Abstract
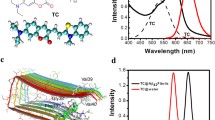
Amyloid-beta proteins that form cytotoxic aggregates called amyloid-β derived diffusible ligands are responsible for various neurodegenerative diseases like Alzheimer’s and Parkinson’s disease. Novel methods for the early detection of such aggregates and inhibition of further fibrillation are highly important and need in the current situation. In this paper, we present a novel method based on fluorescence resonance energy transfer (FRET) between carbon dots and Ag nanoparticle for sensing various fibrillation stages of beta-amyloid proteins. The addition of Ag nanoparticles to carbon dot colloid is found to significantly enhance the inhibition of beta-amyloid fibrillation due to the modified hydrophobic and electrostatic interactions introduced by Ag nanoparticles and is monitored using thioflavin T (ThT) assay. Further, fluorescence quenching of carbon dots in the presence of Ag particles is found to get reduced with the increase in the incubation time of beta-amyloid fibrils. We could observe a linear trend in the variation of Stern–Volmer constants calculated based on FRET between carbon dots and Ag nanoparticles with the incubation time of beta-amyloid, indicating the potential of using the proposed FRET-based method for sensing beta-amyloid fibrillation.






Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in the published article (and its supplementary information files).
References
Guo S et al (2016) Hydrothermal synthesis of nitrogen-doped carbon dots with real-time live-cell imaging and blood–brain barrier penetration capabilities. Int J Nanomedicine 11:6325–6336
Zheng M et al (2015) Self-targeting fluorescent carbon dots for diagnosis of brain cancer cells. ACS Nano 9:11455–11461
Kim D et al (2018) Graphene quantum dots prevent α-synucleinopathy in Parkinson’s disease. Nat Nanotechnol 13:812–818
Wen Y et al (2015) Graphene quantum dots for the inhibition of β amyloid aggregation. Nanoscale 7:19060–19065
Huang H et al (2017) Graphene quantum dots for detecting monomeric amyloid peptides. Nanoscale 9:5044–5048
Chung YJ, Kim K, Lee B. Il, Park CB (2017) Carbon Nanodot-sensitized modulation of Alzheimer’s β-amyloid self-assembly, disassembly, and toxicity. Small 13:1–9
Ogomori K et al (1989) Beta-protein amyloid is widely distributed in the central nervous system of patients with Alzheimer’s disease. Am J Pathol 134:243–251
William LK (2002) Aβ toxicity in Alzheimer’s disease: globular oligomers (ADDLs) as new vaccine and drug targets. Neurochem Int 41:345–352
Klein WL (2006) Synaptic targeting by Aβ oligomers (ADDLS) as a basis for memory loss in early Alzheimer’s disease. Alzheimer’s Dement 2:43–55
Krafft GA, Klein WL (2010) ADDLs and the signaling web that leads to Alzheimer’s disease. Neuropharmacology 59:230–242
Apelt J, Bigl M, Wunderlich P, Schliebs R (2004) Aging-related increase in oxidative stress correlates with developmental pattern of beta-secretase activity and beta-amyloid plaque formation in transgenic Tg2576 mice with Alzheimer-like pathology. Int J Dev Neurosci 22:475–484
MacLeod R, Hillert EK, Cameron RT, Baillie GS (2015) The role and therapeutic targeting of α-, β- and γ-secretase in Alzheimer’s disease. Futur Sci OA 1(3), FS011:1–16
Murphy MP, LeVine H III (2010) Alzheimer’s disease and the β-amyloid peptide. J Alzheimer’s Dis 19:1–17
Bature F, Guinn BA, Pang D, Pappas Y (2017) Signs and symptoms preceding the diagnosis of Alzheimer’s disease: a systematic scoping review of literature from 1937 to 2016. BMJ Open 7, e015746:1–9
Perinchery SM et al (2016) High resolution iridocorneal angle imaging system by axicon lens assisted gonioscopy. Sci Rep 6:1–10
Shinoj VK, Murukeshan VM, Baskaran M, Aung T (2014) Note: A gel based imaging technique of the iridocorneal angle for evaluation of angle-closure glaucoma. Rev Sci Instrum 85:2–5
Haes AJ, Chang L, Klein WL, Van Duyne RP (2005) Detection of a biomarker for Alzheimer’s disease from synthetic and clinical samples using a nanoscale optical biosensor. J Am Chem Soc 127:2264–2271
Queiroz L, Nucci A, Facure NO, Facure JJ (1979) Massive spinal cord necrosis in schistosomiasis. Arch Neurol 36:517–519
Georganopoulou DG et al (2005) From The Cover: Nanoparticle-based detection in cerebral spinal fluid of a soluble pathogenic biomarker for Alzheimer’s disease. Proc Natl Acad Sci 102:2273–2276
Sharma N, Singh AN (2016) Exploring biomarkers for Alzheimer’s disease. J Clin Diagnostic Res 10:KE01–KE06
Lancioni GE et al (2011) Persons with acquired brain injury and multiple disabilities access stimulation independently through microswitch-based technology. Percept Mot Skills 111:485–495
Cuttler JM, Moore ER, Hosfeld VD, Nadolski DL (2016) Treatment of Alzheimer disease with CT scans: a case report. Dose-Response 14:1–7
Chandra A, Dervenoulas G, Politis M (2019) Magnetic resonance imaging in Alzheimer’s disease and mild cognitive impairment. J Neurol 266(6):1293-1302
Marcus C, Mena E, Subramaniam RM (2014) Brain PET in the diagnosis of Alzheimer’s disease. Clin Nucl Med 39:e413–e426
Hane F, Leonenko Z (2014) Effect of metals on kinetic pathways of amyloid-β aggregation. Biomolecules 4:101–116
Gladytz A, Wagner M, Häupl T, Elsner C, Abel B (2015) Structure-making effects of metal nanoparticles in amyloid peptide fibrillation. Part Part Syst Charact 32:573–582
Goadsby PJ, Kurth T, Pressman A (2016) HHS Public Access 35:1252–1260
Nair RV, Murukeshan VM (2020) ( Cu 2 O-Au ) – Graphene - Au layered structures as efficient near Infra - Red SERS substrates. Sci Rep 10:1–10. https://doi.org/10.1038/s41598-020-60874-x
Xia Y, Padmanabhan P, Sarangapani S, Gulyás B, Vadakke Matham M (2019) Bifunctional fluorescent/Raman nanoprobe for the early detection of amyloid. Sci Rep 9:1–11
Zhao J, Nussinov R, Ma B (2017) Mechanisms of recognition of amyloid-β (Aβ) monomer, oligomer, and fibril by homologous antibodies. J Biol Chem 292:18325–18343
Yun S et al (2007) Role of electrostatic interactions in amyloid β-protein (Aβ) oligomer formation: a discrete molecular dynamics study. Biophys J 92:4064–4077
Velander P et al (2017) Natural product-based amyloid inhibitors HHS Public Access Author manuscript. Biochem Pharmacol 139:40–55
Nie Q, Du XG, Geng MY (2011) Small molecule inhibitors of amyloid β peptide aggregation as a potential therapeutic strategy for Alzheimer’s disease. Acta Pharmacol Sin 32:545–551
Morelli L et al (2004) β-sheet breaker peptides inhibit fibrillogenesis in a rat brain model of amyloidosis: implications for Alzheimer’s therapy. Nat Med 4:822–826
Solomon B, Koppel R, Hanan E, Katzav T (2002) Monoclonal antibodies inhibit in vitro fibrillar aggregation of the Alzheimer beta-amyloid peptide. Proc Natl Acad Sci 93:452–455
Feng BY et al (2009) NIH Public Access 4:197–199
Sun Y, Qian Z, Wei G (2016) The inhibitory mechanism of a fullerene derivative against amyloid-β peptide aggregation: an atomistic simulation study. Phys Chem Chem Phys 18:12582–12591
Magdysyuk OV, Adams F, Liermann HP, Spanopoulos I, Trikalitis PN, Hirscher M, Morris RE, Duncan MJ, McCormick LJ, Dinnebier RE (2014) Understanding the adsorption mechanism of noble gases Kr and Xe in CPO-27-Ni, CPO-27-Mg, and ZIF-8. Phys Chem Chem Phys 16:23908–23914
Li H, Luo Y, Derreumaux P, Wei G (2011) Carbon nanotube inhibits the formation of β-sheet-rich oligomers of the Alzheimer’s amyloid-β(16–22) peptide. Biophys J 101:2267–2276
Kim JE, Lee M (2003) Fullerene inhibits β-amyloid peptide aggregation. Biochem Biophys Res Commun 303:576–579
Lindberg DJ, Wranne MS, Gilbert Gatty M, Westerlund F, Esbjörner EK (2015) Steady-state and time-resolved Thioflavin-T fluorescence can report on morphological differences in amyloid fibrils formed by Aβ(1–40) and Aβ(1–42). Biochem Biophys Res Commun 458:418–423
Zielińska A, Skwarek E, Zaleska A, Gazda M, Hupka J (2009) Preparation of silver nanoparticles with controlled particle size. Procedia Chem 1:1560–1566
Chou KF, Dennis AM (2015) Förster resonance energy transfer between quantum dot donors and quantum dot acceptors. Sensors (Switzerland) 15:13288–13325
Liu H et al (2016) Negatively charged hydrophobic nanoparticles inhibit amyloid β-protein fibrillation: the presence of an optimal charge density. React Funct Polym 103:108–116
Funding
The authors acknowledge NTU, COLE-EDB, and Ministry of Education, MoE Singapore (RG 192/17) for financial support. B.G. and P.P. thank the support from Lee Kong Chian School of Medicine Imaging Probe Development Platform and the support from the Cognitive Neuro Imaging Centre (CONIC) at NTU.
Author information
Authors and Affiliations
Contributions
R.V.N along with V.M.M. conducted the research presented in this manuscript. R.V.N, P.P., and B.G. performed beta-amyloid fibrillation experiments. All authors prepared, corrected, and finalized the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nair, R.V., Padmanabhan, P., Gulyás, B. et al. Fluorescence Resonance Energy Transfer (FRET)-Based ThT Free Sensing of Beta-Amyloid Fibrillation by Carbon Dot-Ag Composites. Plasmonics 16, 863–872 (2021). https://doi.org/10.1007/s11468-020-01338-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11468-020-01338-w