucleation for regulation of particle-size in monodisperse gold suspensions. Nat Phys Sci 241:20–22
Vainrub A, Pustovyy O, Vodyanoy V (2006) Resolution of 90 nm (λ/5) in an optical transmission microscope with an annular condenser. Opt Lett 31:2855. https://doi.org/10.1364/OL.31.002855
Kromidas S (2011) Validation in Analytics. Wiley, VCH, Weinheim, Germany
Grasseschi D, Toma HE (2017) The SERS effect in coordination chemistry. Coord Chem Rev 333:108–131. https://doi.org/10.1016/j.ccr.2016.11.019
Mie G (1908) Colloidal solutions. Anallen Der Phys 25:377–445
Aravind PK, Nitzan A, Metiu H (1981) The interaction between electromagnetic resonances and its role in spectroscopic studies of molecules adsorbed on colloidal particles or metal spheres. Surf Sci 110:189–204. https://doi.org/10.1016/0039-6028(81)90595-1
Aravind PK, Metiu H (1982) Use of a perfectly conducting sphere to excite the plasmon of a flat surface. 1. Calculation of the local field with applications to surface-enhanced spectroscopy. J Phys Chem 86:5076–5084. https://doi.org/10.1021/j100223a007
Aravind PK, Metiu H (1983) The effects of the interaction between resonances in the electromagnetic response and sphere-plane structure applications to surface-enhanced spectroscopy. Surf Sci 124:506–528. https://doi.org/10.1016/0039-6028(83)90806-3
Jain PK, El-Sayed MA (2010) Plasmonic coupling in noble metal nanostructures. Chem Phys Lett 487:153–164. https://doi.org/10.1016/j.cplett.2010.01.062
Quinten M, Kreibig U (1986) Optical properties of aggregates of small metal particles. Surf Sci 172:557–577. https://doi.org/10.1016/0039-6028(86)90501-7
Halas NJ, Lal S, Chang W-S et al (2011) Plasmons in strongly coupled metallic nanostructures. Chem Rev 111:3913–3961. https://doi.org/10.1021/cr200061k
Ghosh SK, Pal T (2007) Interparticle coupling effect on the surface plasmon resonance of gold nanoparticles: from theory to applications. Chem Rev 107:4797–4862. https://doi.org/10.1021/cr0680282
Fleischmann M, Hendra PJ, McQuillan AJ (1974) Raman Spectra of pyridine adsorbed at a silver electrode. Chem Phys Lett 26:163–166. https://doi.org/10.1016/0009-2614(74)85388-1
Albrecht AC (1961) Theory of Raman intensities. J Chem Phys 34:1476
Albrecht MG, Creighton JA (1977) Anomalously intense Raman spectra of pyridine at a silver electrode. J Am Chem Soc 99:5215–5217
Jeanmaire DL, Van Duyne RP (1977) Surface Raman spectroelectrochemistry. J Electroanal Chem Interfacial Electrochem 84:1–20. https://doi.org/10.1016/S0022-0728(77)80224-6
Aroca RF (2006) Surface-enhanced vibrational spectroscopy. John Wiley & Sons, Ltd., Chichester
Lombardi JR, Birke RL (2012) The theory of surface-enhanced Raman scattering. J Chem Phys 136:144704. https://doi.org/10.1063/1.3698292
Lombardi JR, Birke RL (2009) A unified view of surface-enhanced Raman Scattering. Acc Chem Res 42:734–742
Zamarion VM, Timm RA, Araki K, Toma HE (2008) Ultrasensitive SERS Nanoprobes for Hazardous Metal ions based on trimercaptotriazine-modified gold nanoparticles. Inorg Chem 47:2934–2936. https://doi.org/10.1021/ic800122v
Grasseschi D, Parussulo ALA, Zamarion VM et al (2013) How relevant can the SERS effect in isolated nanoparticles be? RSC Adv. https://doi.org/10.1039/c3ra41818a
Grasseschi D, Parussulo ALA, Zamarion VM et al (2014) SERS studies of isolated and agglomerated gold nanoparticles functionalized with a dicarboxybipyridine-trimercaptotriazine-ruthenium dye. J Raman Spectrosc 45:758–763. https://doi.org/10.1002/jrs.4562
Grasseschi D, Zamarion VM, Toma HE (2018) Probing the dynamics of dithiooxamide coordinated to gold nanoparticles using SERS. J Raman Spectrosc 49:1478–1486. https://doi.org/10.1002/jrs.5398
Assumpcao AMC, Bonacin JA, Toma SH et al (2014) Probing surface – complex interactions with the bis(4-thienylterpyridine)iron(II) complex anchored on TiO2 and gold nanoparticles. Can J Chem 92:918–924. https://doi.org/10.1139/cjc-2014-0025
Yang Y (2017) SERS enhancement dependence on the diameter of au nanoparticles. J Phys Conf Ser. https://doi.org/10.1088/1742-6596/844/1/012030
Sacco A, Mangino S, Portesi C et al (2019) Novel approaches in tip-enhanced Raman Spectroscopy: Accurate Measurement of Enhancement factors and Pesticide Detection in Tip Dimer configuration. J Phys Chem C 123:24723–24730. https://doi.org/10.1021/acs.jpcc.9b07016
He S, Chua J, Tan EKM, Kah JCY (2017) Optimizing the SERS enhancement of a facile gold nanostar immobilized paper-based SERS substrate. RSC Adv 7:16264–16272. https://doi.org/10.1039/c6ra28450g
Funding
The financial support from FAPESP – Fundação de Amparo à Pesquisa do Estado de São Paulo, grant 2018/21489-1, is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
M.D. Ramos Jr, A. L. Hennemann, and L. M. Sihn performed the analytical and spectrophotometric measurements, M. Nakamura carried out the CytoViva study, K. Araki and H. E. Toma were responsible for conceptualization, Raman investigation, and writing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hennemann, A.L., Ramos, M.D., Sihn, L.M. et al. Plasmonic Interaction of Gold Nanoparticles with the Anti-hypoglycemic Medicament Metformin. Plasmonics (2024). https://doi.org/10.1007/s11468-024-02341-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11468-024-02341-1