Abstract
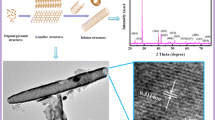
Antimony oxychloride Sb8O11Cl2(H2O)6 products with various morphologies including sheaf-like, rhombic-plate, oval leaf-like and quasi-wafer have been successfully synthesized via a mild and facile solution route at room temperature. The morphologies and structures of the as-prepared samples were characterized by X-ray powder diffraction (XRD), scanning electron microscopy (SEM) and transmission electron microscopy (TEM). A possible formation mechanism of these structures is proposed according to the experimental results and analysis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Min Y, Akbulut M, Kristiansen K, et al. The role of interparticle and external forces in nanoparticle assembly. Nat Mater, 2008, 7: 527–538
Wang D, Lieber C M. Inorganic materials: Nanocrystals branch out. Nat Mater, 2003, 2: 355–356
Nguyen T D, Dinh C T, Do T O. Shape- and size-controlled synthesis of monoclinic ErOOH and cubic Er2O3 from micro- to nanostructures and their upconversion luminescence. ACS Nano, 2010, 4: 2263–2273
Jang S Y, Song Y M, Kim H S, et al. Three synthetic routes to singlecrystalline PbS nanowires with controlled growth direction and their electrical transport properties. ACS Nano, 2010, 4: 2391–2401
Gao J H, Gu H W, Xu B. Multifunctional magnetic nanoparticles: Design, synthesis, and biomedical applications. Acc Chem Res, 2009, 42: 1097–1107
Xiong Y J, Wiley B J, Xia Y N. Nanocrystals with unconventional shapes-A class of promising catalysts. Angew Chem Int Ed, 2007, 46: 7157–7159
Lu X B, Wen Z H, Li J H. Hydroxyl-containing antimony oxide bromide nanorods combined with chitosan for biosensors. Biomaterials, 2006, 27: 5740–5747
Li J H, Zhang J Z. Optical property and applications of hybrid semiconductor nanomaterials. Coordin Chem Rev, 2009, 253: 3015–3041
Costa L, Paganetto G, Bertelli G, et al. Thermal decomposition of antimony oxyhalides: I. Oxychlorides. J Therm Anal, 1990, 36: 1141–1153
Chen X Y, Huh H S, Lee S W. Hydrothermal synthesis of antimony oxychloride and oxide nanocrystals: Sb4O5Cl2, Sb8O11Cl2, and Sb2O3. J Solid State Chem, 2009, 181: 2127–2132
Tang J J, Wang Y, Jiao Z, et al. Self-assembly nanostructures of one-dimensional antimony oxide and oxychloride. Mater Lett, 2009, 63: 1481–1484
Li B J, Zhao Y B, Xu X M, et al. A simple method for the preparation of containing Sb nano- and microcrystallines via an ultrasound agitation. Ultraso Sonochem, 2007, 14: 557–562
Sui Y M, Fu W Y, Yang H B, et al. Low temperature synthesis of Cu2O crystals: Shape evolution and growth mechanism. Cryst Growth Des, 2010, 10: 99–108
Wang H H, Tian F, Li X P, et al. Preparation and shape evolution of cuprous oxide in the solution phases of copper (II) dodecyl sulfate. Powder Technol, 2010, 197: 298–302
Zhou J, Zhao H W, Li L D, et al. One-step synthesis and flame retardancy of sheaf-like microcrystal antimony oxychloride. J Nanosci Nanotech, 2011, doi: 10.1166/jnn.2011.4736
Tang J, Alivisatos A P. Crystal splitting in the growth of Bi2S3. Nano Lett, 2006, 6: 2701–2706
Li L S, Sun N J, Huang Y Y, et al. Topotactic transformation of single-crystalline precursor discs into disc-Like Bi2S3 nanorod networks. Adv Funct Mater, 2008, 18: 1194–1201
Tian L, Tan H Y, Vittal J J. Morphology-controlled synthesis of Bi2S3 nanomaterials via singleand multiple-source approaches. Cryst Growth Des, 2008, 8: 734–738
Hu Y H, Chen K Z. Crystal splitting in the growth of β-FeO(OH). J Cryst Growth, 2007, 308: 185–188
Luo Z J, Li H M, Xia J X, et al. Controlled synthesis of different morphologies of BaWO4 crystals via a surfactant-assisted method. J Cryst Growth, 2007, 300: 523–529
He J H, Han M, Shen X P, et al. Crystal hierarchically splitting in growth of BaWO4 in positive cat-anionic microemulsion. J Cryst Growth, 2008, 310: 4581–4586
Deng H, Liu C M, Yang S H, et al. Additive-mediated splitting of lanthanide orthovanadate nanocrystals in water: Morphological evolution from rods to sheaves and to spherulites. Cryst Growth Des, 2008, 8: 4432–4439
Chen G Y, Dneg B, Cai G B, et al. The fractal splitting growth of Sb2S3 and Sb2Se3 hierarchical nanostructures. J Phys Chem C, 2008, 112: 672–679
Ota J, Roy P, Srivastava S K, et al. Morphology evolution of Sb2S3 under hydrothermal conditions: Flowerlike structure to nanorods. Cryst Growth Des, 2008, 8: 2019–2023
Zhang M, Xu X D, Zhang M L. Hydrothermal synthesis of sheaf-like CuO via ionic liquids. Mater Lett, 2008, 62: 385–388
Pan Q T, Huang K, Ni S B, et al. Synthesis of sheaf-like CuO from aqueous solution and their application in lithium-ion batteries. J Alloys Comp, 2009, 484: 322–326
Kim S, Lee J S, Mitterbauer C, et al. Anomalous electrical conductivity of nanosheaves of CeO2. Chem Mater, 2009, 21: 1182–1186
Zhang F, Wong S S. Ambient large-scale template-mediated synthesis of high-aspect ratio single-crystalline, chemically doped rare-earth phosphate nanowires for bioimaging. ACS Nano, 2010, 4: 99–112
Liu K, You H P, Jia G, et al. Hierarchically nanostructured coordination polymer: Facile and rapid fabrication and tunable morphologies. Cryst Growth Des, 2010, 10: 790–797
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhou, J., Zhao, H., Li, L. et al. Shape evolution of antimony oxychloride from sheaf-like to quasi-wafer structures. Chin. Sci. Bull. 56, 3817–3822 (2011). https://doi.org/10.1007/s11434-011-4735-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-011-4735-1